Research Article
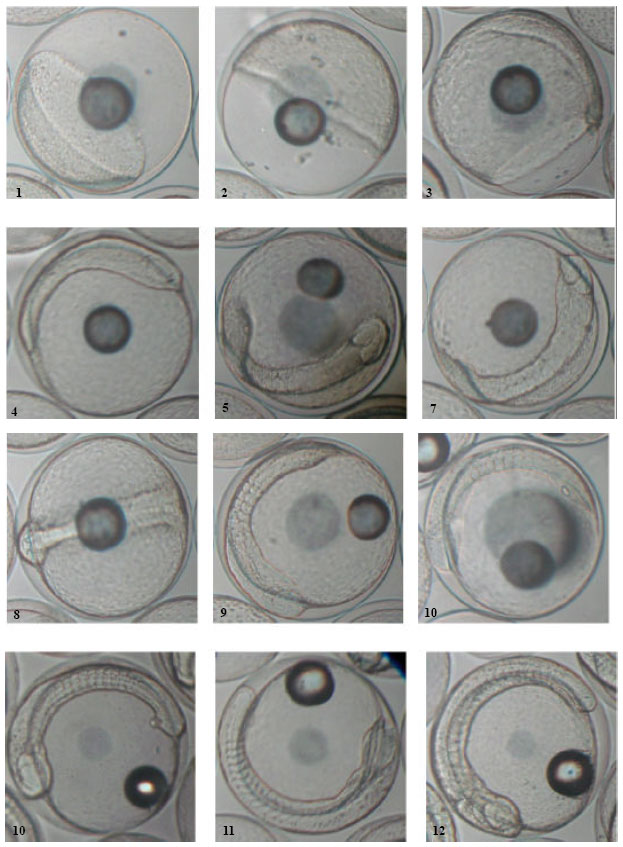
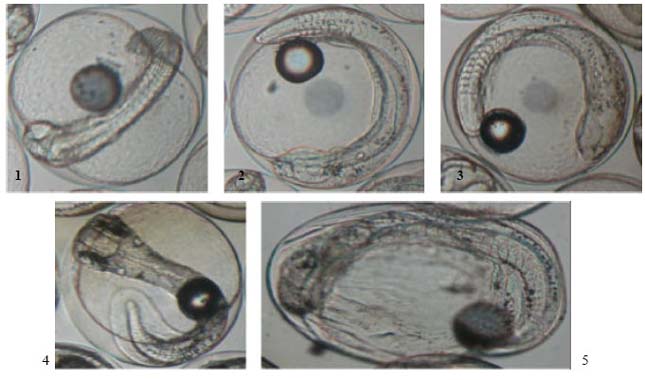
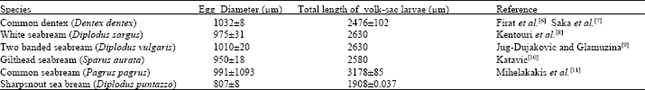
Determination of Embryonic Development Stages of Sharpsnout Seabream (Diplodus puntazzo Cetti, 1777) Eggs in Rearing Conditions
Department of Aquaculture, Faculty of Fisheries, Ege University, 35440, Uria-lskele, In-nir, TURKEY
Kursat Firat
Department of Aquaculture, Faculty of Fisheries, Ege University, 35440, Uria-lskele, In-nir, TURKEY
Sahin Saka
Department of Aquaculture, Faculty of Fisheries, Ege University, 35440, Uria-lskele, In-nir, TURKEY
Musa Bulut
Department of Aquaculture, Faculty of Fisheries, Ege University, 35440, Uria-lskele, In-nir, TURKEY