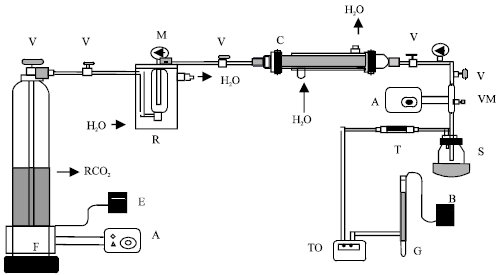
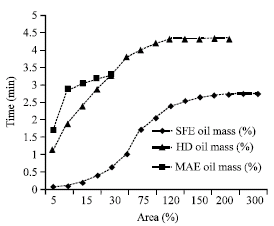
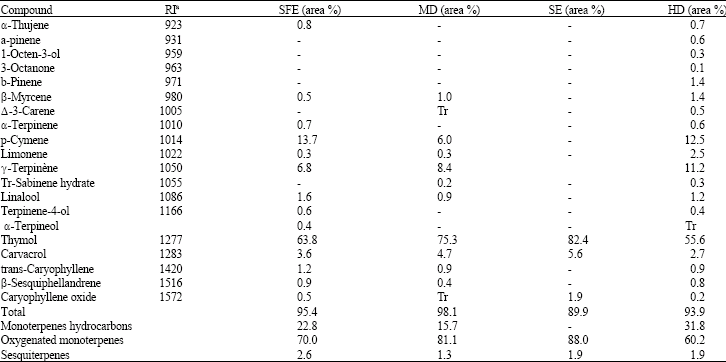
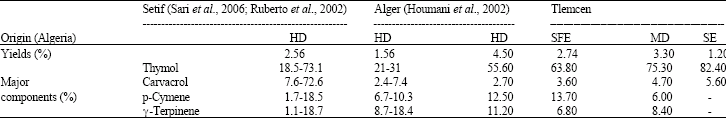
Research Article
Influence of the Processes Extraction on Essential Oil of Origanum glandulosum Desf
Department of Biology: Laboratory COSNA, University Aboubekr Belka�l of Tlemcen, Algeria
M. Benyoucef
Faculty of Medicine, University Aboubekr Belka�l of Tlemcen, Algeria
D. Benkada
Departemento de Engenharia Quimica-Universidade Federal do Rio Grande-Brazil
M.B.D. Soussa Elisa
Department of Biology: Laboratory COSNA, University Aboubekr Belka�l of Tlemcen, Algeria
E.L. Galvao
Departemento de Engenharia Quimica-Universidade Federal do Rio Grande-Brazil
M.M.O. Marques
Departemento de Engenharia Quimica-Universidade Federal do Rio Grande-Brazil
A. Muselli
Departemento de Engenharia Quimica-Universidade Federal do Rio Grande-Brazil
J.M. Desjobert
University of Corse, Chemical of Naturels Products Group, UMR-CNRS 6134, Quartier Grossetti, BP 52, 20250 Corti, France
A.F. Bernardini
University of Corse, Chemical of Naturels Products Group, UMR-CNRS 6134, Quartier Grossetti, BP 52, 20250 Corti, France
J. Costa
University of Corse, Chemical of Naturels Products Group, UMR-CNRS 6134, Quartier Grossetti, BP 52, 20250 Corti, France