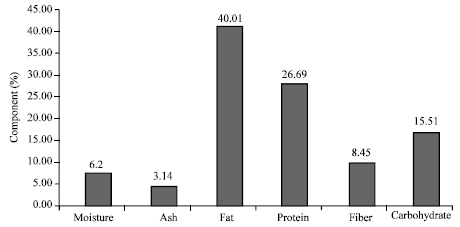
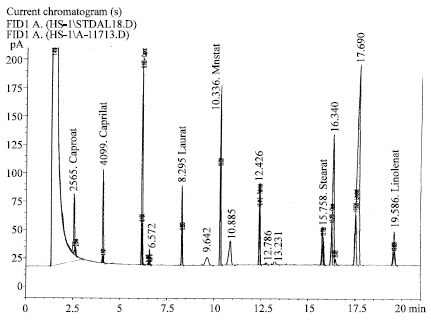
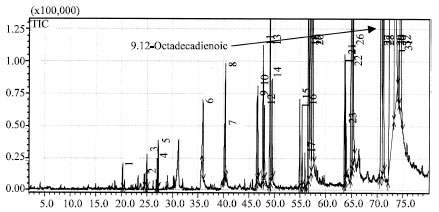
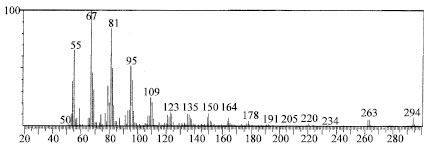
Research Article
Gas Chromatography and Gas Chromatography-mass Spectrometry Analysis of Indonesian Croton tiglium Seeds
Department of Agro industrial Technology, Faculty of Agricultural Technology, Bogor Agricultural University, Bogor, Indonesia
Djumali Mangunwidjaja
Department of Agroindustrial Technology, Faculty of Agricultural Technology, Bogor Agricultural University, Bogor, Indonesia
Sapta Raharja
Department of Agroindustrial Technology, Faculty of Agricultural Technology, Bogor Agricultural University, Bogor, Indonesia
Leonardus B.S. Kardono
Department of Chemistry, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, Bogor, Indonesia
Dyah Iswantini
Department of Chemistry, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, Bogor, Indonesia