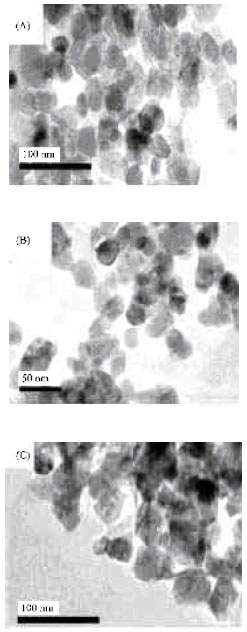
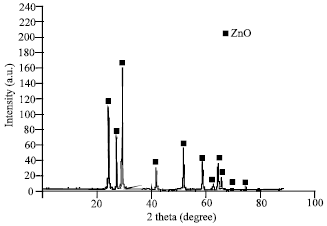
Research Article
The Effect of PVP Addition and Heat-treatment Duration on Zinc Oxide Nanoparticles
Institute of Nano-Science and Technology, Central China Normal University, Wuhan 430079, China
Yiwen Tang
Institute of Nano-Science and Technology, Central China Normal University, Wuhan 430079, China
Zhan Xin
Institute of Nano-Science and Technology, Central China Normal University, Wuhan 430079, China
Zhijie Jia
Institute of Nano-Science and Technology, Central China Normal University, Wuhan 430079, China