Research Article
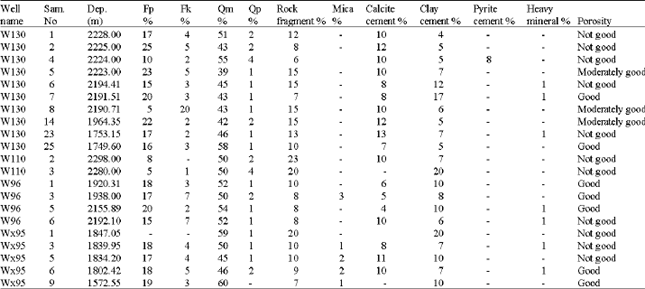
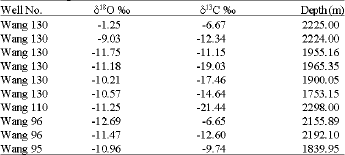
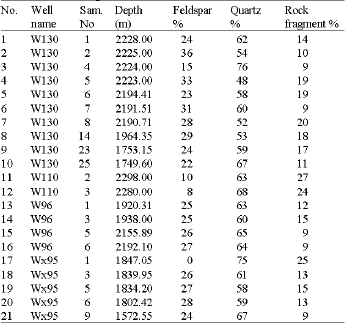
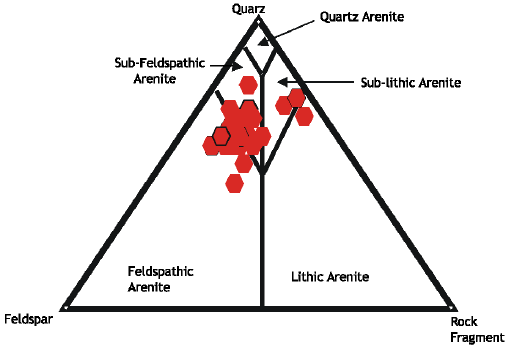
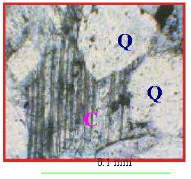
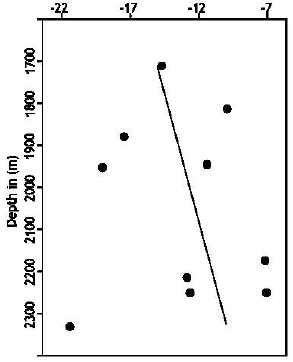
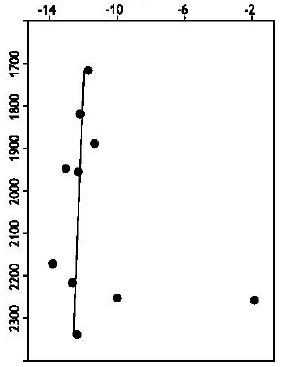
Type of Sandstone and Source of Carbonate Cement in the Kongdian Formation (Upper Part), South Slope of the Dongying Depression, East China
Faculty of Earth Sciences and Mining, Dongola University, Sudan
Fei Qi
Department of Petroleum Geology, China University of Geosciences, Wuhan 430074, China
Yang Xaing Hua
Department of Petroleum Geology, China University of Geosciences, Wuhan 430074, China