ABSTRACT
A Carbon Paste Electrode (CPE) was fabricated using lectin isolated from black sea cucumber (Holothuria scabra Jaeger). The prepared electrode was found to have high affinity towards mercury (II) ions. Analysis of mercury (II) was conducted by Differential Pulse Adsorptive Stripping Voltammetry (DPAdSV) using 0.1 M NaOH as supporting electrolyte. The following conditions gave the best results; pH 7, 20% modifier composition, 240 sec accumulation time and 300 sec deposition time. Electrode regeneration was successfully done by soaking the electrode for at least 5 min in 0.01 M EDTA solution. A linear relationship was observed at the range of 0.1 to 5 mg L -1 with a correlation coefficient of 0.9821. The limit of detection was calculated as 0.0474 mg L -1. Analysis for mercury content in a laboratory waste sample using the lectin modified electrode gave comparable result as that of AAS method.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2005.1461.1465
URL: https://scialert.net/abstract/?doi=jas.2005.1461.1465
INTRODUCTION
Chemically Modified Electrodes (CME) have received considerable attention in recent years[1]. The CME can accumulate metal ions on the basis of the interaction with a functional group on the electrode surface. In addition to its sensitivity, which is useful in analytical chemistry, other advantages of the CME include the detection of electroinactive substances and elimination of interferences from the sample matrix[2].
In particular, a Carbon Paste Electrode (CPE) is a type of CME which can be easily prepared and regenerated. CPE, which make use of graphite powder mixed with various water-immiscible organic binders (pasting liquids)[3] offer a renewable and modified surface, low cost and very low background current contributions[4-6]. CPE containing organic ligands that serve as modifiers made possible the accumulation and detection of metals such as copper (I)[7], nickel (II)[8], silver (I)[9], lead (II)[10] and mercury (II)[11-14] and other substances like sugars[15] and amino acids[16].
Lectin being a protein may contain a thiol group and disulfide, which can interact with mercury. Earlier studies made use of 18-crown-6-ether[11], 1,5-diphenyl carbazide[12], silica gel[13] and clay[14] as modifiers in the voltammetric determination of mercury. This study made use of a purified lectin isolated from black sea cucumber (Holothuria scabra) as modifier of CPE in the voltammetric detection of mercury.
MATERIALS AND METHODS
Apparatus and reagents: All voltammetric measurements were performed in a Metrohm 693 VA processor which is connected via the RS 232 interface to the computer which have a 693 VA back-up software compatible with the Metrohm output data. The data obtained was exported in the Microcal Origin Version 5. A platinum auxiliary electrode and Ag/AgCl reference were used. All potentials were measured against the Ag/AgCl electrode.
The standard solution containing 1000 mg L-1 of mercury (II) was obtained from J.T. Baker. Working solutions of mercury were prepared just before use by dilution with deionized distilled water obtained from the use of a Barnstead E-Pure Deionizer. All chemicals were analytical grade reagents.
Preparation of lectin modified electrode: Lectin was isolated by affinity chromatography wherein Holothuria scabra extracts were passed through an agarose column containing fetuin. The collected solution was lyophilized and then mixed with carbon powder and Nujol oil.
A 100 mg graphite (Sigma) powder and freeze-dried H. scabra lectin were mixed with approximately 20 μL of Nujol oil to form a paste. A portion of the paste was packed into the end of a hard plastic tube (with a thickness of 2 mm diameter) where a copper rod was inserted to establish electrical contact.
Voltammetric analysis: The lectin modified electrode was immersed in 10 mL of mercury (II) sample solution at a preconcentration cell with constant stirring for a variable time (accumulation time) at open circuit. The electrode was then taken out of the preconcentration solution, rinsed with water and transferred to the voltammetric cell containing 0.1 M sodium hydroxide solution (Ajax Chemicals) as supporting electrolyte. Cyclic voltammetry was performed using both bare CPE and lectin-modified CPE. A potential range of -1000 to 1000 mV was applied and reversed. Voltammetric measurements (DPASV) were used for the detection of mercury (II) ions and optimization. Different heavy metal standard solutions (100 mg L-1) aside from mercury (II) ions were prepared and used in the screening of the analyte that can be readily detected by the prepared electrode. Optimization of parameters like modifier concentration, accumulation time, pH of the preconcentration solution, deposition time and deposition potential was performed on the heavy metal that was most readily detected.
Regeneration: The ability of the lectin modified CPE to be reused or regenerated was also studied. Two procedures were used namely: multiple stripping and soaking in a regenerating solution. Multiple stripping was done by preconcentrating the modified electrode at 100 mg L-1 Hg2+ solution and by successive stripping of the electrode using DPASV analysis with fresh 0.1 M NaOH as the supporting electrolyte. This was repeated several times to determine whether or not the electrode surface was stripped off of Hg2+ ions completely.
The other regeneration technique was performed by soaking the used electrode in 0.01 M Ethylene Diammine Tetraacetic Acid (EDTA) solution. The time required to regenerate the electrode in EDTA was recorded.
Figure of merits: The linearity of the fabricated electrode was determined by plotting peak current response versus various concentrations of Hg (II) prepared from stock solution using the optimized parameters. The direct relationship between concentration of Hg (II) and peak current was obtained in the range of 0.1 to 5 mg L-1.
Limit of Detection (LOD) which is the concentration at which one is 95% confident that the analyte is present in the sample, was determined by performing ten voltammetric runs using a single electrode that is regenerated after each use. LOD was calculated by obtaining the standard deviation of the ten voltammetric runs, then multiply by 3 and divide by the sensitivity or slope of standard curve.
Application: The mercury content of a laboratory waste sample was determined using the lectin-modified electrode. This result was compared with Atomic Absorption Spectrophotometric (AAS) standard method using the same sample. Spiking the samples with known amount of mercury (II) solution was also performed.
RESULTS AND DISCUSSION
The first and most important preliminary analysis done on modified electrodes is the investigation of its electrochemistry including the determination of its potential window wherein the modifier and the electrolyte is not electroactive. This is usually done using cyclic voltammetry. Broadening of the cyclic voltammogram in the modified carbon paste electrode was observed (Fig. 1). This suggest the inherent redox reaction at minimal extent of the modifier.
In the screening procedure using different heavy metals, mercury ions (Hg2+) was the most easily detected analyte by the lectin-modified electrode (Fig. 2). Optimization of different parameters for analysis of mercury ions was done using DPASV.
Amount of modifier: The dependence of the electrochemical response on the amount of lectin (modifier) used ranged from 5 to 30% (by weight) modifier (Fig. 3). The peak current was observed to increase up to 20% but decreased with increasing modifier concentration. This is a consequence of the increasing availability of the complexation sites for Hg (II) collection, which consequently gave an increase in current peak.
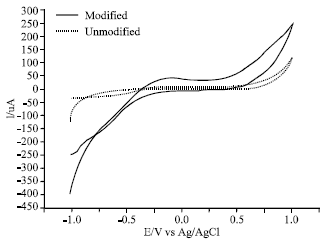 | |
| Fig. 1: | Cyclic voltammogram of unmodified and lectin modified carbon paste electrode in 0.1 M NaOH: 20% modifier, -650 mV applied potential and scan rate of 100 mV s-1 |
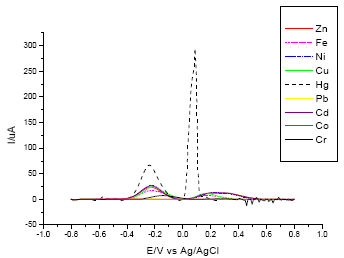 | |
| Fig. 2: | Screening for the most suitable analyte for the lectin-modified electrode: increasing applied potential from -650 mV to 0 mV at a scan rate of 40 mV s-1, pulse amplitude of 50 mV and pulse time of 30 sec |
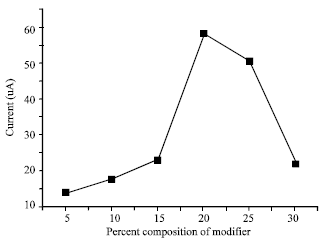 | |
| Fig. 3: | Effect of increasing amount of modifier: accumulation for 4 min in 50 mg L-1 Hg (II) solution, applied potential of -650 mV, scan rate of 40 mV s-1, pulse amplitude of 50 mV and pulse time of 30 sec |
Evidence of the decrease in current above 20% is an indication that the conductivity of the electrode is influenced by the degree at which the electrode was modified. The increase in the number of donor groups or active moieties decreased considerably the amount of carbon, which, was responsible for the conductivity of the electrode. At 20% modifier concentration, the amount of modifier is probably sufficient to attain maximum conductivity.
Effect of pH: The resulting pH profile showed that pH 7 is the optimum pH for maximum current response. However, the values at pH 5-9 are considered acceptable for generating a relatively high current response (Fig. 4).
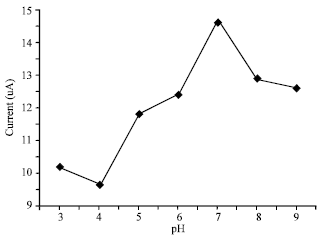 | |
| Fig. 4: | Effect of increasing pH: 20% modifier, accumulation for 4 min in 10 mg L-1 Hg (II) solution, applied potential of -650 mV, scan rate of 40 mV s-1, pulse amplitude of 50 mV and pulse time of 30 sec |
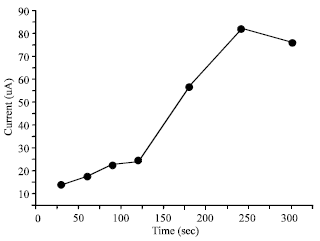 | |
| Fig. 5: | Effect of increasing accumulation time with current response: 20% modifier, accumulated in 50 mg L-1 Hg (II) solution, applied potential of -650 mV, scan rate of 40 mV s-1, pulse amplitude of 50 mV and pulse time of 30 sec |
At higher pH using phosphate buffer, dissociation of the protons in the hydroxyl group is possible, thus, making it significant for the Hg2+ free for cation exchange.
Accumulation time: In conventional stripping voltammetry, the accumulation step serves as a preconcentration step. The peak current increases gradually with increasing accumulation time (Fig. 5) demonstrating that the transport of mercury ions toward the surface of the electrode increases with time. Highest peak current was observed at 240 sec. The curve levels off after 240 sec probably due to the saturation of binding sites on the electrode surface.
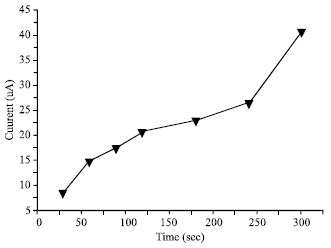 | |
| Fig. 6: | Effect of increasing deposition time with current response: 20% modifier, accumulation for 4 min in 50 mg L-1 Hg (II) solution, applied potential of -650 mV, scan rate of 40 mV s-1, pulse amplitude of 50 mV and pulse time of 30 sec |
Deposition time: Deposition time refers to the time required for the reduction of mercury (II) to its metallic state, which takes place on the electrode surface. Peak current increases as the deposition time increased to 300 sec (Fig. 6). The optimum deposition time, however, was not determined in this study since more than 300 sec of deposition time is too impractical for a rapid method like ASV. Therefore, 300 sec was used as the optimum deposition time for this study.
Regeneration: Results of multiple stripping showed that the concentration of Hg2+ decreased to a certain extent with more stripping steps. The decrease in the ion concentration reached a point where current peak height remains constant which means that regeneration by multiple stripping which means that the removal of the Hg2+ ions bound in the electrode surface was not successful.
Regeneration of the electrode by soaking the electrode in 0.01 M EDTA was achieved after 5 min of soaking (Fig. 7). In addition , a much more practical way of reusing the prepared electrode is by pushing and removing the outermost surface of the electrode and polishing the new electrode surface.
Probable mechanism: Accumulation of Hg2+ in the lectin-modified electrode occurred either by ion-exchange or by complexation mechanism between Hg2+ and H+ ion attached to polysulfhydryl group present in the modifier. After which, during deposition, Hg (II) is converted to its metallic form, Hg (0) upon application of negative potential.
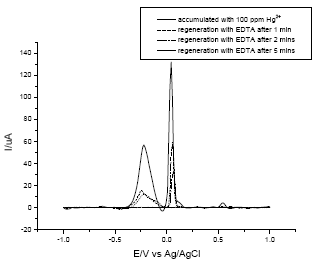 | |
| Fig. 7: | Regeneration of used electrodes soaked in 0.01 M EDTA solution |
This form of mercury would remain complexed with the sulfur groups in lectin. Lectin as a modifier served as ‘host’ site or binding site since it provided the functional group where Hg (II) could bind and it also amplify the signal generated by the Hg (0) present in electrode. Lastly, the Hg (0) was stripped in an appropriate electrolyte using the reversed potential. Regeneration of the electrode surface was successfully done using 0.01 M EDTA where in chelation between the EDTA and Hg (II) occurs.
The current generated is proportional to the concentration of mercury binding with the lectin. Current is usually generated after application of potential in which oxidation takes place. Upon oxidation, electrons are generated which results in current formation. The more mercury that is bound to the lectin, the greater the current unless saturation of binding sites occurs.
Mercury whether in ionic form or metallic form is complexed with sulfur present in the lectin. It is possible that the interaction of mercury with the polysulfhydryl group of lectin is somewhat similar to the interaction of magnesium in chlorophyll and the interaction of iron in heme where the metal is complexed with nitrogen.
Figure of merits: Using the optimized parameters, the direct relationship between concentration of Hg (II) and peak current was linear over the range of 0.1 to 5 mg L-1 with a correlation coefficient of 0.982. The limit of detection was calculated as 0.0474 mg L-1 (2.34 x 10-7 M). This detection limit is higher than the detection limit of the carbon paste electrodes modified with functionalized silica gel (0.013 mg L-1)[13], diphenylcarbazone bulk modified graphite electrode (1x10-7 M)[12] and sodium montmorillonite-modified carbon paste electrode (1x10-10 M)[17] but lower than the detection limit of diphenylcarbazone bulk modified graphite electrode using nitric acid (5 x 10-5 M)[12].
Application: The average value calculated for the mercury content using the lectin modified electrode was 0.808±0.013 mg L-1 while the calculated average amount obtained from AAS was 0.279±0.001 mg L-1, which is lower than that obtained in DPASV using the lectin modified CPE. However, using the same sample spiked with 5 mg L-1 mercury (Hg2+) ion standard solution gave an average amount of 5.75±0.06 mg L-1 using the lectin-modified electrode and 5.28±0.03 mg L-1 using AAS method. In terms of sample preparation, no sample pretreatment was done using the lectin modified electrode in comparison to AAS method.
CONCLUSIONS
CPE was fabricated using lectin isolated from the internal organs of black sea cucumber (Holothuria scabra Jaeger). The prepared electrode was found to have high affinity towards mercury. Analysis of mercury (II) was conducted by Differential Pulse Adsorptive Stripping Voltammetry (DPAdSV) using 0.1 M NaOH as supporting electrolyte. Optimization of the parameters like pH, modifier composition, accumulation time and deposition time was performed. Using the optimized parameters, the lectin-modified electrode was found to be linear in the range 0.1 to 5 mg L-1 and have an LOD of 0.0474 mg L-1. The mercury content of a laboratory waste sample was determined using the modified electrode and was found to be comparable with the one obtained using AAS, the standard method of analysis for lead.








