ABSTRACT
In most developing countries, the use of Herbal Medicinal Preparations (HMPs) is common due to poverty and disillusionment with conventional medical care. In Nigeria, these products are not subjected to stringent regulation and control even with the increasing cases of adulteration and contamination (especially with heavy metals) in similar products in Asia and the Middle East. Lead and Cadmium content of some common Nigerian medicinal preparations were determined with flame atomic absorption spectrometer after acid digestion with 1:3 mixture of HNO3/HCl. Lead levels > 10 μg g-1 was detected in about 37% of the samples whereas 21% of the samples contains Pb levels ≥100 μg g-1 (range ND-213.6 μg g-1) The Cd content of the samples was generally less than 7 μg g-1 (range 0.3-6.61 μg g-1). Higher Pb levels were observed in preparations used in the treatment of eye infections, (as eye cleansers and in cosmetics) (8.06-213.6 μg g-1), compared to herbal soaps (ND-54.9 μg g-1), powdery preparations (ND-138.19 μg g-1) and the oil based preparations (ND-4.91 μg g-1). About 63% of the samples studied may be considered relatively safe if a Pb toxicity threshold of 10 μg g-1 is applied. These data indicate an urgent need for stringent monitoring and regulation of these products. The health implications of the application of these products can only be monitored using the Blood Lead Level (BLL) of groups engaging in such practices.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2006.2907.2911
URL: https://scialert.net/abstract/?doi=jas.2006.2907.2911
INTRODUCTION
People are becoming increasingly disillusioned with conventional medical care and are seeking to gain some measure of control over their illness through alternative health care practices (Crone and Wise, 1998; Hamzah et al., 2004). The use of herbal medicine, the dominant form of medical treatment in developing countries has been increasing in developed countries in recent years (Wojcikowski et al., 2004; Lunch and Braithwaite, 2005; Lekouch et al., 2001; Garg and Hershey, 2003).
Herbal medicine in which plants (dried or in extract form) are used as therapeutic substances, is one of a number of practices encompassed by the term Complementary and Alternative Medicine (CAM) and referred to as ‘Herbal Medical Products’ (HMPs) (Ernst, 2004; Drew and Myers, 1997). Spices and medicinal plants are known to contain trace metals which play vital roles as structural and functional components of metalloproteins and enzymes in the living cells (Ansari et al., 2004).
Although many of these remedies are used safely, there have recently been an increasing number of case reports of heavy metal poisoning (especially Pb, Hg, As and Cd) after use of traditional remedies (Lynch, 2005; Ernst, 2003, 2004; Susan, 2005).
Increasing toxicity of herbal remedies is speculated to be related to lack of child-resistant packaging, new issues of contamination, proliferation of multiple ingredient products, excessive concentration of active ingredients and discovery of new drug-herb interactions (Susan, 2005).
Consumption of some herbal preparations including herbal teas earlier considered to be safe for long periods of time can result in acute hepatic failure and renal impairment (Vanderperren et al., 2005; Ernst, 2004).
In most countries, including Nigeria, HMPs are not submitted to stringent regulation and control. This unreliable quality can be a problem especially when HMPs are contaminated (e.g., with heavy metals) or adulterated (e.g., with prescription drugs) (Ernst, 2004). Heavy metals particularly Pb have been identified as a regular constituent of traditional Indian and Asian remedies and this has repeatedly caused serious harm to patients taking such remedies (Garvey et al., 2001; Ernst, 2002; Ang et al., 2004). High levels of Pb (5-37000 ppm) and Hg (28-104000 ppm) have been reported in Ayurvedic herbs manufactured in South Asia (Saper et al., 2004).
There is paucity of data on the heavy metal contents of Nigerian medicinal preparations. In this study we evaluated the Pb and Cd content of some common Nigerian herbal medicinal preparations.
MATERIALS AND METHODS
Samples of common local medicinal preparations taken orally, used in skin infections treatment and for eye infections treatments (as eye cleansers and in cosmetics) were purchased from retail outlets or collected from traditional practitioners in Isuikwuato and Umuahia in Southeastern Nigeria. The common names of the medicinal preparations/remedies (mostly in Igbo language of Nigeria) studied are given in Table 1. The solid samples were dried to constant weight in an oven at 105°C for 12 h. One gram of sample was digested with two portions of 5 mL each of 1: 3 HNO3/HCl, heated to near dryness on a programmable hot plate and then cooled. The digest was taken up in 1 M HNO3 filtered through a Whatman No. 4 filter paper into 10 mL volumetric flask and made up to volume with the 1 M HNO3. The sample solutions were subsequently analyzed for Pb and Cd using an air-acetylene flame atomic absorption spectrometry (SOLAAR 32AA). Reagents used were of analytical grade and calibration standards were made by dilution of the high purity commercial BDH metal standards for atomic absorption analysis. Internal quality control with re-tests of Pb and Cd standards prepared in 1 M HNO3 was undertaken and results reported are average of duplicates.
| Table 1: | Common/brand names of the local Nigerian medicinal preparation used in this study |
 | |
RESULTS AND DISCUSSION
Lead was detected in only one of the powdery preparations. This product is used on neonates and the pediatric age group and by adults in the treatment of skin rashes, prickle heat and other skin infections. The high level of lead in this preparation (138.19 μg g-1) can expose users to intermittent high doses of Pb (Table 2). Exposure to Pb by dermal contact can contribute to significant toxicity (Moyer et al., 1999; Nnorom et al., 2005). Similarly, Pb was detected in only one of the oil based preparations (eleaku) a crude extraction of palm kernel oil. This may have resulted from contamination during the processes of cracking the palm kernel to obtain the nuts (kernel) or in the extraction processes. The oil is extracted usually by elderly women in the rural areas by heating the kernel to high temperature in a ceramic or stainless steel pot.
Higher mean Pb values was observed in the ophthalmologic preparations (106.37 μg g-1) compared to the mean values of the other sample forms. Most of the samples in this group are also used in cosmetics. In the past decade, Pb exposure to the eyes as a result of the use of traditional cosmetics (henna, surma, kwali, kohl, otangere) in Asia, Africa and the Middle East has been a subject of debate for scientists (Lekouch et al., 2001; Al-Saleh 1995; Funtua and Oyewale, 1997; Nnorom et al., 2005, Chukwuma, 1997). The materials used in the preparation of these products in Nigeria are scavenged from the Benue valley trough of which the Enyingba-Abakiliki lead and zinc mines are part (Chukwuma, 1997; Hearly et al., 1984). These products are used in the treatment of eye infections and in cosmetics. The levels of Cd in the samples are generally below 7 μg g-1 (range 0.30-6.61 μg g-1). This can, though considered safe, expose users to low doses of Cd when these products are used for long periods of time or in large quantities.
The observed high levels of especially Pb in these medicinal preparations indicate the need for registration of these products with emphasis on the quality, efficacy and safety (including heavy metal contamination) of these products. One of the quality requirements for traditional medicines in Malaysia is that they should not exceed 10ppm of Pb (Ang et al., 2004, 2005). A study of the Pb content of such products in Malaysia indicated a range of 10.23-23.05 μg g-1 in some registered products and 12.24-20.72 μg g-1 in yet to be registered products (Ang et al., 2004). A study of Asian remedies evaluated for levels of As, Pb and Hg reported values ranging from toxic to values exceeding public health guidelines for prevention of illness when consumed according to the direction given in or on the package (Garvey et al., 2001).
| Table 2: | Result of Pb and Cd content of some common Nigerian medicinal preparations (μg g-1) |
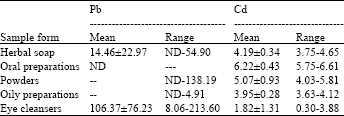 | |
Similarly, studies indicates that about 64% of such products in India contained significant amounts of Pb (Ernst, 2002).
The public health implication of the use of kohl, bint al dhahab and henna in the Middle East has been a source of worry to physicians and environmentalists. The modification of these products by the addition of various herbs or other substances in order to strengthen them has been observed to increase the Pb content of these products (Lekouch et al., 2001). Mean Pb concentration of 5.5 μg g-1 (range 1.29-16.48 μg g-1) was reported in henna samples used in Saudi Arabia (Al-Saleh and Coate, 1995). Large amounts of Pb, Sb and Cd have been observed in bint al dhahab, a herbal remedy common in Oman, used in the treatment of stomach complaints (Ernst, 1998). Bulk analyses of bint al dhahab indicated 91 g of PbO, 600 mg of antimony oxide and 50mg of Cd per 100 g of bint al dhahab (Worthing et al., 1995).
Because of the lack of requirement for quality control and safety and efficacy data, consumers cannot determine whether an herb’s active ingredients are actually in the product, whether the ingredients are bioavailable, whether the dosage is appropriate, whether the next product they buy will have the same components and what else is in the drug preparation besides the claimed ingredients (Desai et al., 2003).
Most of the preparations studied are used on the pediatric age group. This practice can result in baseline human exposure that may place large populations of infants and children at risk of developmental lead toxicity. The observation that this practice is common among low income earners corroborates the observation that poverty is crucial in the elevation of whole blood lead levels (Nordin et al., 1998; Bernard and McGeelin, 2003). Race, dietary habits, cigarette smoking, use of topically applied ophthalmic drugs and low socioeconomic status are some of the suggested obscure causes of senile cataract (Cekic, 1998). Elevated blood lead levels in children have been associated with behavioral problems, loss in intelligence and other neurological disorders (Kalavska 1992; Rabinowitz et al., 1992; Rosen, 1992). Children are more vulnerable to Pb exposure for three reasons: young children are more at risk of ingesting environmental Pb through the normal mouthing behavior and craving for non-food items referred to as pica, (Johnson and Tenuta, 1979; Francek et al., 1994; Lanphear et al., 2002; Brunekreef et al., 1981); absorption from the gastrointestinal tract is higher in children than in adults and the developing nervous system is thought to be far more vulnerable to the toxic effects of Pb than the mature brain (Lindskey and Schneider, 2003; Koller et al., 2004). Lead toxicity can affect several organ systems including the hematopoietic system, the peripheral and central nervous system, the kidney, the cardiovascular system and the reproductive system (Skerfving et al., 1998).
Studies have reported a lowering of blood lead levels in children with treatment with succimer. This however did not improve scores on tests of cognition, behavior, or neuro-psychological function in children with high blood lead levels (Rogan et al., 2001). This confirms the need for collective and concerted efforts to prevent Pb poisoning in children (Rosen and Mushak, 2001). The health implications of indulging in this practice can only be monitored by a survey of the blood lead levels of groups engaging in this practice because Pb determination in blood (B-Pb) is presently the prevailing indicator of Pb exposure and risk (Skerfving et al., 1998).
Even with compliance with the stipulate quality requirements for registration, strict regular surveillance is required as such products cannot be assumed to be safe from heavy metal contamination because of batch-to-batch inconsistencies (Dasai et al., 2003; Ang et al., 2004). There is an urgent need for periodic monitoring of herbal medicinal preparations in Nigeria and to seek means of protecting the consumer from such risks.
REFERENCES
- Al-Saleh, I.A. and L. Coate, 1995. Lead exposure in Saudi Arabia from the use of traditional cosmetics and medical remedies. Environ. Geochem. Health, 170: 29-31.
CrossRefDirect Link - Ang, H.H., K.L. Lee and M. Kiyoshi, 2004. Determination of lead in similax myosotiflora herbal preparations obtained in Malaysia. Int. J. Environ. Health Res., 14: 261-272.
Direct Link - Ang, H., K. Lee and M. Kiyoshi, 2005. Determination of lead in similax luzonensis herbal preparations in Malaysia. Int. J. Toxicol., 24: 165-171.
Direct Link - Bernard, S.M. and M.A. Mc Geelin, 2003. Prevalence of blood lead levels among US children 1 to 5 years of age and socioeconomic and demographic factors associated with blood lead levels 5 to 10 μg dLG1: The 3rd national health and nutrition examination survey, 1988-1994. Pediatrics, 112: 1308-1313.
Direct Link - Brunekreef, B., S.J. Veenstra, K. Biersteker and J.S.M. Boleij, 1981. The Arnhem lead study. Lead uptake by 1-to-3-years old children living in the vicinity of a secondary lead smelter in Arnhem the Netherlands. Environ. Res., 25: 441-448.
PubMedDirect Link - Crone, C.C. and T.N. Wise, 1998. Use of herbal medicines among consultation liaison population: A review of current information regarding risk, interactions and efficacy. Psychosomatics, 39: 3-13.
Direct Link - Desai, A.K. and G.T. Grossberg, 2003. Herbals and botanicals in geriatric psychiatry. Am. J. Geriatric Psychiatry, 11: 498-506.
Direct Link - Drew, A.K. and S.P. Myers, 1997. Safety issues in herbal medicine implications for the health professions. Med. J. Aust., 166: 538-541.
Direct Link - Ernst, E., 1998. Harmless herbs? A review of the recent literature. Am. J. Med., 104: 170-178.
CrossRefPubMedDirect Link - Ernst, E., 2002. Heavy metals in traditional Indian remedies. Eur. J. Clin. Pharmacol., 57: 891-896.
Direct Link - Ernst, E., 2003. erious adverse effects of unconventional therapies for children and adolescents: A systematic review of recent evidence. Eur. J. Pediatr., 162: 72-80.
CrossRefDirect Link - Ernst, E., 2004. Requested contribution: Risks of herbal medicinal products. Pharmacoepidemiol. Drug Safety, 113: 767-771.
Direct Link - Garvey, J.G., G. Hahn, R.V. Lee and R.D. Harbison, 2001. Heavy metal hazards of Asian traditional remedies. Int. J. Environ. Health Res., 11: 63-71.
Direct Link - Hamzah, A., C.N. Beh, S.B. Sarman, J.Y. Liow and L. Abugassa, 2004. Studies on elemental analysis of Chinese traditional herbs by neutron activation technique and their mutagenic effect. J. Radioanal. Nuclear Chem., 259: 499-503.
CrossRefDirect Link - Hearly, M.A., M. Aslam and O.A. Bamgboye, 1984. Traditional medicines and lead-containing preparations in Nigeria. Public Health, 98: 26-32.
Direct Link - Kalavska, D., 1992. Blood lead level as a criterion of global pollution. Bull. Environ. Contam. Toxicol., 48: 487-493.
CrossRefDirect Link - Lanphear, B.P., R. Hornung, M. Ho, C.R. Howard, S. Eberle and K. Knauf, 2002. Environmental lead exposure during early childhood. J. Paediatr., 140: 40-47.
Direct Link - Lekouch, N., A. Sedki, A. Nejmeddine and S. Gamon, 2001. Lead and traditional Moroccan pharmacopoeia. Sci. Total Environ., 280: 39-43.
CrossRefDirect Link - Lindsky, T.I. and J.S. Schneider, 2003. Lead neurotoxicity in children: Basic mechanisms and clinical correlates. Brain, 126: 5-19.
Direct Link - Lynch, E. and R. Braithwait, 2005. A review of the clinical and toxicological aspects of traditional (herbal) medicines adulterated with heavy metals. Expert Opin. Drug Safety, 4: 769-778.
Direct Link - Nnorom, I.C., J.C. Igwe and C.G. Oji-Nnorom, 2005. Trace metal contents of facial (make-up) cosmetics commonly used in Nigeria. Afr. J. Biotechnol., 4: 1133-1138.
Direct Link - Nordin, J., S. Rolnick, E. Ehlinger, A. Nelson, T. Arneson, L. Cherney-Stafford and J. Griffin, 1998. Lead levels in high-risk and low-risk young children in the Minneapolis St. Paul metropolitan area. Pediatrics, 101: 72-76.
Direct Link - Rabinowitz, M.B., J.D. Wang and W.T. Soong, 1992. Children`s classroom behavior and lead in Taiwan. Bull. Environ. Contam. Toxicol., 48: 282-288.
Direct Link - Rogan, W.J., K.N. Dietrich, J.H. Ware, D.W. Dockery and M. Salganik et al., 2001. The effect of chelation therapy with succimer on neuropsychological development in children exposed to lead. New Engl. J. Med., 344: 1421-1426.
Direct Link - Rosen, J.F. and P. Mushak, 2001. Primary prevention of childhood lead poisoning-the only solution. New Engl. J. Med., 344: 1470-1471.
Direct Link - Saper, R.B., S.N. Kales, J. Paquin, M.J. Burns, D.M. Eisenberg and R.B. Davis et al. 2004. Heavy metal content of ayurvedic herbal medicine products. J. Am. Med. Assoc., 292: 2868-2873.
Direct Link - Skerfving, S., L. Gerhardsson, A. Schutz and U. Stromberg, 1998. Lead-biological monitoring of exposure and effects. J. Trace Elements Exp. Med., 11: 289-301.
Direct Link - Susan, C., 2005. Herbal product contamination and toxicity. J. Pharm. Practice, 18: 188-208.
Direct Link - Vanderperren, R., M. Rizzo, L. Angenot, V. Haufroid, M. Jadoul and P. Hantson, 2005. Acute liver failure with renal impairment related to the abuse of senna anthraquinone glycosides. Ann. Pharmacother., 39: 1353-1357.
CrossRefDirect Link - Wojcikowski, K., D.W. Johnson and G. Glenda, 2004. Medicinal herbal extracts: Renal friend or foe? Part one: The toxicity of medicinal herbs. Nephrology, 9: 313-318.
Direct Link - Worthing, M.A., H.H. Sutherland and K. Al-Riyami, 1995. New information in the composition of bint al dhahab, a mixed lead monoxide used as a traditional medicine in Oman and the United Arab Emirates. J. Trop. Pediatr., 41: 246-247.
CrossRefDirect Link - Koller, K., T. Brown, A. Spurgeon and L. Levy, 2004. Recent developments in low level lead exposure and intellectual impairment in children. Environ. Health Perspect., 112: 987-994.
CrossRef








