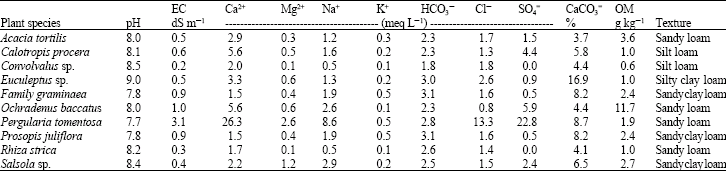
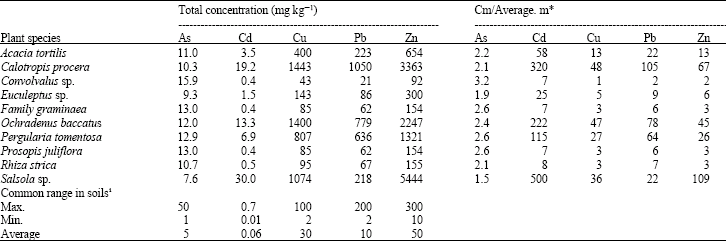
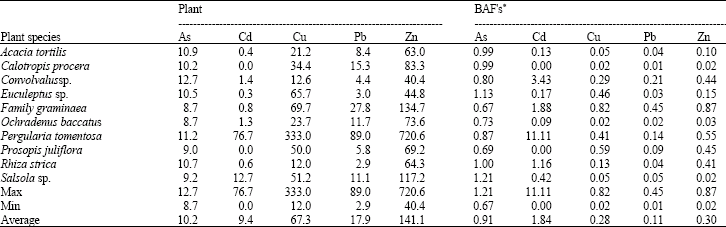
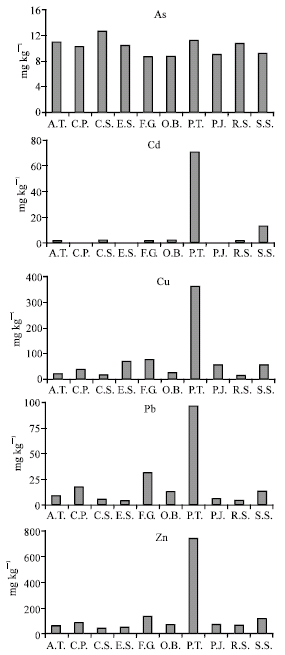
Research Article
Heavy Metals Accumulation of Some Plant Species Grown on Mining Area at Mahad AD’Dahab, Saudi Arabia
Department of Soil Science, College of Food and Agricultural, King Saud University, Riyadh 11451, P.O. Box 2460, Saudi Arabia
M.I. Al-Wabel
Department of Soil Science, College of Food and Agricultural, King Saud University, Riyadh 11451, P.O. Box 2460, Saudi Arabia