Research Article
Characterization of Essential Oils from Helichrysum odoratissimum Using Different Drying Methods
Not Available
D.S. Grierson
Not Available
A.J. Afolayan
Department of Botany, University of Fort Hare, Alice 5700, South Africa
The members of the genus Helichrysum are mostly aromatic perennial herbs belonging to the family Asteraceae. They are widely distributed in southern Africa with about 245 species. H. odoratissimum (L.) Sweet is one of the most commonly used member of the group for medicinal purposes (Van Wyk et al., 1997). In the Eastern Cape of South Africa some rural dwellers burn the herb and inhale the smoke as a sedative. A decoction of the plant is taken to relieve cough and cold. The plant is also boiled and used as a facial ointment for pimples. In Lesotho, the burnt plant is used to fumigate sick people’s rooms as a repellant against parasitic insects, thus, ensuring a good night rest. (Hutchings and Van Staden, 1994). Scientific evidence for the traditional use of the plant for infections has been reported. (Van Puyvelde, 1989) The essential oil from the plant was reported to have antioxidant activity and also showed marked action against Staphylococcus aureus, Pseudomonas aeruginosa and Proteus vulgaris (Gundidza and Zwaving, 1993).
Published reports on the essential oil composition of H. odoratissimum from some African countries showed the dominance of monoterpenoids, in particular α-pinene, over other components (Gundidza and Zwaving, 1993; Lwande et al., 1993; Kuiate et al., 1999).
It has been reported that drying plant materials before distillation could affect the yield of the oil considerably (Díaz-Maroto et al., 2003; Venskutonis, 1997; Whish and Williams, 1996; Omidbaigi et al., 2004). Oil components are however, lost during drying, therefore, the method of drying is very important. In this research, we report the chemical composition of the essential oils obtained from H. odoratissimum and the effect of drying methods on the yield and quality of the oils.
Plant material: The aerial parts of H. odoratissimum were collected randomly from wild-growing plants in the Eastern Cape Province of South Africa during winter (July, 2005). The species was authenticated in the Department of Botany, University of Fort Hare. The plant samples were used as fresh, air-dried (under normal air at room temperature), sun-dried (maximum temperature 24°C) and oven-dried (40°C).
Essential oil distillation and analysis: The essential oils were obtained by hydrodistillation of plant material (100 g each) using the modified Clevenger-type apparatus as described by Asekun and Ekundayo, 1999. GC-MS analyses were performed on a Hewlett-Packard HP 5973 mass spectrometer interfaced with an HP-6890 gas chromatograph. The following column and temperature conditions were used: initial temp 70°C, equilibration time 3.00 min, ramp 4°C min–1, final temperature 240°C; inlet: split less, initial temperature 220°C, pressure 8.27 psi, purge flow 30 mL min–1, purge time 0.20 min, gas type helium; column: capillary, 30 mx0.25 mm i.d., film thickness 0.25 μm, initial flow 0.7 mL min–1, average velocity 32 cm/sec; MS: EI method at 70 eV.
Identification of components: The components of the oils were identified by comparing their mass spectra data with those of authentic standards held in the computer library (Wiley 275 library, New York). The retention indices of the components were determined in relation to a series of n-alkanes run at the same conditions used for the essential oils for control and also compared with those in literature (Shibamoto, 1987; Adams, 1995).
The yield and chemical compositions of oils distilled from H. odoratissimum were affected by the method of moisture removal from the plant material. Generally, drying the plant material before distillation resulted in a higher essential oil yield. Air-dried plant material had the highest oil content (0.46%), the parts dried in the sun and in the oven at 40°C afforded oil yields of 0.33 and 0.36%, respectively, while the yield of the oil from the fresh aerial part was 0.28%.
A total of 22 compounds were identified in the oil from the fresh aerial material. The air-dried had 24 compounds, sun-dried material was 25 compounds, while 30 compounds were identified from the oven-dried plant (Table 1).
The major compounds of the essential oil from the fresh plant were monoterpene oxides, p-menthone (35.4%), pulegone (34.2%) and 1, 8-cineole (13.0%). However, the oil profile changed in the dried materials to terpene hydrocarbons. The air-dried oil had limonene (22.6%), β-caryophyllene (12.0%), caryophyllene oxide (7.9%) and α-pinene (7.7%) as the major compounds. In the oven-dried oil, limonene (31.6%), β-caryophyllene (13.0%) and α-pinene (10.0%) were the main components, while the sun-dried oil had limonene (25.6%), β-caryophyllene (12.6%), α-pinene (8.2%), viridiflorol (7.2%) and caryophyllene oxide (6.5%) as its main components. The results also showed that the monoterpenoids dominance in the fresh oil (95.6%) was reduced to 35.0, 39.1 and 47.6% in the air-dried, sun-dried and oven-dried plants, respectively. It is noteworthy that the fresh oil consisted mainly of monoterpene oxides (p-menthone, pulegone and 1, 8-cineole), whereas the dried oils contained mainly monoterpene hydrocarbons (limonene and α-pinene) as major compounds. The sesquiterpenoid content in the dried plant was high compared to that from the fresh oil. β-caryophyllene and caryophyllene oxide which were 1.3 and 0.1%, respectively in the fresh oil were increased to between 12.0-13.0% for β-caryophyllene and 4.1-7.9% for caryophyllene oxide in the dried oils.
| Table 1: | Compounds of the essential oils of fresh and variously dried portions of H. odoratissimum |
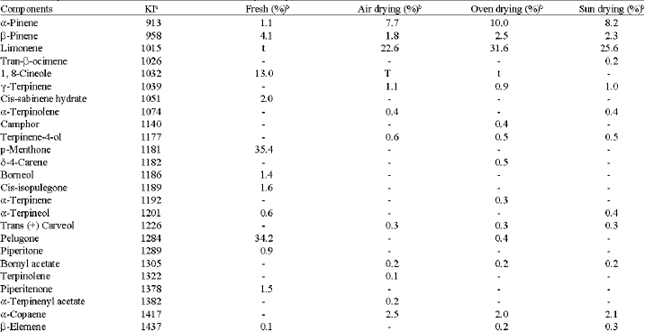 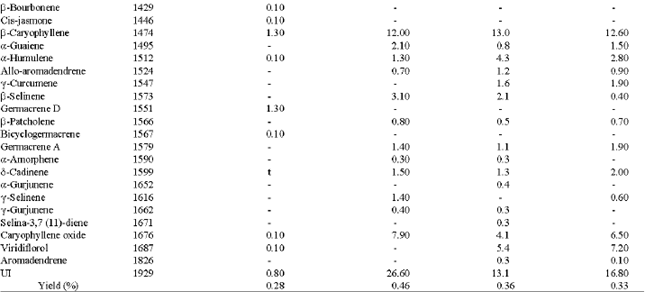 | |
| aKovat Index; t = trace (0.05%); btotal oil content calculated from peak area (values expressed as mean of triplicates); UI = Unidentified; - = Compound not present | |
Menthone and pulegone present in the fresh oil have been reported to be toxic compounds (Gordon et al., 1987; Madsen et al., 1986), hence their complete removal in the air-dried and sun-dried oils makes the oils safer.
The oven-dried oil contained higher quantity of limonene than the rest oils. This compound has been reported to be responsible for the fragrance in citrus plants (Vekiari et al., 2004) which is used as an important alternative to toxic, hazardous and dangerous petroleum-derived chemicals used as cleaner and engine degreaser.
We are grateful to the Govan Mbeki Research and Development Centre of the University of Fort Hare, South Africa, for the post-doctoral fellowship granted to Dr OT Asekun and to the University of Lagos, Nigeria for releasing her during the period.