Research Article
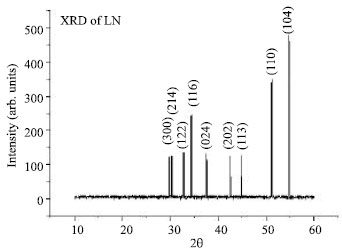
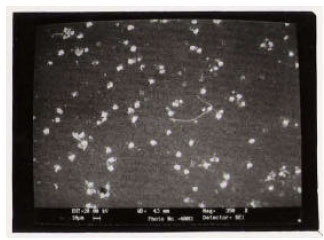
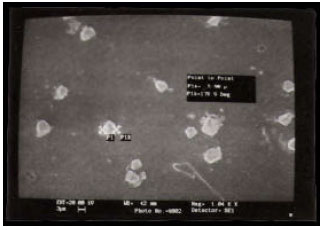
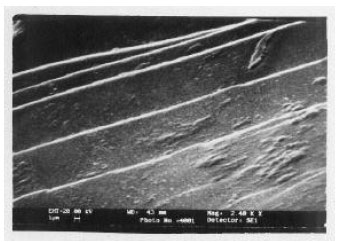
Structural Investigations on Lithium Niobate Grown by Czochralski Technique
Growth Centre, Anna University, Chennai 600 025, India
V. Vaithianathan
Growth Centre, Anna University, Chennai 600 025, India
S. Moorthy Babu
Growth Centre, Anna University, Chennai 600 025, India
P. Ramasamy
Growth Centre, Anna University, Chennai 600 025, India















jegatheesan Reply
This is Jegatheesan, working as a Lecturer in Private Engineering College and also doing research in Nonlinear Optics. The paper is very useful to go through my research in a correct root. The way of presentations in paper is very nice.