Research Article
Levels of Lead and Cadmium in Some Nigerian Confection Wrappers
Not Available
M.K. Morakinyo
Not Available
O.S. Agboola
Not Available
L.M. Durosinmi
Not Available
Food is a basic ingredient that makes up the diet of a living organism for proper growth and development of body tissues. Food consist of a complex mixture of water, fats, proteins and carbohydrates as well as minor constituents such as vitamins, minerals and synthetic compounds added during processing e.g. colours, antioxidants, preservatives, stabilizers, flavouring etc. Data on the composition of foods has been compiled by Nursten[1] .
Safe and nutritious food packaging is very important in our society not only to ensure that food gets to its consumer in good condition but also to conserve and preserve it so that an abundance obtained during harvest or production can be stored for use at a later date. Nowadays industrialists are using different colourful packing materials to make their products attractive to the consumers without bordering about the problem of food contamination. To ordinary people, a neatly packed food is poison free not knowing that such neatly packed food may contain some poisonous substances whose effects may not be known immediately until they are accumulated beyond tolerance level in the body.
The term confectionery has somewhat different meaning in the various parts of the world. In the United Kingdom, it is usually applied to all sweets and often includes fancy cakes, but in the trade there are three divisions namely: sugar confectionery, chocolate confectionery and flour confectionery. Sugar confectionery includes boiled sweets, toffees, fudges, fondants, jellies, pastilles and others not covered in chocolate.
Chocolates confectionery includes much of sugar confectionery covered in chocolate. However, flour confectionery includes baked fancy cakes, iced or chocolate covered and biscuits. There has been paucity of data on the fate of trace metals on confectioneries and their wrappers. Some studies that were carried out only centered on the phytic acid content of wheat flour and bread[2] and efficiency of antioxidants from natural sources in bakery products[3]. The earlier study on trace element levels in food wrappers and coloured magazine pages was carried out by Hankin et al.[4] who reported concentration of Lead of up to 30,000 ppm. Two years later, Watkins et al.[5], reported lead content of between 30,200 to 1,350 μg g-1 for bread, ice cream bar, bubble gum and raisin wrappers using Atomic Absorption Spectrometric methods. It was reported by these authors that the presence of lead was frequently associated with yellow and/or orange pigments.
The intense interest in lead and cadmium arises from their known toxicity and the possibility of human exposure to them. Lead absorbed from food and the atmosphere is retained in tissues like lungs, liver, kidney and bones. The toxic effects of lead in adult is characterised by abdominal pain, anaemia, renal disease, pheripheral neophathy with demyelination of long neurons, ataxia and memory loss[6,7] . There are indications that lead in blood as low as 10 mg dL-1 may be harmful to growing children. Excessive intakes of lead in young children may result in irreversible damage of the Central Nervous System (CNS) and cause permanent mental retardation[8-10]. In the adult the uptake of lead from the gastrointestinal tract is about 10% while in children it is probably much higher[11].
The presence of cadmium in food is dangerous to human health and adsorption of a few milligrams can result in a number of serious diseases or neoplasia. The toxic effect of chronic exposure to a low level of cadmium constitutes a continuing health concern[12,13] .
Packaging of sweet and biscuits plays two major roles-protections against breakages and physical damage and consumer appeal, which come through attractive design and colour scheme. The use of various packaging materials such as paper, aluminum foil, polyethylene, cellulose derivatives etc are well known and may contribute significantly to trace element contamination in sweet and biscuits apart from contamination which may occur during manufacturing process. This is unfortunate since the route of entry of most elements into the body is through diet apart from the elements involved directly in respiration[14] .
Considering the health implication of acute toxicity of Pb and Cd coupled with the fact that knowledge on the fate of these metals in sweet and biscuit wrappers does not exist in our country to the best of our knowledge, it is worth while to investigate the levels of these toxic elements in wrappers of commonly consumed biscuit and sweet in Nigeria. This is because school children are more predisposed to the danger of possible exposure to these metals as some of them may accidentally chew the wrappers with the sweet and biscuit. Concentration of lead as high as 5,140 and 1,400 μg g-1 has been reported for bubble gum wrapper and ice cream bar wrapper respectively[5]. This is unfortunate since these wrappers, are in constant contact with the edible part of their contents. It is a common practise in the study area that parents often package biscuits and sweets along with their Kids’ lunch when they go to school.
Materials: A market survey of the commonly consumed sweets and biscuits of various brands was carried out in supermarkets in Nigeria for analysis. A total of 40 brands of sweets and 60 brands of biscuits were discovered from the market survey. Twenty and eighteen brands of biscuits and sweets respectively were selected and purchased from the supermarkets for analysis. Our selection was based on those that are well liked by people especially children and wide availability. The selected sweet and biscuit brands that were purchased were enclosed in polythene bags, the open ends of which were tied to keep the contents away from dust and insects while being kept in a cupboard in the laboratory prior to analysis.
All reagents used-Nitric acid (BDH Chemicals Limited, England); Ammonium Nitrate (Courtin and Warner Lewis, Sussex) and Aluminum Nitrate (BDH Chemicals Limited, England) were of analytical grade.
Pre-treatment: All the glass wares used- crucibles, 25 mL volumetric flasks, sample bottles after thorough washing with hot detergents were rinsed with distilled water and soaked in 10% Nitric acid for 48 h and finally rinsed with deionised water to preclude trace metals contamination.
Methods: The method of Watkins et al.[5] was adopted for the sample preparation and analysis of Pb and Cd in both sweet and biscuit wrappers.
Wrappers were first wiped clean with a clean damp white napkin. The coloured portions were cut up into approximately 0.5x0.5 mm sections and dried at 110°C for 2 h.
About 0.5 g of samples of sweet and biscuit wrappers were accurately weighed separately into clean dried silica crucibles. To the content of the crucibles was added 1.0 mL of concentrated nitric acid each and the mixture was heated on a hot plate at about 100°C for 15 min until a partial digest was formed. Small amount of deionised water was added to the mixture intermittently to prevent the mixture from becoming dried up. Furthermore, 1 mL of 0.1 M ammonium nitrate-aluminum nitrate solution was added to the mixture and the mixture heated in an oven at 110°C for 2 h to dry the sample, followed by dry ashing in a muffle furnace at 450°C for 12 h. The residual ash was dissolved in 1 mL of hot nitric acid. This was transferred quantitatively into a 25 mL volumetric flask and the solution made to the mark with deionised water. The final solution obtained was analysed for lead and cadmium by measurement of the absorptions on the Chemtech (UK) Flame Atomic Absorption Spectrophotometer at the Center for Energy Research and Development at the Obafemi Awolowo University, Ile-Ife Nigeria, using the appropriate hollow cathode lamps. Measurements were made at 217.0 and 213.9 nm for lead and cadmium, respectively. The slit widths were 3 cm for lead and 2 cm for cadmium. The instrument was operated as per the instrument’s handbook and data were acquired on a gateway 2000 pc system using the Alphaster software.
Recovery was carried out to determine the efficiency of the method by spiking a Lemon Plus sweet wrapper sample of known amount of lead and cadmium with known amount of these elements and then redetermining these elements following the procedure described above.
The results of the quality assurance study for the spiked sweet wrapper sample based on three replicate addition experiments gave high percentage recoveries which ranged between 75±0.14 and 80±0.30% for Cd and Pb, respectively which show that the described analytical procedure for the analysis of the trace elements in the wrappers is reliable and satisfactory.
| Table 1: | Lead and cadmium content (μg g-1) of sweet wrappers |
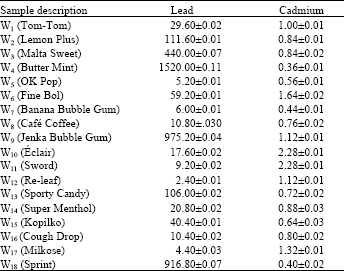 | |
| *Values are means of triplicate analysis±Standard Deviation, r = -0.3286 | |
| Table 2: | Lead and cadmium content (μg g-1) of biscuit wrappers |
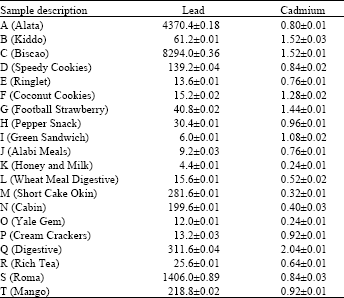 | |
| *Values are means of triplicate analysis±Standard deviation, r = 0.0024 | |
Table 1 shows the results of analysis of 18 different brands of sweet wrappers obtained from various supermarkets of the study area. Levels of lead in the sweet wrappers ranged from 1520.00±0.11 μg g-1 in Butter mint (W4) to 2.40±0.01 μg g-1 in Re-leaf (W12). The observation in this study showed that levels of lead were consistently higher than those of cadmium in all the sweet wrappers investigated and occurred most frequently in wrappers with yellow and/or orange pigment[5]. This explains the high levels of lead recorded for samples W3 (440.00±0.07 μg g-1), W4 (1520.00±0.11 μg g-1), W9 (975.20±0.04 μg g-1) and W13 (106.00±0.02 μg g-1) in Table 1, whose wrappers are either yellow and/or orange in colours. Generally, cadmium occurred at a very low levels in all the sweet wrappers investigated. The lowest level of cadmium was surprisingly found in Butter mint sweet wrapper that gave the highest level of lead. This may suggest none common source of these elements in this sweet wrapper. Result of correlation study between the two elements in all the sweet wrappers shows negative correlation (r = -0.3286). This shows that lead and cadmium are not correlated and hence cannot be from the same source.
Lead levels in biscuit wrappers ranged from 8294.0±0.36 μg g-1 in Biscao biscuit (Sample C) to 6.0±0.01 μg g-1 in Green sandwich biscuit (Sample I). Results of present analysis also showed that levels of lead are consistently higher in biscuit wrappers studied than those levels obtained for cadmium. Lead was also found at higher concentrations in wrappers with orange and/or yellow pigments (Samples A, C, D, G, H, N, Q, S and T) (Table 2) compared with others. Our observation agreed with those made by Watkins et al.[5] who reported high levels of lead for wrappers with orange and/or yellow pigments. Furthermore, cadmium levels are generally low in the biscuit wrappers studied compared with levels of lead. The lowest levels (0.24±0.01 μg g-1) being found in both samples K (Honey and milk) and O (Yale Gem) while the highest level of cadmium (2.04±0.10 μg g-1) was found in biscuit wrapper of sample Q (Digestive). An attempt was also made in case of biscuit wrappers to investigate a possible relationship between lead and cadmium by determining the correlation between cadmium and lead in the wrappers. Again, there is no significant correlation (r = 0.0024) between lead and cadmium in the biscuit wrappers suggesting none common source of these elements in the wrappers.
The variations in the levels of lead and cadmium of both biscuit and sweet wrappers obtained in this study may not be unconnected with the variation in the colours/colour mixtures of the designs made on the wrappers by different manufacturers and the chemical composition of the wrappers themselves. The use of wrappers such as paper, aluminum foil, polyethylene and cellulose derivatives are well known. Most of the results obtained for lead in both biscuit and sweet wrappers in this study are very much lower compared to those of between 30,200-1,350 μg Pb g-1 reported elsewhere for some food wrappers[4,5].
Although there are no guidelines for trace metals in food wrappers, lead and cadmium levels obtained for both sweet and biscuit wrappers in this study are quite high and may give cause for concern particularly if children accidentally chew the wrappers with either sweet or biscuit since excessive intake of lead in children may result in irreversible damage of the central nervous system and can cause permanent mental retardation[7-10]. An average of 0.09 mg kg-1 of lead and a range of 0.01-0.04 mg kg-1 of cadmium contents have been recommended for metals in total diet sample[15].
Levels of lead and cadmium in biscuit and sweet wrappers have been determined in this study. In both receptors, levels of lead are higher in wrappers with yellow and/or orange pigment. Cadmium levels are much less than those of lead in both sweet and biscuit wrappers. The levels of lead and cadmium reported in this work are quite high and should give cause for concern.