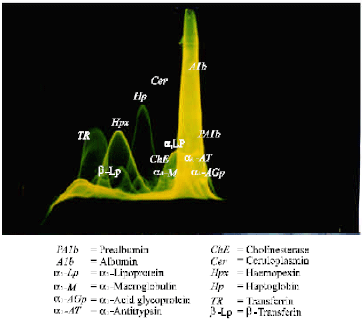
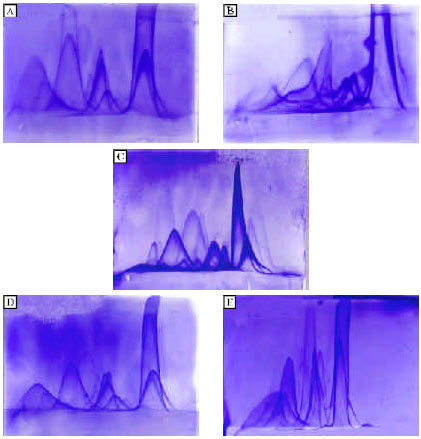
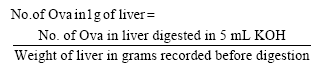Research Article
Effect of Citrus reticulata on Serum Protein Fractions of Mice After Schistosoma mansoni Infection
Departments of Medicinal Chemistry, National Research Centre, Dokki, Cairo, Egypt
Mona H. Hetta
National Research Center, Dhokki, Cairo, Egypt