Research Article
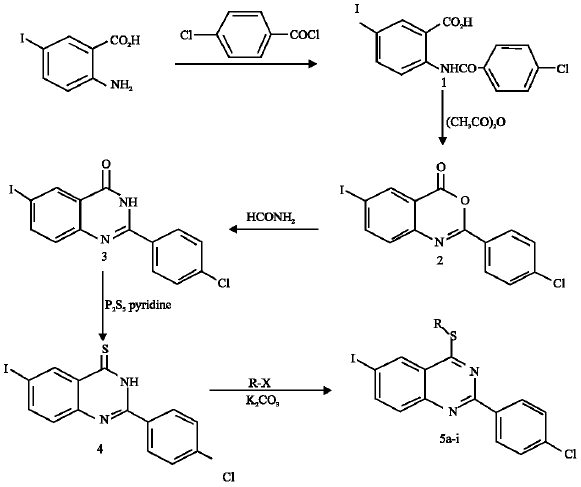
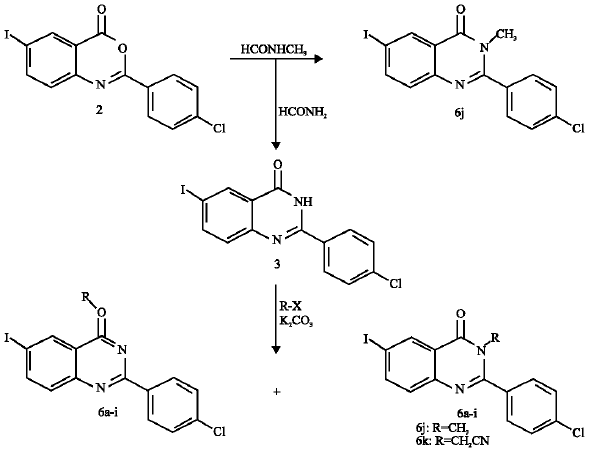
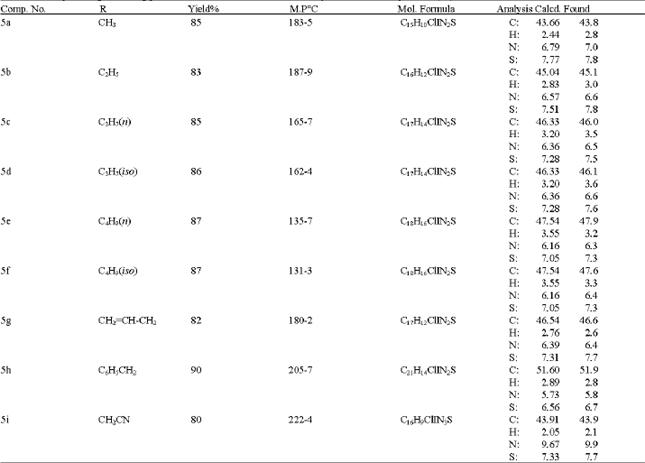
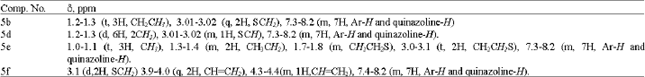
Synthesis of Certain New 6-Iodoquinazolines as Potential Antitubercular Agents
Department of Pharmaceutical Organic Chemistry, Faculty of Pharmacy, Helwan University, Egypt
K.I. Mohammed
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Al-Azhar University, Cairo, Egypt
M.A. Ahmed
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Al-Azhar University, Cairo, Egypt
S.G. Abdel-Hamide
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Al-Azhar University, Cairo, Egypt










Sunayana Gajare Reply
A very good paper