Research Article
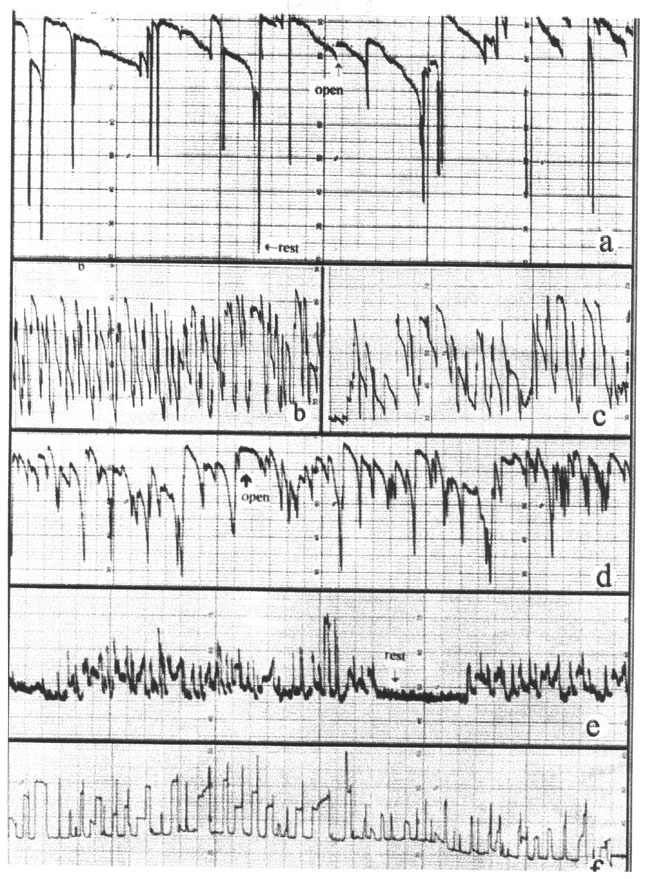
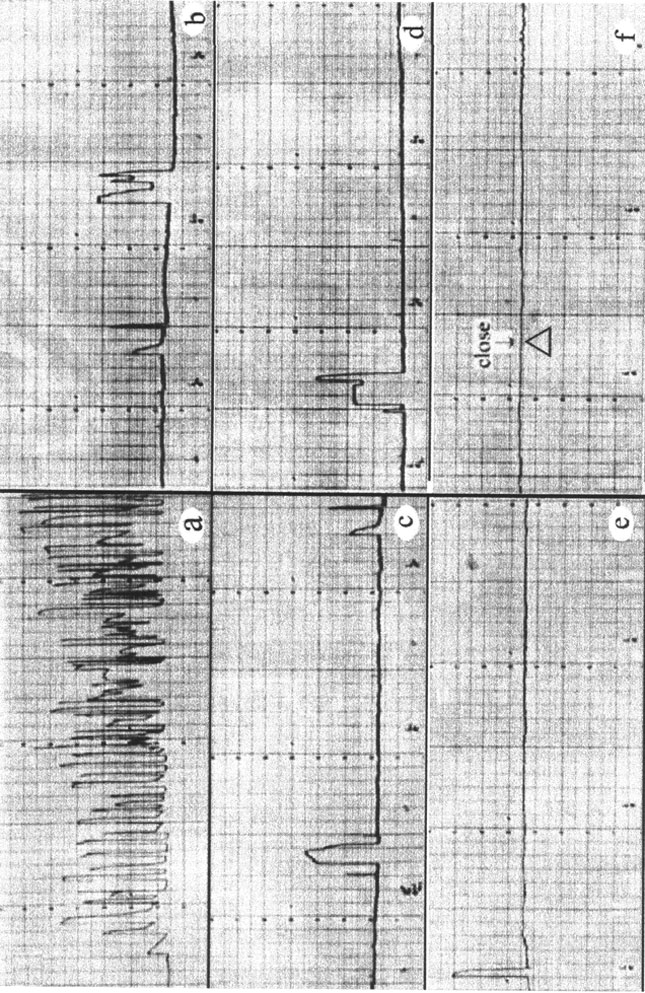
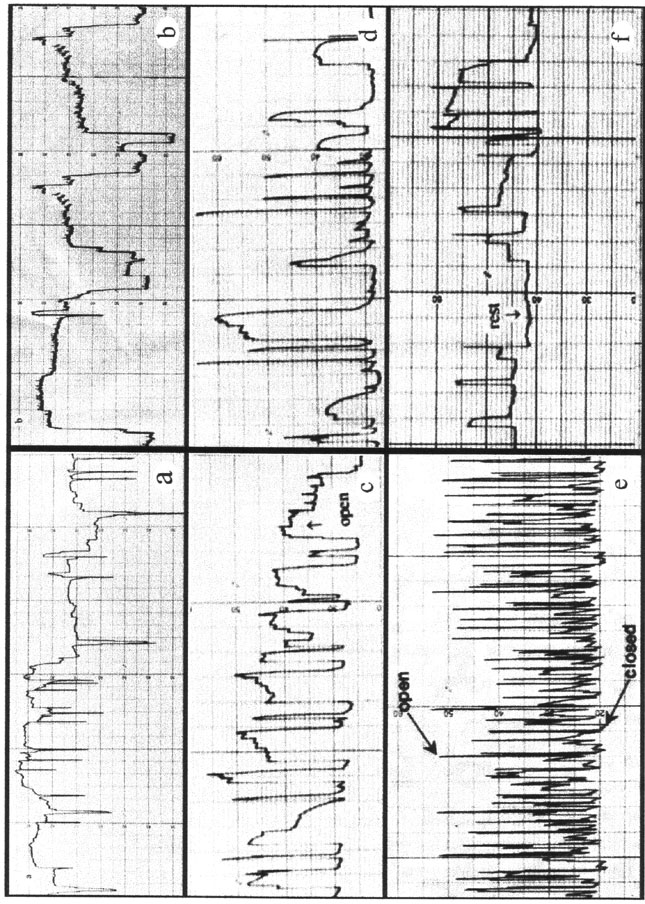
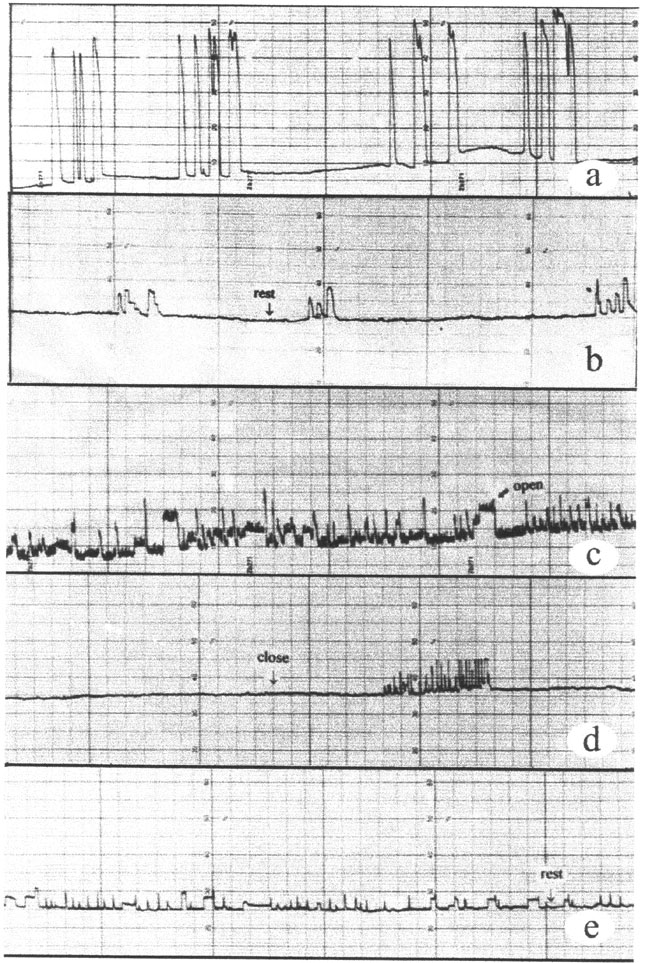
Evaluation of Lindane and Atrazine Toxicity in Mussels (Mytilus Edulis)
Department of Zoology, Faculty of Science, Suez Canal University, Egypt
Richard Greenwood
Biology Department, Portsouth University, UK
Zohour I. Nabil
Basic Medical Science, Department of N.R.C. Egypt
Ismail M. Abdel-Nabi
Department of Zoology, Faculty of Science, Suez Canal University, Egypt
Raafat A. Hanna
Department of Zoology, Faculty of Science, Suez Canal University, Egypt