ABSTRACT
The water in-crude oil emulsion has great importance in the oil industry. Experimental data are presented to investigate the stability of water-in-crude oil emulsions in both creaming and coalescence states were measured as a function of sodium chloride concentration. Also the stability of w/o emulsion is investigated over a wide range of parameters. These parameters are salt concentration (0-5.5%), mixing speed (800-1600 rpm), water concentration (10-80%) and temperature. For economic and operational reasons, it is necessary to separate the water completely from the crude oils before transporting or refining them. The emulsions were found to fall into three categories on the basis of both rheological properties and visual appearance: stable; mesostable and unstable. The present research found that, microwave radiation method can enhance the demulsification of water-in-crude oil emulsions in a very short time compared to the conventional methods.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2006.2895.2900
URL: https://scialert.net/abstract/?doi=jas.2006.2895.2900
INTRODUCTION
It is well known that the formation of water-in-crude oil emulsion is in the center of several economic and environmental problems. Water-in-crude oil emulsions can be encountered at many stages during drilling, producing, transporting and processing of crude oils. These increase the cost of production and transport of petroleum. Environmental problems arise because of the difficulty in cleaning up of the environment after oil-spillage by techniques such as burning, use of sorbants, use of dispersants and pumping (Mingyuan et al., 1992). Crude oil is a mixture of aliphatic, aromatic hydrocarbons and oxygen, nitrogen and sulphur containing compounds such as resins and asphaltenes. There is no doubt that the interfacially active components come from resins and asphaltenes of the crude oils. Many studies have proved that the primary mechanism of asphaltene of w/o emulsions is through the formation of viscous, cross-linked three dimensional film network with high mechanical rigidity (Kilpatrick et al., 2001). Authors (Sjoblom et al., 2003), (McLean et al., 1998) and (Strassner, 1968) were reported that, asphaltene and resins adsorb at the water-oil interfaces and form interfacial films that confer stability against phase separation. In most cases colloids absorb at the oil-water interface, providing steric hindrance to drop-drop coalescence. In addition, interfacially active molecules are also preferentially partitioned to the interface. The interfacial structure that arises is, therefore, a combination of surface-active molecules in association with colloidal particles. As reported by author Krawczyk et al. (1991), this can lead to extremely large resistances to film drainage and ultra-stable macro-emulsions. Depending on salt concentration, Bink et al. (2000) suggested three phenomena contributing to the emulsion stability. The first is the creaming process, which is enhanced as a result of Ostwald ripening and is governed primarily by the buoyancy motion of single drops. This process takes place at low electrolyte concentrations. The second is the flocculation process, which is dominated at high electrolyte concentration. The third process is coalescence, which takes place after creaming and flocculation process. Zapryanov et al. (1983) reported that coalescence occurs in three steps: (1) approach of the droplets through the continuous phase; (2) deformation of the droplets to form a thin between them; and (3) thinning of this film to a critical thickness, below which the droplets coalesce. Also Auflem (2002) reported that the main mechanisms that influence coalescence are film drainage and film rupture.
The aim of this study is conducted to investigate the influence of salt concentration, mixing speed, water concentration, as well as temperature on emulsion stability. Present findings showed there is a strong relation between these parameters and emulsion stability.
MATERIALS AND METHODS
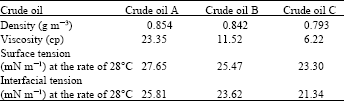
Materials: The materials used in this study are three crude oils; Crude oil A; from Iran oilfield, crude oil B and C from Malaysia oil fields, the crude oils were obtained from Petronas refinery at Melaka. Their respective compositions are given in Table 1.
Emulsion preparation: Water-in-crude oil emulsions were prepared by adding internal phase (water) slowly to the mixing phase (solution with stabilizer) in a glass beaker (900 mL) with ranges by volume of water and oil phase. Emulsions were agitated vigorously using a standard three blade propeller at speed from 800 to 1800 rpm and temperature 28-30°C for 10 min. In order to prepare water-in-oil emulsions, the agent in-oil method was followed, that is; in this study, the emulsifying agent dissolved in the continuous phase (oil) and water added gradually to the mixture (oil + emulsifying agents). The volume of the water settled to the bottom was read from the scale on the beaker with different times. The amount of water separation in percent was calculated as separation efficiency from the water observed in the beaker.
The prepared emulsions were used to check for w/o or o/w emulsions. All emulsions investigated were type of water-in-oil emulsions (w/o).
Separation of crude oil fractions: The interfacially active fractions, such as the saturate, aromatic, resins and asphaltenes used in these experiments were separated from crude oil obtained from Petronas Refinery. The separation of asphaltene fractions from the crude oil was carried out by pentane precipitation. Two hundred grams of crude oil was agitated with 2500 mL of pentane at room temperature (30°C) for 15 min. The mixture was then left to stand for 6 h. The precipitated asphaltene fraction was filtered and washed with smaller portion of pentane and dried.
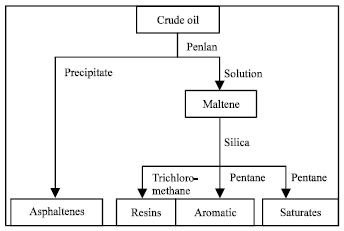 | |
| Fig. 1: | Separation of crude oil fractions |
| Table 1: | Physico-chemical properties of crude oils; A, B and C |
 | |
| Table 2: | Physical properties of crude oils used in the experiments |
 | |
The filtrate from above was then poured on a column containing silica. The wax fraction was extracted by 1300 mL of pentane. The remaining resin fraction was then extracted by 2000 mL of benzene/methanol (50-50% v/v) mixture. The remaining oil fraction was used for the preparation of water-oil (W/O) emulsions as shown in Fig. 1. The physical properties of the crude oils and their fractions were shown in Table 2.
RESULTS AND DISCUSSION
The percentage of water separated is the best indicator of emulsion stability. The prepared water-in-crude oil emulsions were placed in a baker and observed over 30 h (1800 min). The volume of water settled to the bottom was read from the scale on the beaker with different times. The amount of water separation in percent was calculated as separation efficiency (e) from volume of water observed in the beaker as follows:
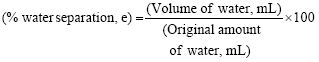 | (1) |
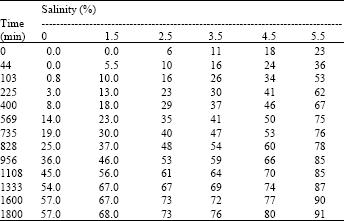
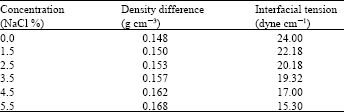
The results obtained from the sedimentation experiment and Eq. (1) as a function of salinity (NaCl) is shown in Table 3 and Fig. 2. Separation of water layer was used as a measure of physical instability for water-in-oil emulsions. As illustrated in Table 3 and Fig. 2, it is pronounced that separated water was increased for emulsions having high salt concentrations. In the sedimentation experiments, a continuous oil phase, the amount of water separated with high salt concentration (5.5%) was found as 91% compared to 0% salt concentration which produced water separation amount is 57% after 1800 min. The emulsion with relatively lower salt concentration (1.5%) had separated into three layers after 735 min, while after 1800 min it was separated into two clear phases and the maximum amount of water separated was found as 68%.
| Table 3: | Data for emulsion stability test, at different salinity (NaCl %) |
 | |
| Table 4: | Data for interfacial tension test at different salinity (NaCl %) |
 | |
| Table 5: | Effect of volume fraction on emulsion viscosity |
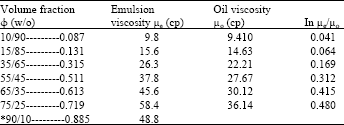 | |
| * Note: when the volume fraction of emulsion reached (0.885), the emulsion was inverted from water-in-oil (w/o) to oil-water (o/w) emulsions (Fig. 7). | |
The influence of salt concentration (NaCl%) on crude oil emulsion stability was illustrated as follows: 0% NaCl (57%v/v), 1.5% NaCl (68%v/v), 2.5% NaCl (73% v/v), 3.5% NaCl (76% v/v), 4.5% NaCl (80% v/v) and 5.5% NaCl (91% v/v), respectively.
Figure 3 shows the separation time required as function of salinity for emulsion separation. A long duration is necessary for separation of water-in-oil emulsions with no heat and salt is remarkable for sedimentation experiments. However, an unstable emulsion can be eventually destroyed leading to complete separation of the two phases. In this regard, there is no stabilization in the case of water-in-oil emulsion with relatively high salt concentration (5.5%).
To understand this observation, one may consider the characteristic changes in the oil water emulsion behavior as a result of salt (NaCl) addition. From the diffuse ion theory, it is known that for the same water content, as salt concentration increases, the internal energy of the system increases Mohamed et al. (2003).
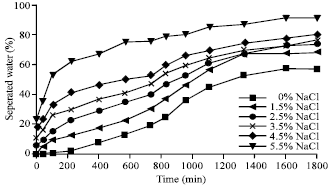 | |
| Fig. 2: | Emulsion stability evaluation under sedimentation process; percentage of separated water vs. time for w/o emulsions at different NaCl concentration. |
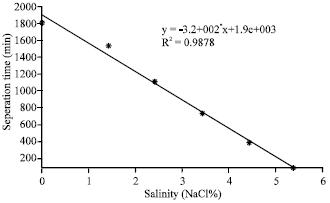 | |
| Fig. 3: | Separation time for complete emulsion breakdown at different salinity (%) |
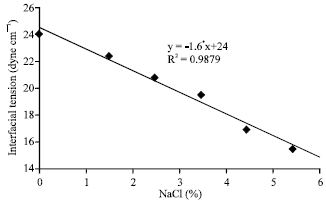 | |
| Fig. 4: | The interfacial tension of crude oil at different (NaCl%) concentrations |
Consequently, the emulsions are not thermodynamically stable and droplets (water) merge with each other to produce big droplets and increase the coalescence rate. Coalescence is one of the possible mechanisms of destruction of emulsions, which occurs when the energy of adhesion between two droplets is larger than the turbulent energy causing dispersion, Calderon and Poulin (1999).
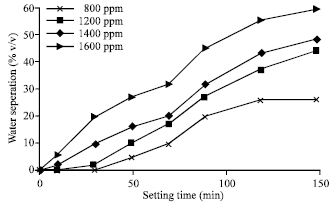 | |
| Fig. 5: | Effect of surfactant concentration (ppm) on emulsion stability (50-50% w/o) |
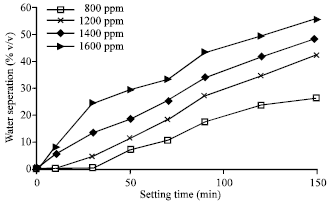 | |
| Fig. 6: | Effect of Surfactant concentration (ppm) on emulsion stability (20-80% w/o) |
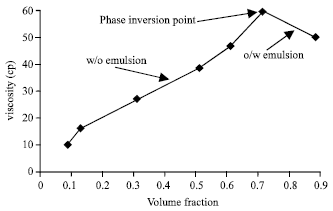 | |
| Fig. 7: | Effect of volume fraction on emulsion viscosity |
Interfacial tension is an important factor in emulsion stability. It arises because the intermolecular forces in the interior of a liquid are equal in all directions. The interfacial tension falls to low values at higher sodium chloride (NaCl%) concentration as shown in Table 4 and Fig. 4, respectively. To investigate the effect of salt on the emulsion stability, one would into the free energy of the system. For a constant volume of droplets, as the droplet stretches, its surface area as well as the free energy of the surface increases.
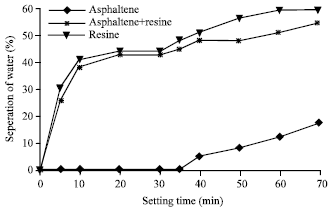 | |
| Fig. 8: | Stability of (w/o) emulsions with asphaltenes, resin and a mixture of asphaltenes and resins of crude oil A |
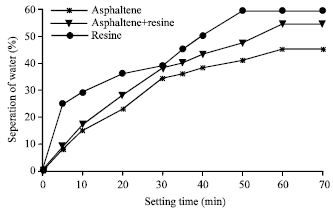 | |
| Fig. 9: | Stability of (w/o) emulsions with asphaltenes, resin and a mixture of asphaltenes and resins of crude oil B |
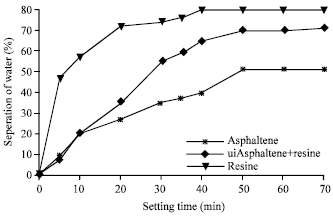 | |
| Fig. 10: | Stability of (w/o) emulsions with asphaltenes, resin and a mixture of asphaltenes and resins of crude oil C |
On the other hand, the longer the droplet dimension and smaller the radius, the lower is the rotational energy. The results shown in Table 4 indicated that as salt concentration (NaCl%) increases, the density difference increases and interfacial tension decreases. As shown in Table 4, at salt concentration NaCl (0%) the density difference is 0.148 g cm-3 and the interfacial tension is 24.00 dyne/cm, while when the concentration of NaCl reached (5.5%), the density difference increased to 0.168 g cm-3 and the interfacial tension decreased to 15.30 dyne/cm. with increase of salinity NaCl%, the chance for the molecules to collide with each other increases.
The effect of surfactant concentration and volume fraction on emulsion stability is studied by two ratios of water-in-oil emulsions that; 50-50 and 20-80% water-in-oil emulsions. It found that as the volume fraction (φ) decreases, the separation time for water to separate from the emulsion decreased. In this regard, the volume fraction (φ) for 50-50% w/o is 0.459 while for 20-80% w/o is 0.175. The concentration of surfactant mainly affects the viscosity of emulsion. Experiments show that the viscosity of emulsion increases as surfactant concentration increases. Figure 5 and 6 show the effect of surfactant concentrations for 50-50 and 20-80% w/o on emulsion stability, respectively.
The effect of volume fraction of internal phase (water) on emulsion stability was also studied with emulsion viscosity. Table 5 shows the viscosity of emulsion increases greatly as internal volume fraction (φ) increases, the result leads to emulsion stability. An interesting and surprisingly, when the volume fraction reached (φ = 0.885) the emulsion converted from w/o to o/w as shown in Fig. 7.
The stability of emulsions again was determined by measuring the water separation from the emulsions at room temperature (30°C) using surface-active agents (asphaltene and resin). The interfacially active fractions such as asphaltene and resins used in this study were extracted from the different crude oils. When surface –active agents added to the emulsions, the interfacially active agents were adsorbed or deposited at the interface of the oil/water system. The process takes place over a period of time until the system attains equilibrium. Plots describing the stabilities of emulsion system for the three crude oils emulsion with asphaltene and resins as interfacially active components are shown in Fig. 8-10 for crude oils, A, B and C, respectively. From these Fig. 8-10 crude oil A was found more stable compared to crude oil B and C. The maximum percentage of water separated was 58%. While for crude oil B and C, the percentage of water separation were found as 60 and 80%, respectively. Crude C is less stable. These figures shown that the emulsions made by asphaltenes were more stable than those made by resins or mixture agents (asphaltene+resins).
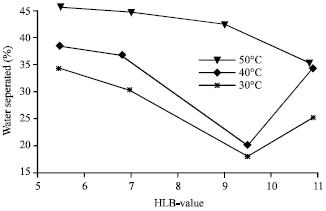 | |
| Fig. 11: | Effect of temperature on (w/o) Emulsion Stability for 50-50% w/o emulsions |
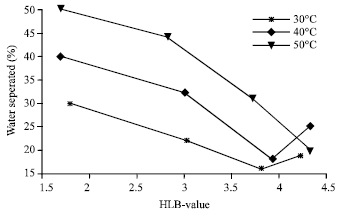 | |
| Fig. 12: | Effect of temperature on (o/w) emulsion stability for 20-80% w/o emulsions |
The effect of temperature on emulsion stability was studied. In ordinary emulsification, a change of temperature has only an indirect effect as result of altering the interfacial tension, adsorption of emulsifier and viscosity. However, significant changes in temperature cause changes in interfacial tensions, viscosities, nature (hydrophilicity-lipophilicity) of the surfactants, vapor pressures of the liquid phases and in the thermal agitation of the molecules. Thus, emulsions tend to be very sensitive to temperature changes. Emulsions are more stable when the temperature is near the point of minimum solubility of emulsifying agents. Emulsion stability decreases when temperature in increased. Figure 11 depicted the effect of Hydrophilic-Lipophilic Balance (HLB) and temperature on emulsion stability for 50-50% w/o, while Fig. 12 shows for 20-80% w/o, respectively.
CONCLUSIONS
The water-in-crude oil emulsions have great important in the oil industry. The formation and stabilization of water-in-oil (w/o) emulsions are investigated over a wide range of parameters. These parameters are surfactant concentrations, temperature, volume fractions of dispersed phase, phase ratio water-oil (10-80%) and surfactant concentration (800-1800 ppm).
The physical properties of water-in-crude oil emulsions in terms of density, viscosity, surface tension and interfacial tension were also measured. The SARA-separation analysis was used to measure the surface-active molecules.
REFERENCES
- Bink, B.P., W.G. Cho, P.D.I. Fletcher and D.N. Petsev, 2000. Stability of oil in water emulsions in a low interfacial tension system. Langmuir, 16: 1025-1034.
Direct Link - Mohamed, A.M.O., M. El Gamal and A.Y. Zekri, 2003. Effect of salinity and temperature on water cut determination in oil reservoirs. J. Petroleum Sci. Eng., 40: 177-188.
Direct Link - Sjoblom, J., N. Aske, I.H. Auflem, O. Brandal and T.E. Havre et al., 2003. Our current understanding of water-in-crude oil emulsions. Recent characterization techniques and high pressure performance. Adv. Colloid Interface Sci., 100: 399-473.
Direct Link








