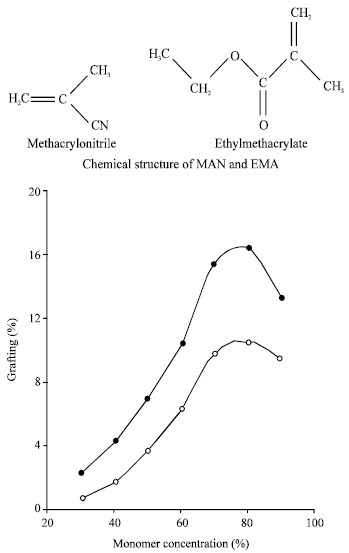
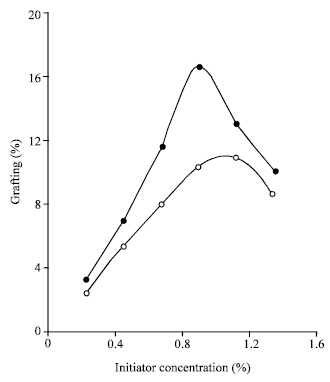
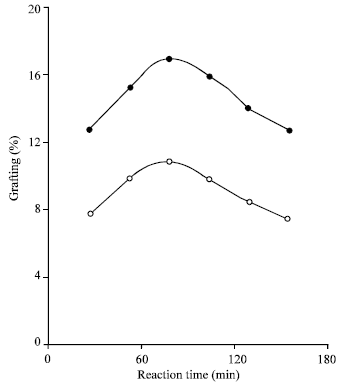
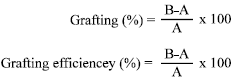Research Article
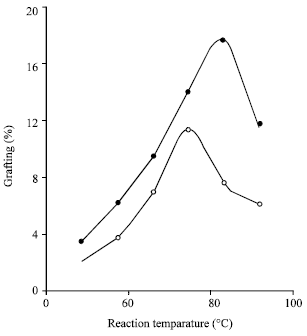
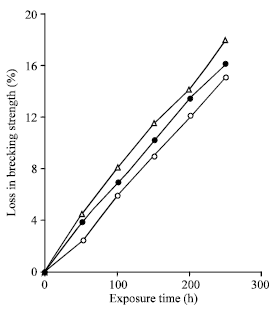
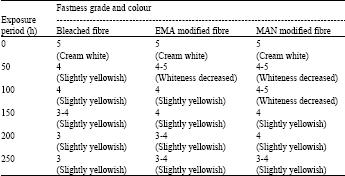
Grafting of Methacrylonitrile and Ethyl Methacrylate onto Jute Fibre: Physico-chemical Characteristics of Grafted Jute
Department of Applied Chemistry and Chemical Technology, Rajshahi University, Rajshahi 6205, Bangladesh
Faisul Islam Farouqui
Department of Applied Chemistry and Chemical Technology, Rajshahi University, Rajshahi 6205, Bangladesh
Md. Abu Hanif
Department of Applied Chemistry and Chemical Technology, Rajshahi University, Rajshahi 6205, Bangladesh
G.M. Shafiur Rahman
Department of Materials Science and Technology, Rajshahi University, Rajshahi 6205, Bangladesh
Md. Asadul Hoque
Department of Materials Science and Technology, Rajshahi University, Rajshahi 6205, Bangladesh