ABSTRACT
The influence of ignition manner on micropyretic synthesis of low exothermic reaction with Ni+Al is numerically investigated in this study. The different ignition powers and ignition regions are chosen to illustrate the influences on the length of pre-heating zone and propagation velocity of the specimens. The numerical results indicate that a lower ignition power or igniting in the center region accelerates the heat loss. Thus, the length of pre-heating zone is increased and the average propagation velocity is correspondingly decreased. In the extreme case, the combustion front may not complete the propagation for the reaction with a lower pre-exponential factor. In addition, it is also shown that a higher ignition power consumes the additional ignition energy to ignite the specimens. Such an increase in the required ignition energy enhances the processing cost, but also probably results in the unstable propagation. The optimal ignition manner for micropyretic synthesis of Ni+Al reaction is generated in this research.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2005.202.206
URL: https://scialert.net/abstract/?doi=jas.2005.202.206
INTRODUCTION
Many exothermic non-catalytic solid-solid or solid-gas reactions, after being ignited locally, can release enough heat so as to sustain the self-propagating combustion front throughout the specimen without additional energy[1-8]. Since the 1970’s, this kind of exothermic reaction has been used in the process of synthesizing refractory compounds in the former Soviet Union[2]. This novel technique, so-called micropyretic/combustion synthesis, has been intensively studied for process implication[1-3]. This technique employs exothermic reaction processing, which circumvents difficulties associated with conventional methods of time and energy-intensive sintering processing. The advantages of micropyretic synthesis also include the rapid net shape processing and clean products.
Several numerical and analytical models of micropyretic synthesis in a composite system have been well developed[3,8-10]. Lakshmikantha and Sekhar[8] firstly explored the numerical model that includes the effects of dilution and porosity and melting of each constituent of the reactants and products[3,8]. The analytical modeling of the propagation of the combustion front in solid-solid reaction systems has also been reported[9]. The analytical model gives good results when compared with the experimentally determined numbers and the numerically calculated values. In addition, a multi-dimensional numerical model and dynamic modeling of the gas and solid reaction have also been carried out to illustrate the effects of various parameters on the micropyretic synthesis[8-10]. These numerical and analytical analyses provide the better understanding of the reaction sequence during micropyretic synthesis reactions.
Since the micropyretic synthesis is normally occurred in a few seconds, any small variations in the ignition manners have been reported to dramatically influence the propagation during micropyretic synthesis[11]. The selection of the ignition manner is important while materials processing by micropyretic synthesis. Thus, the numerical calculation is used to investigate the correlation of the ignition manners with the propagation velocity in this study. The low exothermic Ni-Al reactions with the different pre-exponential factors, which correspond to the various reactant sizes, are chosen to illustrate the effects of the ignition manners. The results generated in this study will be used to provide the useful information for selecting the optimal igniting manners.
Numerical calculation procedure: The middle-difference approximation and an enthalpy-temperature method coupled with Guass-Seidel iteration procedure are used to solve the energy equations of the micropyretic synthesis problems. The detailed numerical calculation procedure has been reported in the previous studies[4,7]. In the computational simulation, a one-dimensional sample of 1 cm long is divided into 1201 nodes (regions) to calculate the local temperature using an enthalpy-temperature method. First, the proper initial and boundary conditions are used to initialize the temperatures and enthalpies at all nodes. The initial conditions in the simulation are taken as follows: (1) At the ignition node, at time t≥0, the temperature is taken to be the adiabatic combustion temperature. (2) At the other nodes, at time t=0, the temperatures are taken to be the same as the substrate temperature. During the ignition, the specimens are heated at the different ignition powers. The proceeding of the reaction is assumed in the isolate thermal environment and the ignition power is immediately removed as the combustion front starts to propagate.
The various thermophysical/chemical parameters, such as thermal conductivity, density and heat capacity of the reactants and product are also assumed to be independent of temperature, but they are different in each state. The average values of these parameters vary when the reaction proceeds, depending upon the degree of reaction. In this study, the combustion temperature is defined as the highest reaction temperature during micropyretic synthesis and the propagation velocity is the velocity of a propagating combustion front.
The calculations are normally performed 500 to 2000 times, depending upon the calculated thermal parameters; to make all 1201 sets (nodes) meet the criterion for each time step. At least 600 time steps are calculated to allow the propagation of the combustion front across the 1 cm long specimen completely. The pre-exponential factors in this study are respectively taken as 9x108 and 9x107 s-1, which correspondingly denote the different reactant sizes and reaction barriers. In this study, the porosities of the reactants and product are both taken to be 0%.
RESULTS AND DISCUSSION
Nickel aluminum compound (Ni:Al atomic ratio=1:1) without diluent is chosen to illustrate the effect of the ignition manner on micropyretic synthesis. Figure 1 shows the temperature profiles of combustion fronts at various times along the Ni+Al specimens with the ignition powers of 500 Joule and 1500 Joule, respectively. The micropyretic reaction is ignited at the position 0 cm and the combustion front starts to propagate from left to right. The interval time step between two consecutive time steps (profiles) is 0.001 s. It can be seen from Fig. 1a that the combustion front starts to propagate at 0.006 s after the ignition as the ignition power of 500 J s-1 is used. As the ignition power is further increased to 1500 J s-1, the rate of the energy supply is correspondingly increased. The combustion front thus takes the less time, 0.003 s, to start propagating, as shown in Fig. 1b. Table 1 shows the required ignition time and energy for the specimens with the different ignition powers.
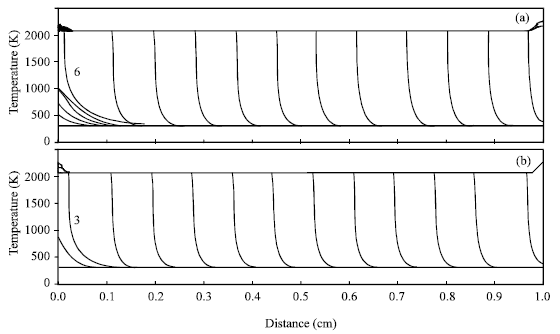 | |
| Fig. 1: | Time variations of the combustion front temperature along the Ni+Al specimen with the pre-exponential factor of 9x108 s-1. The specimen is ignited from the left end. The interval time between two consecutive time steps (profiles) is 0.001 s. The numbers 6 and 3, which, respectively denote the sixth time step (0.006 s) and the third time step (0.003 s) after ignition, are the beginnings of the propagation. The igniting powers in (a) and (b) are 500 and 1500 J s-1, respectively |
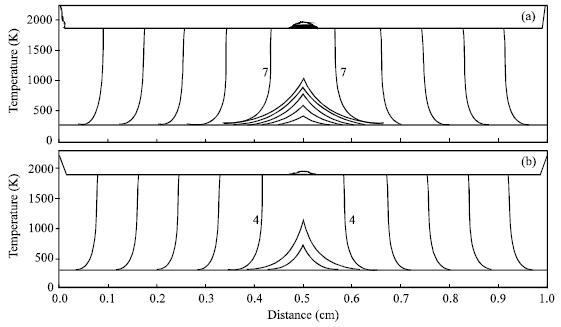 | |
| Fig. 2: | Time variations of the combustion front temperature along the Ni+Al specimen with the pre-exponential factor of 9x108 s-1. The specimen is ignited at the center. The interval time between two consecutive time steps (profiles) is 0.001 s. The numbers 7 and 4, which, respectively denote the seventh time step (0.007 s) and the forth time step (0.004 s) after ignition, are the beginnings of the propagation. The igniting powers in (a) and (b) are 500 and 1500 J s-1, respectively |
| Table 1: | The required ignition time and the required ignition energy for the specimens with different igniting powers. The pre-exponential factor is taken as 9x108 1/s |
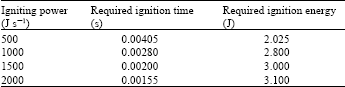 | |
| Table 2: | The calculated initial velocity during the first 0.0002 s after propagation for the specimens with different igniting powers. The pre-exponential factor is taken as 9x108 1/s |
 | |
It is noted that an increase in the ignition power decreases the ignition time required for starting of the propagation. However, the required ignition energy is noted to increase with the increase in the ignition power. This is because the higher ignition power provides a higher energy in each time interval. In the last time step just before the propagation, it is always found that the excessive energy is supplied to the specimen which only needs the less energy to propagate. The supplied energy easy exceeds a critical energy required for propagation.
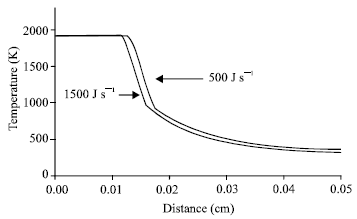 | |
| Fig. 3: | Time combustion temperature profiles after combustion front propagation. The X-coordinate from 0 indicates the start of the reaction zone |
Thus, the over-heating of the specimen is noted and the more energy for igniting a micropyretic reaction is consumed.
In this study, the effect of ignition region on micropyretic synthesis is also investigated. Figure 2 shows the temperature profiles for the specimens are ignited in the middle. As expected, the less ignition time is taken to propagate the combustion front as the higher ignition power is used. In addition, Fig. 2 also illustrates that the ignition times for the specimens ignited in the middle are more than those ignited from the one end. In this study, the reaction is assumed to occur in the isolate thermal system.
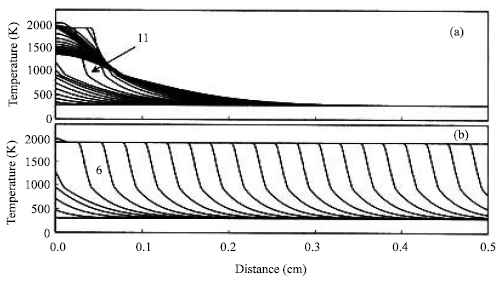 | |
| Fig. 4: | Time variations of the combustion front temperature along the Ni+Al specimen with the pre-exponential factor of 9x107 s-1. The specimen is ignited at the left end. The interval time between two consecutive time steps (profiles) is 0.001 s. The numbers 11 and 6, which, respectively denote the eleventh time step (0.011 s) and the sixth time step (0.006 s) after ignition, are the beginnings of the propagation. The igniting powers in (a) and (b) are 500 and 1500 J s-1, respectively |
Thus, the thermal energy is only transferred to one (right) direction as the specimen is ignited from the left end. When the specimen is ignited in the middle, the thermal energy is simultaneously transferred to both (left and right) directions. The heat loss in the ignition region is thus enhanced as compared with the specimen ignited from the left end. Therefore, Fig. 2 shows that it takes the more time to ignite in the middle as compared with that ignited from the left end.
During igniting the specimens, it offers the sufficient time to have the thermal energy transferring to the next region (pre-heating zone) when the ignition time is increased. The energy transferred to the pre-heating zone increases the initial temperature of this zone, further aiding the reaction to proceed after the ignition at the initial stage of propagation. Table 2 shows that the propagation velocity at the initial stage is higher for the specimen with the small ignition power or the specimen is ignited in the middle. The velocities for the specimens with the different ignition powers are then converged to the constant values as the combustion front further propagates. Figure 1 and 2 both show that the combustion fronts for the specimens with different ignition powers almost take the same time to complete propagation.
The temperature profiles affected by the ignition powers are also studied. Figure 3 illustrates the temperature gradients in the reaction zone and the pre-heating zone for the specimens with different ignition powers. As discussed before, a decrease in the ignition power increases the amount of heat transferred to the pre-heating zone. Thus, the lengths of reaction zone and pre-heating zone are increased, as shown in Fig. 3. Even this leads to an increase in the initial temperature and further enhancing combustion temperature and propagation velocity at the initial stage. However, the previous study[4] also shown such an increase in the length of pre-heating zone aids to disperse the energy during the propagation. It further decreases the propagation velocity and probably stops the propagation. Figure 4 shows the temperature-time variations with the distance for the specimens with lower pre-exponential factor, 9x107 1/s. It has been reported that an increase in the pre-exponential factor is considered equivalent to better spread and improving contact efficiency, further decreasing the kinetics of the reaction. Hence, it is expected that the propagation velocity is decreased as the pre-exponential factor decreases. For a given higher ignition power (1500 J s-1), the propagation velocity is dramatically decreased as the pre-exponential factor is decreased from 9x108 1/s (Fig. 1b) to 9x107 1/s (Fig. 4b). Note from Fig. 1b that the combustion front takes 0.012 s (~12 time step, 0.001 s/time step) to complete propagation. The combustion front in Fig. 4b only propagates to ~0.3 cm in the same time interval. For the specimens with lower ignition power (500 J s-1) and pre-exponential factor (9x107 1/s), the propagation velocity is noted to decrease and the combustion front is found to extinguish in the half way (Fig. 4b). Therefore, it can be concluded that a lower ignition power decreases the propagation velocity of a combustion front and probably stops the combustion front of the reaction with the lower pre-exponential factor.
CONCLUSION
The influence of ignition manner on micropyretic synthesis of low exothermic reaction with Ni+Al has been numerically investigated in this study. The numerical results have shown that a lower ignition power or igniting in the middle accelerates the heat loss. Thus, the length of pre-heating zone is correspondingly increased, further decreasing the propagation velocity. In the extreme case, the combustion front may not complete the propagation for the reaction with the lower ignition power and pre-exponential factor. The results generated in this study indicate that a higher ignition power prevents a combustion front from extinguish and aids to increase the propagation velocity. However, the numerical results also show a higher ignition power consumes the additional ignition energy to ignite the specimens. Such an increase in the required ignition energy enhances the processing cost, but also probably results in the over-heating and unstable propagation during the reaction. Therefore, an optimal ignition manner is required to choose for a stable propagation of micropyretic synthesis.
ACKNOWLEDGMENTS
The supports from the National Center for High-Performance Computing (y48hpl00) and National Science Council in Taiwan (NSC93-2216-E-228-001) are acknowledged.
REFERENCES
- Munir, Z.A., 2000. Modeling and experimental studies on the effect of thermophysical properties on field-activated combustion synthesis reactions. Pure Applied Chem., 72: 2177-2186.
CrossRefDirect Link - Merzhanov, A.G. and B.I. Khaikin, 1998. Theory of combustion waves in homogeneous media. Prog. Energy Combust. Sci., 14: 1-98.
CrossRefDirect Link - Lakshmikantha, M.G., A. Bhattacharya and J.A. Sekhar, 1993. Numerical modeling of solidification combustion synthesis. Metallurgical Mater. Trans. A, 23: 23-34.
Direct Link - Li, H.P., 2003. Numerical study of the second ignition for combustion synthesizing Ni-Al compounds. Metallurgical Mater. Trans. A, 34: 1969-1978.
CrossRefDirect Link - Li, H.P., 2003. The numerical simulation of the heterogeneous composition effect on the combustion synthesis of TiB2 compound. Acta Materialia, 51: 3213-3224.
Direct Link - Li, H.P., 2002. Investigation of propagation modes and temperature/velocity variation on unstable combustion synthesis. J. Mater. Res., 17: 3213-3221.
CrossRefDirect Link - Lakshmikantha, M.G. and J.A. Sekhar, 1994. Analytical modeling of the propagation of a thermal reaction front in condensed systems. J. Am. Ceramic Soc., 77: 202-210.
Direct Link - Lakshmikantha, M.G. and J.A. Sekhar, 1993. An investigation of the effect of porosity and diluents on micropyretic synthesis. Metallurgical Mater. Trans. A, 24: 617-628.
CrossRefDirect Link - Subramanian, V., M.G. Lakshmikantha and J.A. Sekhar, 1996. Modeling of sequential reactions during micropyretic synthesis. Metallurgical Mater. Trans. A, 27: 961-972.
Direct Link - Lee, W.C. and S.L. Chung, 1995. Ignition phenomena and reaction mechanisms of self-propagating high-temperature synthesis reaction in the Ti+C system. J. Mater. Sci., 30: 1487-1494.
Direct Link








