ABSTRACT
This study aims to clarify the induction of the “Crabtree consequence” in liver cells through measuring certain biochemical parameters in livers of Schistosoma mansoni infected mice. The liver is one of most significant target for toxic substances. and represents the main site for detoxication in the organism. The curative effect of oil extract of Curcuma longa (C.Longa) were tested and compared to praziquantel (PZQ) as the effective drug against all schistosome species occurring in man. Measurement of protein, glucose, glucose-6-phopsphatase (G-6-Pase), AMP-deaminase, adensoine deaminase, urea concentration, pyravate kinase (PK), phosphoenol pyruvate carboxykinase (PEPCK) and PK/PEPCK ratio were performed. In addition worm burden and ova count in mice infected with S.mansoni were elucidated. The result showed that C. longa normalized the concentration of protein, glucose, AMP-deaminase and adenosine deaminase which were changed by infection. Moreover it lowered pyruvate kinase (PK) level, which was elevated in infected mice while PZQ-treatment induced more elevation of this enzyme. Regarding the parasitological parameters, PZQ was more effective in lowering worm burden while C.Longa extract was more potent in reducing egg count.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2006.2991.3003
URL: https://scialert.net/abstract/?doi=jas.2006.2991.3003
INTRODUCTION
Schistosomiasis is one of the most common parasitic diseases which mostly affect the liver and intestine, causing granuloma formation and hepatic fibrosis. Schistosomisis also causes certain necrotic changes in the liver tissues (EL-Aaser et al., 1989; Li et al., 2000).
The chemotherapy of schistosomiasis was reviewed extensively (Silva and Andrade, 1995), PZQ compound is highly active against S. haematobium, S. japonicum and S. mansoni in the hamster with no apparent significant differences against the different geographical strains of the parasites (Homeida et al., 1988).
A new trend for treatment of liver disorders as a result of S. mansoni infection is the use of natural plant extracts. Curcumin, a yellow colouring agent from turmeric (Curcuma longa linn, Zingiberaceae) has been shown to inhibit tumor formation in diverse animals’ models (Asai and Miyazawa, 2000). Curcuma xanthorrbiza improved the diabetic symptoms such as growth retardation, elevation of glucose in the serum (Yasni et al., 1991).Curcumin (diferuloyl methane) a natural product obtained from the rhizomes of Curcuma longa, enhanced wound repair in diabetic impaired healing and could be developed as a pharmacological agent in such clinical settings (Piyachaturawat et al., 1999). Cardioprotective effects of C. Longa correlates with the improved ventricular function. Histopathological examination further confirmed the protective effects of C. Longa on the heart (Ipseeta et al., 2004). C. Longa has anti-inflammatory, antioxidant and anti-cancer activities (Radhakrishna et al., 2004).
Regarding the effect of schistosome infection, it was reported that while total liver protein content showed non significant differences in schistosoma infected as compared to control (El-Haieg et al., 1977). A significant decrease in concentration of serum total proteins and albumin were observed in patients with active schistosomiasis (either S. mansoni alone or mixed with S. haematobium) (El-Sharabasy et al., 1994).
Glucose-6-phosphatase activity showing a significant increase in hepatic cells of S. mansoni infected animals (Hamed and Hetta, 2005). After PZQ administration, there was a marked decrease of hepatic G-6-Pase. (Shaheen et al., 1989). It has been recognized for many years that the chief source of the ammonia produced by working muscle is the reaction catalyzed by AMP deaminase (Lowenstein, 1972). There is a high correlation between AMP-deaminase activity and phosphofructokinase activity lends credence to the idea that one function of AMP-deaminase may be to regulate flux through the glycolytic pathway (Winder et al., 1974). Ammonia formed by many parasites, it seems probable that competition for substrates by PEPCK and PK could account for the apparent shift in the proportions of succinate and lactate formed when oxygen is present in the maintenance medium (Saz et al., 1971).However, there is a group of parasites which have been described as homolactate fermenters, Schistosoma mansoni, which possesses PEPCK, belongs to this group (Brazier and Jaffe, 1973).
The liver is the main organ responsible for the biosynthesis, uptake and degradation of number of biological materials, including proteins and enzymes. Liver infection, may therefore, be reflected to some extent on the levels and/or the activities of these circulating biochemical compounds in serum, hepatic schistosomiasis is one of the most prevalent forms of human hepatic fibrosis in the world (Dunn and Kamel, 1981) and a reproducible experimental model of disease can be obtained in infected mice (Souza et al., 1993).
In this study the measured parameters were selected on the basis that they are closely related to the glycolytic flux which could reflected the Crabtree effect usually induced by the schistosome parasite in the definite host.
MATERIALS AND METHODS
Materials
Experimental animals: The animals used were healthy male albino mice of CD strain weighting 20-25 g obtained from the Schistosome Biological Supply Programmes (SBSP), Theoder Bilharz Institute. They were feed stock commercial pellets (El-Kahira Company for oil and soap) and water was supplied ad-libtum.
| • | Praziquantel drug (suspension), a product of Egyptian International Pharmaceutical Industries Company (E.I.P.I. Co). |
| • | Curcuma longa, crude material (obtained from chemistry and pharmacognosy department, National Research Centre) was reduced to a moderately coarse powder, hundred grams were immerged with 500 ml. 70% ethyl alcohol for 72 h., with occasional shaking. The extract was concentrated to dryness (Rana and Avadhoot, 1992). |
Chemicals: All the used reagents were of analytical grade obtained from sigma (U.S.A.), Merck (Germany), BDH (England), Reidel (Germany) and Fluka (Switzerland) Chemical companies.
Methods:
Infection: For the infection of mice, 10-20 Biomphalria alexandrina snails were placed in a beaker containing 200 mL. dechlorinated water. Snails were exposed to sunlight at 8-9 am. In order to shed cercariae. Each mouse was subjected to subcutaneous injection with 50 cercariae (Peters and Warren, 1969).
Animal treatments: Animals were divided equally into three batches first, second and third. These batches were of one, two and three month’s age respectively. Each batch was subdivided into 4 groups. For the 1st and 2nd batches, these 4 groups included group I, act as control, group II as infected group, group III control treated with Praziquantel (PZQ) while group IV was given (PZQ) post infection.
Groups III and IV served as control and infected given PZQ were sacrificed after 7 days of treatment. While the third batch of 3 months age were subdivided into the following four groups: I, served as a control, II infected group, III served as Curcuma longa – treated control while IV, was used as C. longa – treated infected group.
The experimental design and groups distribution is summarized in Table 1.
Praziquantel suspension was given orally 500 mg kg body weight, on two successive days (Piper et al., 1990). Curcuma longa extract was given orally to control and infected mice, one month post infection for a period of two months, twice per week 300 mg kg body weight (Rana and Avadhoot, 1992).
| Table 1: | Experimental design and groups distribution |
 | |
| * Each group contains six animals | |
At the end of each period, a group of six mice were weighed and scarified and the liver was taken for the biochemical analyses: - protein, glucose, glucose-6-phosphatase, AMP – deaminase, adenosine deaminase, urea, pyruvate kinase (PK), phosphoenol pyruvate carboxykinase (PEPCK) and liver and body weights. In addition worm burden and ova count in infected mice.
Preparation of tissue homogenates for biochemical analysis: A- one gram of liver tissue was homogenized in 9 mL. Bidistilled water to yield 10% homogenate, the homogenate was used for determination of protein, glucose, glucose–6-phosphatase and urea concentration were measured.
Determination of protein: Protein was estimated by the method of Gogstad and Krutnes (1982) as modified method of Bradford (1976).
Determination of glucose content: Glucose was determined colorimetrically by the method described by Trinder (1969).
Estimation of phosphate: Inorganic phosphate was estimated by the method of Fiske and Subbarow (1925).
Estimation of glucose -6- phosphatase activity (E. C. 3. 1. 3. 9):
Glucose -6- phosphatase activity was estimated according to the method of Swanson (1955).
Determination of urea content: Urea was determined colorimetrically by the method described by Patton and Crouch (1977).
B- The hepatic tissue homogenized in 10 volume of 20 mM cold potassium phosphate buffer (pH 7.1) containing 100 mM KCl and 0.1%, 2-mercaptoethanol. The mixture was centrifuged for 10 min. The filtrate was used for the measurement of Adenylate degrading enzymes (Fijisawa and Yoshino. 1987).
Determination of AMP - deaminase activity (E. C. 3. 5. 4. 6.): Enzyme activity was determined through the measurement of ammonium ion liberated using phenol hypochlorite reagent (Chaney and Marbach, 1962).
Determination of adenosine deaminase activity (E. C. 3. 5. 4. 4): The activity of the enzyme was determined by enzymatic colorimetric method of Fijisawa and Yoshino (1987).
C-One gram tissue of the liver was homogenized in 5 ml Tris - HCl buffer pH 7.6 according to Umezurike and Anya (1978), the homogenate was centrifuged at 4000 r. p. m. for 15 minutes and the supernatant was used for the measurement of Pyruvate kinase and phosphoenol pyruvate carboxykinase.
Determination of pyruvate kinase activity (PK) (E. C. 2. 7. 1. 40): PK is measured according to the method of Bucher and Pfleiderer (1975).
Determination of phosphoenol pyruvate carboxykinase (PEPCK) (EC 4. 1. 1. 49): PEPCK was assayed spectrophotometrically by the procedure of Suarez et al. (1986). OAA formation from PEP and NaHCO3 was determined by measuring the oxidation of NADH in the presence of malate dehydrogenase at 340 nm.
Recovery of adult worms of infected mice (liver perfusion): After infection of mice with 50 cercariae, adult Schistosoma mansoni worms were recovered from hepatic portal system and liver by the perfusion technique described by Smithers and Terry (1965). The infected mouse was killed by injection intraperitoneally with 0.15 mL of thiopental (0.5 μg). The adult worms were recovered from the hepatic system and the liver by perfusion with citrate saline (0.85% sodium chloride, 1.5% sodium citrate). When the liver, kidney and gut become pale, the perfusion process was stopped. The perfusate was collected in a container attached to the perfusion plate. The coils of the intestine were lifted from the tray and washed down in order to dislodge any worms adhering to them.
Worm counting: The degree of protection or the percent reduction in challenge was calculated as follows (Tendler et al., 1986).
Where: P = % protection, C = mean number of parasites recovered from infected mice and V = mean number of parasites recovered from treated mice.
Ova count: The number of ova in each tissue was counted in the three slides and the average number was calculated (Cheever, 1970; Cheever and Anderson, 1971).
Statistical analysis: The statistical significance of the results was determined by Ronald et al. (1983).
RESULTS
The results presented in Table 2 and Fig. 1. showed significant elevation of protein and glucose concentrations after one month post infection when compared with control group of mice, while at 2nd month post infection, a marked reduction in protein, glucose levels and glucose-6-phosphatase activity with respect to control mice with percentage change -21.7, -52.2 and -34.5% respectively, was observed.
| Table 2: | Effect of Praziquantel drug (PZQ) treatment on liver protein, glucose and glucose-6-phosphatase enzyme of Schistosoma mansoni infected mice |
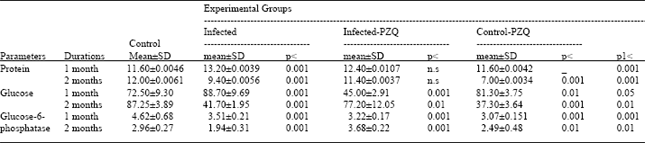 | |
| Protein and glucose were expressed as mg g-1 tissue, glucose-6-phosphatase was expressed as μ mol glucose released /min./mg protein. *p = Level of Significance as compared with control, P1 as compared to infected groups. Where p< 0.001: highly significant, p< 0.010: significant, p< 0.050: slightly significant and n.s: non significant | |
| Table 3: | Effect of Curcuma longa extract (C. longa) treatment on liver protein, glucose and glucose-6-phosphatase enzyme of Schistosoma mansoni infected mice |
 | |
| Protein and glucose were expressed as mg gm-1 tissue, glucose-6-phosphatase was expressed as μ mol glucose released /min. /mg protein. *p = Level of Significance as compared with control, P1 as compared to infected groups. Where p< 0.001: highly significant, p< 0.010: significant and n.s: non significant | |
| Table 4: | Effect of Praziquantel drug (PZQ) treatment on AMP-deaminase, adenosine deaminase and urea of Schistosoma mansoni infected mice |
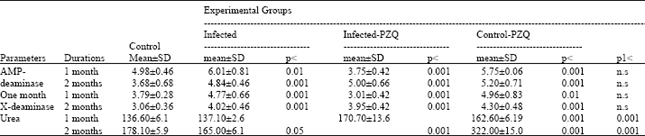 | |
| AMP-deaminase, adenosine deaminase and urea are expressed as μmoles ammonia /min /mg protein, p = Level of Significance as compared with control, P1 as compared to infected groups. Where p< 0.001: highly significant, p< 0.010: significant, p< 0.050: slightly significant and n.s : non significant | |
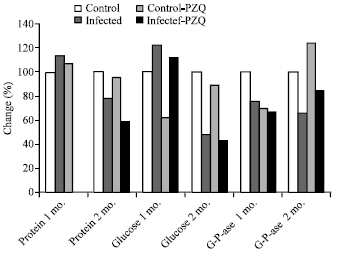 | |
| Fig. 1: | Diagrammatic representation illustrate the percentage change of protein, glucose and G-6-Pase in hepatic mice at different groups given PZQ as compared with control group |
 | |
| Fig. 2: | Diagrammatic representation illustrate the percentage change of protein, glucose and G-6-Pase in hepatic mice at different groups treated with C. longa as compared with control group |
| Table 5: | Effect of Praziquantel drug (PZQ) treatment on pyruvate kinase (PK), phosphoenol pyruvate carboxykinase (PEPCK) activities and PK/PEPCK ratio of Schistosoma mansoni infected mice |
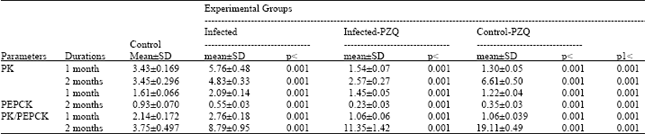 | |
| PK and PEPCK are expressed as μmoles NADH.H+ formed /min /mg protein, p = Level of Significance as compared with control, P1 as compared to infected groups. Where p< 0.001: highly significant, p< 0.010: significant, p< 0.050: slightly significant and n.s: non significant | |
| Table 6: | Effect of Curcuma longa extract (C. longa) treatment on AMP-deaminase, adenosine deaminase, urea, pyruvate kinase (PK), phosphoenol pyruvate carboxykinase (PEPCK) activities and PK/PEPCK ratio of Schistosoma mansoni infected mice |
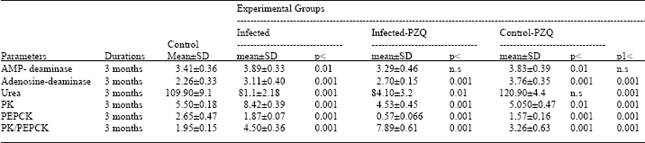 | |
| AMP-deaminase, adenosine deaminase and urea are expressed as μmoles ammonia /min /mg protein, PK and PEPCK are expressed as μmoles NADH.H+ formed /min /mg protein. p = Level of Significance as with control, P1 as compared to infected groups. Where p< 0.001: highly significant, p< 0.010: significant and n.s: non significant | |
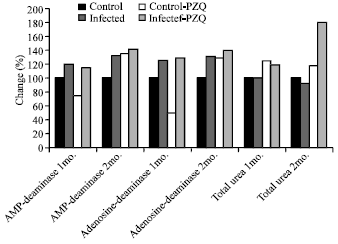 | |
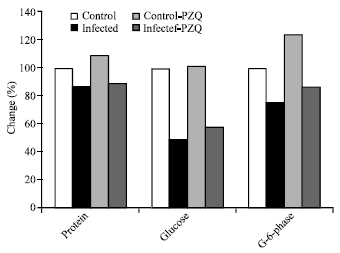
| Fig. 3: | Diagrammatic representation illustrates the percentage change of AMP-deaminase, adenosine deaminase and total urea in hepatic mice at different groups given PZQ as compared with control group |
It could be noticed that treatment of infected mice with PZQ caused a pronounced reduction of the elevated level of protein and reduction of glucose reaching to the level of control mice. While on the other hand, glucose was elevated after treatment with PZQ as compared with control. When control group were given PZQ, show increase in protein concentration and marked decrease in glucose at the first month, when compared to the control untreated.
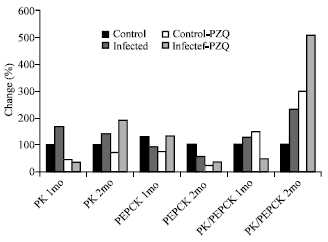 | |
| Fig. 4: | Diagrammatic representation illustrate the percentage change of AMP-deaminase, adenosine deaminase and total Urea in hepatic mice at different groups treated with C. longa |
Table 3 and Fig. 2 record a significant reduction of protein, glucose and G-6-ase with -12.3, -50.7 and -25% respectively, after 3 months of infection. These reduction were reduced with -10.76, -42.59 and -13.9% respectively when treated with C. longa extract.
Table 4 and Fig. 3 showed a significant elevation in AMP-deaminase in infected and infected given PZQ with 20.68, 31.5, 15.5 and 41.3% respectively. Also, adenosine deaminase activity and total urea concentration revealed increase with different percentage at different durations.
| Table 7: | Worm burden and ova count in liver of mice infected with Schistosoma mansoni in groups treated with praziquantel or Curcuma longa |
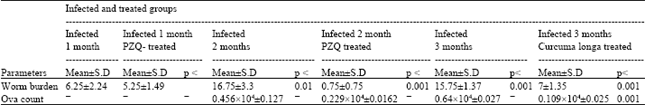 | |
| OVA count is expressed / gm tissue of liver. p = level of significance, Significantly different compared to the infected (untreated) level, Where p < 0.001: highly significant and p < 0.010: significant | |
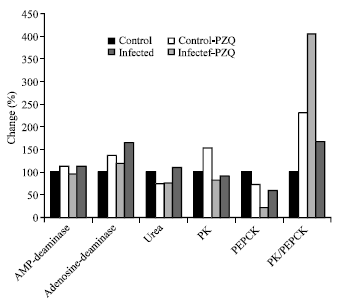 | |
| Fig. 5: | Diagrammatic representation illustrate the percentage change of AMP-deaminase, adenosine deaminase, total urea PK, PEPCK and PK/PEPCK ratio in hepatic mice at different groups treated with C. LONGA as compared with control group |
Table 5 diagrammatically represented in Fig. 4 demonstrate that PK and PEPCK activities significantly reduced in control-treated (groupIII) with percentage change 25.5 and 75.2%, respectively while at 2nd month post infection given PZQ, these enzymes were change with +91.6 and -62.3%, respectively as compared to control (group I ). On the other hand PK/PEPCK ratio was significantly elevated in the two groups III and IV with percentage increase 203 and 409% respectively as compared to groupI.
Table 6 and Fig. 5 record that PK and PEPCK activities were decreased in both control and infected groups treated with C. LONGA. The effect of C. LONGA can be easily noticed through the reduction of PK and PEPCK activities recorded -8.2 and -40.7% percentage, when compared to the control group.
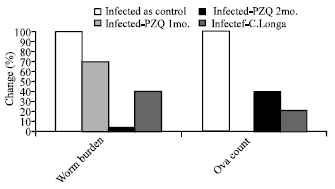 | |
| Fig. 6: | Diagrammatic representation illustrate the percentage change of worm burden and ova count in hepatic mice treated with PZQ or C. LONGA as compared with infected as control |
AMP-deaminase, adenosine deaminase activities and urea concentration were elevated with 12.3, 66.3 and 10.0% changes respectively in infected group treated with C. LONGA.
From Table 7 and Fig. 6 it can be noticed that PZQ treatment one and two months post infection induced reduction in worm burden by 16% and 95.5% respectively, when compared to infected groups. C. LONGA treatment showed a significant reduction in worm burden and ova count compared to the three months infection duration with 55.5 and 83 percentage change respectively. In case of PZQ, ova count recorded only 49.8% inhibition when compared to the infected group.
DISCUSSION
Earlier studies have shown that the interaction between schistosomal parasites and the mammalian host is extremely complex. Many parasitologists have focussed their studies on the epidemiology of schistosomiasis or the physiology of the parasites neglecting to some extent the metabolic changes developed in the host in consequence to infection or drug treatment. Despite the possession of a mouth and functional gut, glucose is taken up by the schistosome parasites across their outer body surface or tegument. All cells appear to move glucose molecules across specific glucose transporter proteins (GTPs) (Camacho and Agenw, 1995). Because mammalian stage of schistosomes live in a high glucose medium, it seems likely that uptake of host glucose accelerated the synthesis of these transporters (Skelly and Shoemaker, 2000).
This study showed that, as early as one month post infection of mice with Schistosoma mansoni cercariae and prior to maturation and egg deposition, significant elevation of protein and glucose was observed in the tissue homogenates of schistosome-infected animals. This may prove that at this stage of infection, the parasite has no disturbing effect on host metabolism. The elevation of the measured parameters could be attributed to the presence of the parasite itself. Total liver protein content showed non significant differences in schistosome infected as compared to control. These findings might be due to either the decrease in hepatic cell population due to the liver fibrosis or the release of the enzyme from the damaged livers into the circulation (El-Haieg et al., 1977). El-Hawry et al. (1971) recorded that the decrease in albumin fraction may be due to decreased anabolism or increased catabolism, malnutrition and/or malabsorption may contribute to the decreased biosynthesis of albumin.
Praziquentel (PZQ) has become the drug of choice in most endemic areas because of its efficacy, its ease of administration, its tolerable side-effects and its cost. As a consequence of this positive trend, two potential dangers have emerged. The possibility that other existing drugs might be discontinued and the diminished interest of major pharmaceutical companies in the quest for novel active compounds (Cioli, 1998). In the present study, PZQ was not effective in restoring normal glucose level. Control, one and two months-infected animals were found to have lower glucose concentration. Moreover, PZQ- treatment reduced the protein content of hepatic cells either in control or infected animals. This could be explained on the basis that this drug may interfere with amino acid and proteins metabolism of treated animals.
There is still intensive search for effective anti-schistosomal drugs with minimal side effects. Natural health products have become increasingly important in the lives during the past few years (Khopde et al., 2000; Groten et al., 2000). The huge global economic potential for the production and processing of medicinal plants has led to important initiatives in research, development and regulatory procedures. In the present study, Curcuma longa extract as a plant with recently reported many medicinal properties (Olajide, 1999; Kawamori et al., 1999). Curcuma longa was used as antibilharzial drug. The obtained data proved that C. longa extract induced a significant elevation of glucose concentration in control and infected C. longa-treated animals. Regarding the enzyme activity, glucose-6-phosphatase (G6Pase), was found to be variably affected by bilharzial infection (Tanabe et al., 1997).In the present study, G-6-Pase was found to be greatly inhibited in schistosome-infected mice (1, 2 and 3 months post infection). The pronounced decrease in G-6-Pase activity reported in the present study due to infection seems to be consistent with the lower adenylate energy charge (AEC) previously reported in schistosome-infected animals at the same experimental durations (El-Ansary and Farouk, 2001). Lower AEC usually accompanied by activated glycogen phosphorylase and glycolytic enzymes and inhibited glycogen synthase and gluconeogenic enzymes (Geoffrey et al., 1995). G-6-Pase as a gluconeogenic enzyme was inhibited with infection. This led to the availability of glucose-6-phosphate as a substrate for phosphohexoisomerase the glycolytic enzymes. Reduced G-6-Pase activity recorded in the present study is in full agreement with El-Merzabani et al. (1977) and Daniele et al. (1997) who reported a significant reduction in G-6-Pase activity between normal and schistosome infected livers respectively.
The decrease of G-6-Pase activity indicated the damaging effect of schistosomiasis on liver parenchymal cells. The hepatic hypoglycemia reported in the present investigation in infected animals may be a normal consequent to the decrease of G-6-Pase activity level. This could be supported through considering the work of El-Haieg et al. (1977) who recorded the inability of hepatic cells to dephosphorylate glucose-6-phosphate into glucose, the marked decrease in glycogen, total liver protein content. Moreover it is good with Hara et al. (1981) who found a significant decrease of enzyme activity G-6-Pase in the liver of mice infected by subcutaneous injection with S. mansoni cercariae. They attributed the decrease in enzyme activity to molecular and biological changes occurring in hepatic and granulomatous cells.
PZQ as uncharged compound was found to induce some morphological alterations in human erythrocytes. Haemolysis of erythrocytes and release of membrane lipids (phospholipids and cholesterol) were shown to be concentration - dependent (Malheiros et al., 2000). These results suggest that distinct cell membrane interaction pathways lead to drug-specific mechanisms of cytotoxicity. The change in the activity of G-6-Pase observed in control and infected animals-treated with PZQ could be correlated to its membrane interaction and increase of hepatocytes permeability. C. longa on the other hand was effective in activating G-6-Pase in control- treated mice. In case of infected animals, C. longa extract could have significantly reactivated G-6-Pase which showed its potent effect in restoring normal hepatocyte permeability, which could be supported by Park et al. (2000) and Joe and lokesh (2000) Moreover, Jayadeep et al. (2000) revealed that the antioxidative and hypolipidaemic action of C. longa is responsible for its protective role against ethanol induced brain injury. In recent years, clinical interest has been directed to the study of other liver enzymes which might offer an improved specificity over the conventional liver function enzymes for the confirmation of the degree and activity of liver disease and the success of its treatment. In the present study, AMP deaminase and adenosine deaminase as adenylate degrading enzymes were studied. These two enzymes were selected since they have a regulatory role on the glycolytic pathway as the most important metabolic pathway in schistosome-infected definitive or intermediate hosts (El-Ansary, 1999; El-Ansary et al., 2000a and b). In the present study, AMP deaminase and adenosine deaminase were biochemically detected in the liver tissues of normal-healthy animals. In the present investigated both enzymes were significantly stimulated in schistosome- infected mice at the three different durations studied. Activation of these two enzymes is consistent with the stimulation of the glycolytic flux previously reported by many authors in schistosome infected animals (Soliman et al., 2000). AMP deaminase activity is closely correlated with the activity of phosphofructokinase and pyruvate kinase as key enzymes of glycolysis in mammalian cells. The ammonia formed by deamination of AMP may accelerate the rate of glycolysis through these two enzymes (Yoshino and Murakami, 1982 a,b). The presence of an activated AMP-deaminase in schistosome-infected liver cells confirms the dependence of energy metabolism of infected animals on the glycolytic flux.
Moreover, because the liver plays a central role in detoxication of free ammonia absorbed from gastrointestinal tract or derived from amino acid breakdowns in a large number of higher organisms, hepatic damage often results in elevation of blood ammonia levels (Tanabe et al., 1989). Actually, hyperammonemia and abnormal ammonia tolerance have been demonstrated in mice experimentally infected with S. mansoni and patients with hepatosplenic schistosomiasis (Page et al., 1972). Such alterations in the amino acid catabolism and in the detoxication of free ammonia are considered to be related with the activity of urea cycle.
In the current study, urea levels presented in the same table together with AMP deaminase and adenosine deaminase is related to these two enzymes which produce ammonia as a metabolic product, since urea is the detoxified product of ammonia. The recorded lower urea concentration in schistosome-infected mice could be easily correlated to the significant decrease in both total and specific activities of carbamayl phosphate synthetase and ornithine carbamyl transferase (Tanabe et al., 1989) of female mice infected with cercariae of S. mansoni. Were significantly inhibited these enzymes. Moreover, this decrease in the urea concentration confirmed the recent work of Rizk et al. (2000) who reported inhibition of carbamayl phosphate synthetase, argininosuccinate lyase and arginase as urea cycle enzymes greatly affected with schistosome infection. Inhibition of the ammonia detoxifying activity in hepatocytes of infected mice could be explained on the basis that infections significantly inhibit glutathione-S-transferase, glutathione reductase, reduced glutathione together with cytochrome P450 and NADH-cytochrome c reductase as a group of drug metabolizing enzymes (Sheweita et al., 1998). The alteration in the activities of these drug- metabolizing enzymes as a result of infection with different levels of S. mansoni may thus change the liver’s capacity to detoxify endogenous compounds as ammonia through its conversion to urea.
The effectiveness of PZQ as an antibilharzial drug is easily noticed in this investigation. PZQ reduced the enzymatic activities of AMP-deaminase and adenosine deaminase to a certain extent. Inhibition of these two enzymes could be related to its inhibitory effect of glycolysis and stimulation of Krebs cycle previously reported by Ahmed and Gad (1995). PZQ also was effective in elevating urea concentration in control and infected mice livers. Elevation of urea concentration with PZQ treatment, two and three months post infection confirmed that liver cells restored its NH3 detoxifying ability. C. longa extract was more effective in normalizing the enzymatic activities of AMP deaminase and adenosine deaminase when compared to PZQ. This could be easily explained on the basis that C. longa has potential role in the stimulation of oxidative phosphorylation and stabilization of adenylate energy charge (AEC) as a biochemical parameter controlling the flux of metabolites through the glycolytic pathway (Atkinson, 1971; El-Ansary and Farouk, 2001). C. longa was also effective as PZQ in initiating the impaired NH3 detoxifying mechanism in schistosome-infected animals. Hasmeda and Polya (1996) reported that curcumin, as a major bioactive secondary metabolite isolated from rhizomes of C. longa inhibits Cyclic-AMP dependent protein kinase. Lower protein kinase activity could be helpful through the cascade mechanism to keep glycogen phosphorylase and glycogen synthase in the dephosphorylated form. This in turn may suppress glycogen breakdown and stimulates glycogen synthesis which could explain the glycogen repletion in C. longa treated-infected animals (Geoffrey et al., 1995; El-Banhawey et al., 2006). It was suggested that an important control point in the respiration of parasitic helminthes exist at the level of phosphoenol pyruvate (PEP) (Horemans et al., 1992). The nature of the end product formed is dependent on the competition for PEP by the two enzymes Pyruvate kinase (PK) and phosphoenol pyruvate carboxykinase (PEPCK). Stimulation of PK in schistosome-infected animals ascertained the stimulation of the glycolytic flux previously reported by and Ahmed and Gad (1995). Stimulation of PK was reflected on the substrate availability of PEP for PEPCK and resulted in its remarkable inhibition two and three months post infection (Prichard, 1976). This in turn led to significantly higher PK/PEPCK ratios in schistosome-infected mice. PK/PEPCK ratio is described as potential regulatory site. It may give an indication about a major pathway in vivo (Moon et al., 1977). Animals that rely on glycolysis generally have a ratio in the region 2-10, whilst those that rely on Krebs cycle have a much lower ratio in the region 0.05-0.1 (Barrett, 1981). Data of the present investigation confirmed the stimulation of the glycolytic flux with bilharzial infection. Values of 2.76, 8.79 and 4.5 PK/PEPCK ratios were recorded for one, two and three months infected mice respectively compared to values of 2.15, 3.75 and 1.95 for the corresponding age-matching controls. PZQ was effective in lowering PK as a rate limiting enzyme of glycolysis, able to switch on/off the whole metabolic pathway. This finding is in good agreement with Ahmed and Gad (1995) who reported a more or less same effect of PZQ on phosphofructokinase (PFK) as another glycolytic regulatory enzyme. In the present study, PZQ caused a significant inhibition of the gluconeogenic enzyme PEPCK in control and infected animals. Inhibition of PEPCK was reflected on the PK/PEPCK ratio resulted in ratios of 19.11 and 11.39 for control and infected PZQ-treated animals. This may be helpful to clarify the impairment of the gluconeogenic activity in case of PZQ-treatment. Inhibition of PEPCK could be easily correlated to the inhibition of G-6-Pase previously discussed in the present study. C. longa was highly effective in normalizing PK activity of infected mice (3 months post infection) and at the same time it has less inhibitory effect on PEPCK showing a value of 40.7% decrease compared to a value of -62.3% in PZQ-treated infected animals. The slight effect of C. longa on this respect could be easily noticed in the remarkable lower PK/PEPCK ratio of 3.26 which showed a slightly impaired gluconeogenic activity in infected mice treated with this extract.
In infection by S. mansoni, the major pathologic changes are not caused by the adult worm itself but by eggs which do not reach the intestinal lumen, but instead, become trapped in other body tissues. At these sites, areas of local inflammation are produced, cumulating in the formation of granulomas around eggs (Giboda and Smith, 1994). The formation of granuloma around schistosome eggs in the liver and the intestine is the major cause of pathology in schistosome infections (Modha et al., 1998). It is the result of a classic Th2 cytokine-mediated immune response that was shown to involve tumor necrosis factor-α (TNF-α) (Boros, 1994 and Hassan et al., 2000).
In the present study PZQ was effective in reducing worm burden, ova count and granuloma size in infected -treated mice. These results coincide with those of Gerges et al. (1994a, b) and Farah et al. (2000) who reported that worm burden and the number of eggs were significantly reduced in PZQ-treated animals when compared to untreated groups. Moreover, Botros et al. (1996) discussed that PZQ orally given (500 mg kg) over 2 consecutive days at 7 weeks to mice caused complete eradication of worms, disappearance of immature egg stages and decrease in the number of mature eggs and an increase in the number of dead eggs. The present study showed that after one month post infection mice treated PZQ, there was few worms still living, in agreement with Yang et al. (1981) who revealed that male worms which were still living but encapsulated by connective tissue, were observed along the liver margin of some rabbits. Histopathological examination of liver sections revealed moderate to small-sized hypocellular granulomas (El-Fakahany et al., 1993, 1995).
Although C. longa extract was less effective in reducing the worm burden (-55.5%) in schistosome-infected-treated animals when compared to PZQ (-95.5%), it was about 2 fold higher in reducing ova count (-83.0%) in treated animals compared to PZQ treatment (-49.8%). This could be more promising in controlling the pathology of this disease which is mostly due to the toxic released on secreted by the eggs and could be attributed to the antifecundity effect of curcumin. Curcumin, obtained from powdered rhizomes of plant Curcuma longa linn, is commonly used as coloring agent in food, drugs and cosmetics (Chuang et al., 2000). In conclusion, the author propose that C. longa extract as a remarkable non-toxic plant with many medicinal properties should be explored for possible intervention in schistosomiasis as a disease involve impairment of metabolism of infected subjects. These inspire more hope for further study on C. longa.
REFERENCES
- Ahmed, S.A. and M.Z. Gad, 1995. Effect of schistosomal infection and its treatment on some key enzymes of glucose metabolism in mice livers. Arzneimittel Forschung, 45: 1324-1328.
Direct Link - Asai, A. and T. Miyazawa, 2000. Occurrence of orally administered curcuminoid as glucuronide and glucuronide/sulfate conjugates in rat plasma. Life Sci., 67: 2785-2793.
PubMedDirect Link - Botros, S.S., B.L. Doughtly, Z.A. Shaker, M.M. Aki, R. Sharmy, T.M. Diab and H.I. Hassanein, 1996. Efficacy of an antipathology vaccine in murine schistosomiasis administered with and without chemotherapy. Int. J. Immunopharmacol., 18: 707-718.
CrossRefDirect Link - Bradford, M.M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248-254.
CrossRefPubMedDirect Link - Camacho, M. and A. Agnew, 1995. Glucose uptake rates by Schistosoma mansoni, S. haematobium and S. bovis adults using a flow in vitro culture system. J. Parasitol., 81: 637-640.
Direct Link - Chaney, A.L. and E.P. Marbach, 1962. Modified reagents for determination of urea and ammonia. Clin. Chem., 8: 130-132.
PubMedDirect Link - Cheever, A.W., 1970. Quantitative comparison of the intensity of S. mansoni infection in man and experimental animals. Trans. R. Soc. Trop. Med. Hyg., 63: 781-795.
Direct Link - Cheever, A.W. and L.A. Anderson, 1971. Rate of destruction of Schistosoma mansoni eggs in tissues of mice. Am. J. Trop. Med. Hyg., 20: 62-68.
Direct Link - Chuang, S.E., A.L. Cheng, J.K. Lin and M.L. Kuo, 2000. Inhibition by curcumin of diethylnitrosamine-induced hepatic hyperplasia, inflammation, cellular gene products and cell-cycle-related proteins in rats. Food Chem. Toxicol., 38: 991-995.
CrossRefPubMedDirect Link - El-Ansary, A., K. El-Shaikh and M. El-Sherbini, 2000. SDS-PAGE-separated tissue proteins of Biomphalaria alexandrina snails in the presence and absence of Schistosoma mansoni. Egypt. J. Parasitol., 30: 125-136.
PubMedDirect Link - El-Fakahani, A.F., K.F. Abdalla, H.M. El-Hadi, S.M. Abd-El-Azez and L.M. Afifi, 1993. The effect of praziquantel treatment on the liver functions worm burden and granuloma size using two drug regimen in murine Schistosoma mansoni infection. J. Egypt. Soc. Parasitol., 23: 877-886.
PubMed - El-Haieg, M.O., I.I. Ibrahim and M.F. Zanaty, 1977. Alpha-fetoprotein in adult normals bilharzial hepatic fibrosis and viral hepatitis in Egypt. J. Egypt. Med. Assoc., 60: 699-706.
PubMedDirect Link - El-Hawary, M.F.S., A.M. Ibrahim, A.H. Shaker and M. Saif, 1971. Studies on serum proteins in bilharziasis by agar gel electrophoresis and the effect of treatment with niridazole. J. Egypt. Med. Assoc., 54: 101-105.
PubMedDirect Link - Farah, I.O., P.W. Mola, T.M. Kariuki, M. Nyindo, R.E. Blanton and C.L. King, 2000. Repeated exposure induces periportal fibrosis in Schistosoma mansoni infected baboons role of TGF-beta and IL-4. J. Immunol., 164: 5337-5343.
PubMedDirect Link - Giboda, M. and J.M. Smith, 1994. Schistosoma mansoni eggs as a target for praziquantel efficacy of oral application in mice. J. Trop. Med. Hyg., 97: 98-102.
PubMedDirect Link - Groten, J.P., W. Butler, V.J. Feron, G. Kozianowski, A.G. Renwick and R. Walker, 2000. An analysis of the possibility for health implications of joint actions and interactions between food additives. Regul. Toxicol. Pharmacol., 31: 77-91.
PubMedDirect Link - Hamed, M.A. and M.H. Hetta, 2005. Efficacy of Citrus reticulata and Mirazid in treatment of Schistosoma mansoni. Mem. Inst. Oswaldo Cruz, 100: 771-778.
PubMedDirect Link - Hara, A., K. Fukuyama and W.L. Epstein, 1981. Angiotensin-converting enzyme and other enzymes in livers of mice with experimental schistosomiasis. Exp. Mol. Pathol., 35: 199-210.
CrossRefDirect Link - Horemans, A.M.C., A.G.M. Tielens and S.G. Van Den Bergh, 1992. The reversible effect of glucose on the energy metabolism of Schistosoma mansoni cercariae and schistosomula. Mol. biochem. Parasitol., 51: 73-90.
PubMedDirect Link - Mohanty, I., D.S. Arya, A. Dinda, S. Joshi, K.K. Talwar and S.K. Gupta, 2004. Protective effects of Curcuma longa on ischemia-reperfusion induced myocardial injuries and their mechanisms. Life Sci., 75: 1701-1711.
PubMed - Rajakrishnan, V., A. Jayadeep, O.S. Arun, P.R. Sudhakaran and V.P. Menon, 2000. Changes in the prostaglandin levels in alcohol toxicity: Effect of curcumin and N-acetylcysteine. J. Nutr. Biochem., 11: 509-514.
CrossRefPubMedDirect Link - Kawamori, T., R. Lubet, V.E. Steele, G.J. Kelloff, R.B. Kaskey, C.V. Rao and B.S. Reddy, 1999. Chemopreventive effect of curcumin a naturally occurring anti-inflammatory agent, during the promotion/progression stages of colon cancer. Cancer Res., 59: 597-601.
PubMedDirect Link - Khopde, S.M., K.I. Priyadarsini, S.N. Guha, J.G. Satav, P. Venkatesan and M.N. Rao, 2000. Inhibition of radiation induced lipid peroxidation by tetrahydro curcumin possible mechanisms by pulse radiolysis. Biosci. Biotechnol. Biochem., 64: 503-509.
Direct Link - Kind, P.R.N. and E.J. King, 1954. Estimation of plasma phosphatase by determination of hydrolysed phenol with amino-antipyrine. J. Clin. Pathol., 7: 322-326.
CrossRefPubMedDirect Link - Li, Y.S., A.C. Sleigh, A.G. Ross, Y. Li, G.M. M. Williams, Tanner and D.P. McManus, 2000. Two year impact of praziquantel treatment for Schistosoma japonicum infection in China reinfection subclinical disease and fibrosis marker measurements. Trans. R. Soc. Trop. Med. Hyg., 94: 191-197.
Direct Link - Malheiros, S.V., M.A. Brito, D. Brites and N.C. Meirelles, 2000. Membrane effects of trifluoperazine dibucaine and praziquantel on human erythrocytes. Chem. Biol. Interact., 126: 79-95.
CrossRef - Moon, T.W., T. Mustafa, W.C. Hulbert, R.B. Podesta and D.F. Mettrick, 1977. The phosphoenol pyruvate branch point in adult hymenolepis diminuta cestoda a study of pyruvate kinase and phosphoenol pyruvate carboxylase. J. Exp. Zool., 200: 325-336.
Direct Link - Olajide, O.A., 1999. Investigation of the effects of selected medicinal plants on experimental thrombosis. Phytother. Res., 13: 231-232.
PubMedDirect Link - Page, C.R., F.J. Etges and J.D. Ogle, 1972. Experimental prepatent schistosoma mansoni quantitative analysis of proteins enzyme activity and free amino acid in mice serum. Exp. Parasitol., 31: 341-349.
PubMedDirect Link - Patton, C.J. and S.R. Crouch, 1977. Spectrophotometric and kinetics investigation of the Berthelot reaction for the determination of ammonia. Anal. Chem., 49: 464-469.
CrossRefDirect Link - Peters, P.A. and K.S. Warren, 1969. A rapid method of infecting mice and other laboratory animals with Schistosoma mansoni: Subcutaneous injection. J. Parasitol., 55: 558-560.
CrossRefDirect Link - Piyachaturawat, P.A., A. Timinkul, A. Chuncharunee and A. Suksamrarn, 1999. Effect of Curcuma comosa extract on male fertility in rats. Pharmaceut. Biol., 37: 22-27.
Direct Link - Prichard, R.K., 1976. Regulation of pyruvate kinase and phosphoenolpyruvate carboxykinase activity in adult Fasciola hepatica Trematoda. Int. J. Parasitol., 6: 227-233.
Direct Link - Pillai, G.R., A.S. Srivastava, T.I. Hassanein, D.P. Chauhan and E. Carrier, 2004. Induction of apoptosis in human lung cancer cells by curcumin. Cancer Lett., 208: 163-170.
PubMed - Rana, A.C. and Y. Avadhoot, 1992. Experimental evaluation of hepatoprotective activity of Gymnema sylvestre and Curcuma zedoaria. Fttoterapia, 63: 60-62.
Direct Link - Silva, L. and Z.A. Andrade, 1995. Repair of schistosomal intestinal vascular lesions after curative treatment. Memo-rias do Instituto Oswaldo ctuz., 90: 401-405.
Direct Link - Skelly, P.J. and C.B. Shoemaker, 2000. Induction cues for tegument formation during the transformation of Schisosoma mansoni cercariae. Int. J. Parasitol., 30: 625-631.
PubMed - Smithers, S.R. and R.J. Terry, 1965. The infection of laboratory hosts with cercariea of S. mansoni and the recovery of adult worms. Parasitology, 55: 695-700.
Direct Link - Soliman, K.M., A.K. El-Ansary and A.M. Mohamed, 2000. Effect of carnosine administration on certain metabolic parameters in bilharzial infected hamsters. J. Egypt. Soc. Parasitol., 30: 455-468.
PubMed - Souza, V.M.R., A.A.J. Barbosa and Z.A. Andrade, 1993. Experimental pulmonary schistosomiasis lack of morphological evidence of modulation in schistosomal pulmonary granulomas. Revista. Medic. Trop. paulo, 35: 423-429.
Direct Link - Tanabe, M., N. Kaneko and T. Takeuchi, 1989. Schistosoma mansoni suppression of carbamoyl phosphate synthetase (Ammonia) and ornithine carbamoyltransferase activities in the liver of infected mice. Exp. Parasitol., 68: 432-442.
PubMed - Tanabe, M., J.F. Goncalves, F.J. Goncalves, S. Tateno and T. Takeuchi, 1997. Occurrence of a community with high morbidity associated with Schistosoma mansoni infection regardless of low infection intensity in North - East Brazil. Trans. Ro. Soc. Trop. Med. Hyg., 91: 144-149.
Direct Link - Tendler, M., R.M. Pinto, L.A. Oliveira, G. Gebara and N. Katz, 1986. Schistosoma mansoni: Vaccination with adult worm antigens. Int. J. Parasitol., 16: 347-352.
CrossRefDirect Link - Winder, W.W., R.L. Terjung, K.M. Baldwin and J.O. Holloszy, 1974. Effect of exercise on AMP deaminase and adenylosuccinase in rat skeletal muscle. Am. J. Physiol, 227: 1411-1414.
Direct Link - Yoshino, M. and K. Murakami, 1982. AMP deaminase reaction as a control system of glycolysis in yeast activation of phosphofructokinse and pyruvate kinase by AMP deaminase ammonia system. J. Biol. Chem., 257: 4729-4732.
PubMedDirect Link - Yoshino, M. and K. Murakami, 1982. AMP deaminase reaction as a control system of glycolysis in yeast mechanism of the inhibition of glycolysis by fatty acid and citrate. J. Biol. Chem., 257: 10644-10649.
Direct Link - Sheweita, S.A., S.A. Mangoura and A.G. El-Shemi, 1998. Different levels of Schistosoma mansoni infection induce changes in drug-metabolizing enzymes. J. Helminthol., 72: 71-77.
CrossRef








