ABSTRACT
Present study was aimed at investigating the toxic effects on the eleven day-old-chick embryo of different concentrations (50, 100, 200 and 400 ppm) of cypermethrin insecticide administered as a single sublethal dose (0.05 ml) into the eggs at day ‘0’ of incubation. Toxicity was evaluated in terms of teratological and biochemical changes in the developing embryo. Among biochemical constituents, activities of a few enzymes and some biochemical contents of the whole embryo were investigated. AST, ALT and LDH activities were affected with cypermethrin treatment. Of the transaminases, the activity of AST was increased whereas, the activity of ALT decreased. Activity of AST was increased at 100 ppm, while the activity of ALT was decreased at 200 and 400 ppm. LDH activity was decreased at 100 ppm. The activities of Amylase AkP and AcP remained unaltered with cypermethrin treatment. Cypermethrin treatment has resulted in the decrease of embryonic glucose, soluble protein and free amino acids contents and increase in the cholesterol and urea contents. Glucose content was decreased at 100, 200 and 400 ppm and soluble protein and free amino acid contents were decreased at 200 and 400 ppm. Cholesterol was increased at 400 ppm. Urea content was increased at 200 and 400 ppm. RNA showed a mixed response, it was increased at the dose of 50 ppm and decreased at 100 ppm. Glycogen, total protein, total lipids, uric acid and DNA contents remained unchanged with cypermethrin treatment. No malformations were observed at the dose of 50 ppm of cypermethrin treatment in eleven-day-old chick embryo However, at the doses of 100, 200 and 400 ppm teratological changes were observed, which included the reduction in crown rump length and in the size of eye ball, incomplete development of beak and totally absence of beak (at some doses), lower jaw, legs, wings and feathers. Crown rump length was reduced at the doses of 100, 200 and 400 ppm of cypermethrin. At the dose of 100 ppm, eye was not developed, however, at 200 and 400 ppm there was significant reduction in the size of eyeballs. At 100 ppm beak was incompletely developed whereas, lower jaw was totally absent. At the doses of 200 and 400 ppm beak, legs, wings and feathers were not developed. All these data suggest that cypermethrin is extremely toxic to developing organisms.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2003.432.445
URL: https://scialert.net/abstract/?doi=jas.2003.432.445
INTRODUCTION
Pyrethroid insecticides, including cypermethrin are widely used against pests all over the world to increase the production of food grains and other agricultural-products (Usmani and Knowles, 2001) and there is increased risk of food being contaminated with the insecticide, which may harm humans and the domesticated animals. Insecticides from insecticides-contaminated feed can be transported to young embryos through eggs and can cause teratological abnormalities, organ dysfunction and mortality in the young embryos thus affects the growth.
Cypermethrin, (RS)-α-cyano-3phenoxy benzyl (IRS)-cis, trans-3-(2,2-dichlorovinyl)-2, 2-dimethyl cyclopropane carboxylate, belongs to type II pyrethroid and possess α-cyano group. Its degradation products are 3-(2,3-dichlorovinyl)-2, 2-dimethyl cyclopropane carboxylic acid (cis+trans isomer) and 3-phenoxy benzoic acid. It is photostable and possesses high insecticidal activity. Cypermethrin produces drastic effects on both the invertebrates (Gowlan et al., 2002) and vertebrates (Das and Mukherjee, 2003). In vertebrates, for example in amphibians, it induces apoptosis in the telencephalon of Physalaemus biligonigerus tadpoles (Anura: Leptodactylidae) (Izaguirre et al., 2000). In fishes, cypermethrin-induced changes have been observed in various biochemical parameters like, DNA and RNA and in some enzyme activities like LDH, SDH and ATPase of muscle, liver, brain and kidney (Das and Mukherjee, 2003). In chicks, cypermethrin induces NADPH-cytochrome c reductase and cytochrome b5 (Kapoor et al., 1988).
In mammals, cypermethrin toxicity has been evaluated to a greater extent. Sheets (2000) observed that young rats are more sensitive to pyrethroids than old rats and this greater susceptibility of the neonates to cypermethrin appears to be due to limited metabolic capacity. Cypermethrin is known to affect the blood and immune system. Santoni et al. (1997) noted the cypermethrin-induced increases in peripheral natural killer cell and antibody dependent cytotoxic activity in rats. Haratym-Maj (2002) observed the increase in the number of leukocytes in peripheral blood, inhibition and mobilization of hemopoietic system in female mice following cypermethrin administration. Cypermethrin also effects the reproduction, reduces fertility in male rats through affecting testosterone, follicle-stimulating hormone and luteinizing hormone. It is also known to cause congestion and haemorrhage in testes (Elbetieha et al., 2001). Cypermethrin affects the nervous system through the release of acetyl cholinesterase from the brain synaptosomes as observed in rats and also affects the voltage-sensitive sodium channel (Eells et al., 1992). Cypermethrin is metabolised through cytochrome P450 (El-tawil and Abdel-Rahman, 2001) and is also known to induce cytochrome P450 CYP2B1 in rat hepatocyte cultures (Heder et al., 2001).
Although a lot of work has been done on the toxicity of cypermethrin on fishes and mammals, but a little work has been performed on chicks (Kapoor et al., 1988). Since a little information was available on the toxicity of cypermethrin in chick embryos, the present study was designed to investigate the toxic effects of a single dose of cypermethrin on the development of chick in terms of biochemical and teratological changes. As cypermethrin is commercially used in large quantity, the studies of its secondary affects in developing chicks are of great toxicological importance.
MATERIALS AND METHODS
Fertilized eggs were administered with different doses of cypermethrin insecticide. Dilutions were prepared in acetone. LD50 was obtained using probit analysis and was found to be 800 ppm. A single sublethal dose (0.05 ml) of the insecticide of each concentration (50, 100, 200 and 400 ppm) was administered through injection to 4 groups (6 eggs in each), respectively, into the yolk of each egg at vegetal pole by disposable tuberculin syringes at day '0' of incubation. Equal volume of acetone was injected into the controls. The eggs were incubated at 38 ± 0.5°C in incubators with a relative humidity of 70% with proper ventilation. The eggs were rotated every 2 h to avoid the sticking of the embryo to the shell membranes.
At day ‘11’ of incubation, embryos were taken out from the eggs, weighed, minced and mixed and then divided into two parts. One part was used for making saline homogenate, while the other part was used for the extraction of lipid, cholesterol and nucleic acid contents. The saline homogenate was used for the estimation of various enzyme activities and some biochemical components.
Estimation of enzyme activities
The activities of alkaline phosphatase (AkP, orthophosphoric monoester phosphohydrolase, alkaline optimum, EC: 3:1:3:1) and acid phosphatase (AcP, orthophosphoric monoester phosphohydrolase, acid optimum, EC: 3:1:3:2) were estimated according to the method of Kind and King (1954). Lactate dehydrogenase (LDH, L, lactate: NAD oxidoreductase (EC 1:1:1:27) activity was estimated by a method based on Cabaud and Wroblewski (1958). The activities of aspartate aminotransferase (ASAT; L, aspartate: 2 oxoglutarate aminotransferase, EC 2:6:1:1) and alanine amino transferase (ALAT; L, alanine: 2 oxoglutarate aminotransferase (EC 2:6:1:2) by the method of Reitman and Frankel (1957). The amylase (1, 4 a-D glucanhydrolase, EC 3:2:1:1) activity was estimated according to the procedure described in Wootton (1964).
Soluble proteins were determined from saline tissue extract, while same saline extract was digested in 0.5 N NaOH for 24 h and used for the estimation of total proteins. Both total and soluble proteins were estimated according to Lowry et al. (1951).
Glucose content was estimated by O-toluidine method of Hartel et al. (1969). Glycogen content in the supernatant left after centrifugation of saline homogenate was precipitated with ethanol and then dissolved in distilled water and estimated by the Anthrone method of Consolazio and Lacono (1963). Amino acid contents were estimated according to the Ninhydrin method of Moore and Stein (1957). Estimation of urea was performed according to the DAM method as described by Natelson et al. (1951). Uric acid content was determined according to the method described by Carraway (1963).
For the extraction of total lipid and cholesterol, the tissue was ground in hot ethanol (60°C) and kept for extraction overnight. After centrifugation at 5,000 rpm for 10 min, the supernatant was obtained and used for the estimation of total lipid by Vanillin reagent (Zollner and Kirsch, 1962) and cholesterol content according to Liebermann and Burchardt Reaction (Henry and Henry, 1974). Nucleic acids were extracted according to the method described by Shakoori and Ahmed (1973). The pellet left during lipid extraction was used for preparation of DNA and RNA extracts. DNA was extracted in hot PCA and estimated according to diphenylamine method, while RNA extract was prepared in cold PCA and estimated according to the orcinol method. Both these estimations follow the procedure as described in Schneider (1957).
Results
Enzyme Activities
Tables 1 and 2 show the effect of various concentrations (50, 100, 200 and 400 ppm) of cypermethrin administered at ‘0’ day of incubation on the activities of amylase, AkP, AcP, ALT, AST and LDH of 11th-day-old whole chick embryo. ALT, AST and LDH activities were affected with cypermethrin treatment, whereas, the activities of amylase and AkP and AcP remained unaltered. Among transaminases the activity of ALT was decreased, while the activity of AST increased. The activity of ALT showed the decrease of 35 and 46% at the doses of 200 and 400 ppm, respectively. Activity of AST was increased at 100 ppm by 94% while at the remaining doses it remained unaffected. Like AST activity, LDH activity was also affected at 100 ppm but was decreased (26%).
Biochemical components
Results are presented in the Tables 3 and 4. A marked decrease in the glucose content was observed in the embryo following cypermethrin treatment. The decrease was 56, 53 and 58% at 100, 200 and 400 ppm, respectively. In contrast, the embryonic glycogen content was not affected with this insecticide. Among proteins, total protein content remained unaltered,
| Table 1: | Toxicological effects on some Enzymes Activities of 11th day chick embryo developed from eggs treated with a single sublethal dose of Cypermethrin of various concentrations (50, 100 and 200 and 400 ppm) |
 | |
| Table. 1 shows the toxic effects of cypermethrin on amylase, AkP, AcP, ALT, AST and LDH activities of kidney of newly hatched chick. Cypermethrin of various concentrations (50, 100, 200 and 400 ppm) dissolved in acetone was administered into the eggs at day ‘0’ of incubation. Control eggs received acetone only. Embryos were taken out at the day 11 of incubation from the eggs and analysed for the enzyme activities. *, significantly different from controls at P < 0.05, using student ’t’.test. **, significantly different from controls at P < 0.01, using student ’t’.test. ***, significantly different from controls at P < 0.001, using student ’t’.test. | |
Abbreviations used
AkP, Alkaline Phosphatase; AcP, Acid Phosphatase; ALT, Alanine aminotransferase; AST. Aspartate aminotransferase; LDH, Lactate dehydrogenase; IU: International unit, the amount of enzyme which under defined assay conditions will catalyse the conversion of 1 micro mole of substrate per min.; SoU: Somogyi Unit: The amount of enzyme that catalyses digestion of 5 mg of starch under the experimental condition; KAU: King Armstrong Unit: The amount of enzyme that transforms 1 mg of phenol in 15 min.
| Table 2: | Percent change in the Enzymes Activities of 11th day chick embryo developed from eggs treated with a single sublethal dose of Cypermethrin of various concentrations (50, 100 and 200 and 400 ppm) |
 | |
| Table. 2 shows the percent change in the activities of amylase, AkP, AcP, AST and LDH in the 11th day chick embryo developed from eggs administered with (0.05 ml) of cypermethrin of various concentrations (50, 100, 200 and 400 ppm). Cypermethrin was dissolved in acetone. Control eggs received acetone only. Embryos were taken out at the day 11 of incubation from the eggs and analyzed for the enzyme activities. Only statistically significant changes were considered. | |
| Table 3: | Toxicological effects on some of the Biochemical constituents of 11th day chick embryo developed from eggs treated with a single sublethal dose of Cypermethrin of various concentrations (50, 100 and 200 and 400 ppm) |
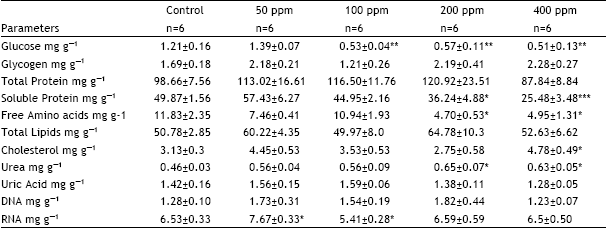 | |
| Table.3 shows the toxic effects of cypermethrin on glucose, glycogen, total proteins, soluble proteins, free amino acids, total lipids, cholesterol, urea, uric acid, DNA and RNA contents of 11th day chick embryo. Cypermethrin of various concentrations (50, 100, 200 and 400 ppm) dissolved in acetone was administered into the eggs at day ‘0’ of incubation. Control eggs received acetone only. Embryos were taken out at the day 11 of incubation from the eggs and analysed for some biochemical components. *, significantly different from controls at P < 0.05, using student ’t’.test. **, significantly different from controls at P < 0.01, using student ’t’.test. ***, significantly different from controls at P < 0.001, using student ’t’.test | |
whereas, soluble protein content was depleted at 200 and 400 ppm by 27 and 49%, respectively. However, at the doses of 50 and 100 ppm of cypermethrin soluble protein content remained unaffected. Free amino acid content showed the same pattern of change as soluble protein content did. It was decreased by 60% at 200 ppm and by 58% at 400 ppm. Among lipids cholesterol content was increased by 53% at the dose of 400 ppm, whereas, at the remaining dose levels it remained unchanged. In contrast, total lipid contents did not show change at any dose level. Of the nitrogenous wastes, urea content was changed, whereas uric content remained unaltered. Urea content was increased at 200 and 400 ppm by 41 and 37%, respectively.
| Table 4: | Percent change in on some of the Biochemical constituents of 11th day chick embryo developed from eggs treated with a single sublethal dose of Cypermethrin of various concentrations (50, 100 and 200 and 400 ppm) |
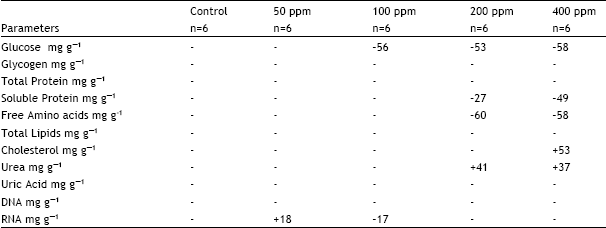 | |
| Table.4 shows the percent change in total proteins, total lipids, cholesterol and DNA contents of 11th day chick embryo developed from eggs administered with (0.05 ml) of cypermethrin of various concentrations (50, 100, 200 and 400 ppm). Cypermethrin was dissolved in acetone. Control eggs received acetone only. Embryos were taken out at the day 11 of incubation from the eggs and analysed for some biochemical components. Only statistically significant changes were considered | |
Among nucleic acids, DNA was not affected with cypermethrin treatment while RNA was changed only at the doses of 50 and 100 ppm. At 50 ppm, it was increased by 18%, whereas, at 100 ppm it showed a decrease of 17%.
Teratological changes
Control embryo
The stage in 11-day-old chick embryo correlates with the stage classified by Hamberger and Hamilton (1951). In the control embryo the crown rump length was 32.9 mm, the diameter of eyeball was 3 mm. Legs, toes and beak were well developed. The length of the beak was 2.9 mm (Fig. 1).
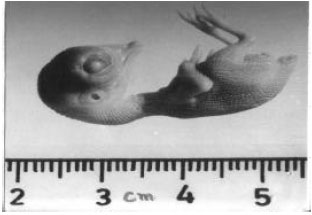 | |
| Fig. 1: | shows the morphology of 11-day-old vehicle-treated control chick embryo. Embryo is quiet normal |
 | |
| Fig. 2: | shows the morphology of 11-day-old chick embryo developed from the eggs treated with 50 ppm (0.05 ml) of cypermethrin. No malformation is seen and the embryo looks quiet normal |
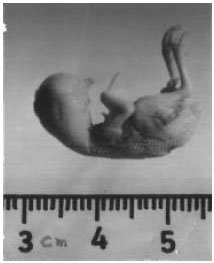 | |
| Fig. 3: | Shows the morphology of 11-day-old chick embryo developed from the eggs treated with 100 ppm (0.05 ml) of cypermethrin. Beak is incompletely developed. Eyes are totally absent. |
Treated embryos
Malformations were not detected in eleven-day-old chick embryo obtained from eggs treated with 50 ppm of cypermethrin (Fig. 2). Teratological abnormalities were observed in embryos developed from eggs treated with 100 and 200 and 400 ppm of cypermethrin (Fig. 6). Crown rump length was reduced in the embryos from the eggs treated with 100, 200 and 400 ppm of cypermethrin and this reduction was 19, 25 and 19%, respectively. At the dose of 100 ppm, eye was not developed, however, at 200 and 400 ppm there was significant reduction in the size of eyeballs. At 100 ppm beak was incompletely developed whereas, lower jaw was totally absent (Fig. 3).
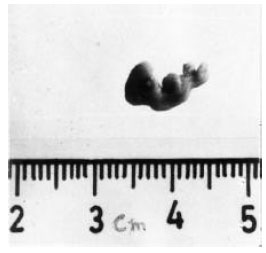 | |
| Fig. 4: | Shows the morphology of 11-day-old chick embryo developed from the eggs treated with 200 ppm (0.05 ml) of cypermethrin. Crown rump length is significantly reduced. Eyeballs are severely malformed. Beak, wings, limbs and feathers are totally absent |
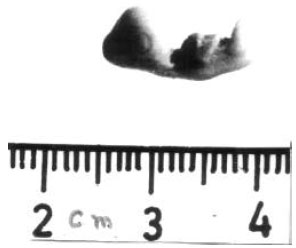 | |
| Fig. 5: | Shows the morphology of 11-day-old chick embryo developed from the eggs treated with 400 ppm (0.05 ml) of cypermethrin. Embryo is severely malformed. Crown rump length and the size of eyeball are significantly reduced. Beak is totally absent. Wings, limbs and feathers are not completely developed, however, their buds are prominent |
At higher doses of 200 and 400 ppm severe deformities were observed and these deformities included the absence of beak, legs, wings and feathers (Figs. 4 and 5).
Discussion
Biochemical components
Significant reduction in embryonic glucose content at 100, 200 and 400 ppm observed in the present study could be due to the utilization of glucose for detoxification of the cypermethrin or its metabolite through the process of glucuronidation (a process by which toxic metabolites combine with glucose phosphate and are excreted).
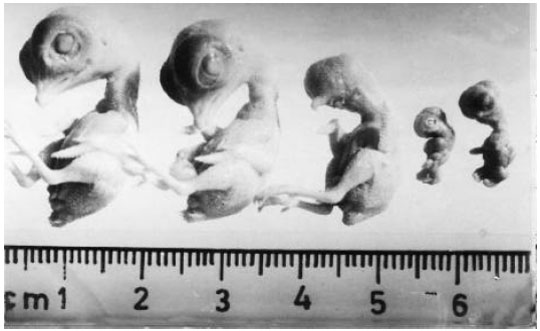 | |
| Fig. 6: | Is a composite photograph of vehicle treated control embryo and the embryos developed from eggs treated with different concentrations of cypermethrin. Control embryo is on the left side and is followed by 50, 100, 200 and 400 ppm of cypermethrin treated embryos. 200 ppm and 400 ppm of cypermethrin treatment has resulted in severely deformed embryos (right) |
Lock and Berry (1981) observed increased blood glucose and lactate in rats following cypermethrin treatment. Increased blood glucose level was also observed in Indian major carp, Labeo rohita following cypermethrin treatment (Das and Mukherjee, 2003). The increased blood glucose reflects the decreased tissue glucose as observed in the present study. Soluble protein was decreased in the present study. Decrease in soluble protein might have occurred as the result of decrease in the level of free amino acid contents, which are the precursors in the protein synthesis. The decrease in free amino acid content of the whole embryo was also observed in the present study. Decrease in the free amino acid content may indicate the utilization of the amino acids either for the oxidation purpose to provide energy or used for the synthesis of protein to repair the injured tissues. Since the embryonic urea content the end product of amino acid breakdown was significantly elevated in the present study, it becomes clear that the amino acids were utilized for the oxidation purpose to provide energy in cypermethrin-induced stress conditions rather than used in the repair of injured tissues through utilization in the protein synthesis. Reddy and Yellamma (1991b) had already observed such increase in urea content in the liver of the fish Tilapia mossabica following cypermethrin administration. Cholesterol content showed a significant increase at the dose of 400 ppm. Since cholesterol is the integral part of cell membrane, its synthesis might have been increased to repair the damaged tissues as the result of cypermethrin toxicity or it might have not been utilized in the synthesis of some steroid hormones, which utilize cholesterol as precursor. RNA content was increased at 50 ppm and decreased at 100 ppm. Sheela and Muniandi (1992) observed decrease in RNA in muscle and liver of fish following cypermethrin treatment. Increase in RNA content may indicate an adaptation to over come the toxic insult through increased protein synthesis. In contrast, decline in the embryonic RNA content may indicate damage to the embryonic tissue caused by cypermethrin. However, the reason for the unchanged RNA content at 200 and 400 ppm dose levels is not clear.
Enzyme activities
From amongst enzymes, only the activities of AST, ALT and LDH were affected whereas, amylase, AkP and AcP remained unaltered with cypermethrin treatment. El-Demerdash et al. (2003) observed decreased ALT in liver, brain and testes of rabbits following administration of cypermethrin. Reddy and Yellamma (1991b) found that cypermethrin causes increase in ALT and AST in liver of the fish Tilapia mossabica. Findings of these authors (Reddy and Yellamma, 1991b; El-Demerdash et al., 2003) indicate that the effect of cypermethrin on the induction and inhibition of the enzymes ALT, AST and LDH is animal specific. For example, Reddy and Yellamma, (1991b) observed an increase in ALT in the liver of fish Tilapia mossabica, while, El-Demerdash et al. (2003) observed decrease in ALT in liver of rabbits. In addition, effects of cypermethrin are also found to be organ specific. For example observations made by Das and Mukherjee (2003) that cypermethrin increases LDH activity in brain and liver but inhibits in kidney of the same animal (Indian major carp, Labeo rohita) also indicate the organ specificity of the cypermethrin. Since the present investigations were made in the whole embryo rather than in organs of the embryo, a mixed response was observed, one enzyme was elevated and other inhibited. However decrease in both the enzymes ALT and AST in tissues indicate damage as the enzymes are leaked out when the cells in the tissues lyse. El-tawil and Abdel-Rahman (2001) observed the decrease in cell viability along with the leakage of ALT and AST enzymes from rat hepatocytes into the culture medium as a result of cypermethrin toxicity. Decrease in the embryonic LDH activity also indicates the damage to the embryo. Decrease in LDH activity was also observed in the fish with cypermethrin treatment (Reddy and Yellamma, 1991a).
Teratological changes
High doses (100, 200 and 400 ppm) of cypermethrin caused severe teratological abnormalities in the chick embryo. These abnormalities included the reduction in crown lump length, size of eyeball, absence of, beak, legs, wings and feathers. These findings are in agreement with the work reported by Clemedson et al. (1989) who noted reduction in crown rump length in the chick embryo following treatment with CCl4 and the work reported by Schom et al. (1979) who found that Azordin treated chick embryos responded by developing a generalized micromelia, parrot beak syndromes and a chondroplasia. The development of beak, legs, wings and feathers all involve bone ossification. Similar changes were observed in the chick embryo induced with diazinon an OP insecticide (Misawa et al., 1981). Abou-Egla et al. (1988) observed skeletal abnormalities in 11-day-old chick embryo developed from the eggs treated with methomyl insecticide.
The observed drastic effects of cypermethrin on the development of beak, legs, wings and feathers might be attributed to the ability of the insecticide to react with cation ionophores and thereby modulate their motion across lipid membranes. Rashatwar and Matsumura (1985) observed that cypermethrin inhibits calcium binding protein calmodulin. Binding of cypermethrin with this protein might have resulted in the decreased influx of Ca2+ across the cell membrane that influenced the bone development as observed in the present study. Findings of Imamura et al. (2000) also show that cypermethrin decreases the Ca2+ influx into neurons as revealed by the decrease in the expression of brain-derived neurotrophic factor (BDNF) and c-fos genes which are activated through Ca2+ influx into neurons, in an activity-dependent manner. Walker (1971) observed abnormal beak development in chick embryos treated with 30 μmoles of maloxon. He also observed inhibition of plasma cholinesterase enzyme with maloxon. Eells et al. (1992) have already shown the release of acetyl cholinesterase from rat brain synaptosomes following cypermethrin administration. Recently, Das and Mukherjee (2003) observed the decreased acetyl cholinesterase activity in the brain of carp Labeo rohita treated with cypermethrin. So the deformities observed in the muscular, nervous and skeletal system of cypermethrin treated chick embryos seem to be due to the interference of cypermethrin with neurotransmitter acetyl cholinesterase activity as well as with the influx of Ca2+ across the cell membrane.
Reduction in the size of head observed in the present study reflects the reduction in the size of brain that occurs as a result of degenerative changes in neurons. One other possibility for degenerative changes in neurons come from the findings of Izaguirre et al. (2000) who reported cypermethrin-induced apoptosis in the telencephalon of Physalaemus biligonigerus tadpoles (Anura: Leptodactylidae). This cypermethrin-induced apoptosis might have resulted in the degenerative changes in brain thus reduced the size of brain as observed in the present study. Apoptosis, a programmed cell death is mediated by reactive oxygen species (ROS) and lipid hydroperoxide radicals (Sarafian and Bredesen, 1994) and these free radicals and ROS have been found to be generated through increased cerebral lipid peroxidation in rat brain following cypermethrin intoxication (Giray et al., 2001). In addition, El-Demerdash et al. (2003) also reported free radicals in plasma, liver, brain and testes of rabbits following cypermethrin administration. ROS, free radicals and reactive metabolites are also generated during the metabolism of toxins via mixed oxidase system including cytochrome P450. El-tawil and Abdel-Rahman (2001) found the involvement of cytochrome P450 in cypermethrin metabolism. Induction of Cytochrome P450 CYP2B1 with cypermethrin was also observed in rat hepatocyte cultures (Heder et al., 2001). In chicks, NADPH-cytochrome c reductase and cytochrome b5 is also induced with cypermethrin (Kapoor et al., 1988). These ROS, free radicals and reactive metabolite are either detoxified through conjugating with glutathione or react with tissue macromolecules to initiate the damage. Glutathione conjugation occurs by glutathione S-transferase (GST), a detoxification enzyme, which is also known to be induced with cypermethrin in shore crab (Gowlan et al., 2002). Toxic effects of cypermethrin on chick embryo at higher doses and no effect at lower doses indicate that cypermethrin is metabolised to its toxic metabolites, which at lower doses can be detoxified by conjugating with glutathione but at higher doses are unable to be detoxified as the glutathione level in the tissue drops. Thus damage caused by ROS, free radicals and reactive metabolite can also lead to the development of teratological abnormalities in the growing embryos. Teratological changes can also occur through the alterations in genes. This genotoxic effect of cypermethrin has been observed in the bone marrow cells in a murine test system in vivo through the induction of sister chromatid exchanges (Giri et al., 2003). All these effects, 1) the cypermethrin-induced biochemical changes, 2) its interference with Ca2+ influx, 3) induction of apoptosis and 4) its genotoxic effects contribute towards the changes, like, teratological abnormalities in the developing chick embryos. It is quite clear that treatment of eggs with insecticide will give abnormal embryos, which may be attributed to the dysfunction caused by the insecticide to the living cells involved in the growth of embryos and their skeleton formation. In the light of these observations it is recommended that cypermethrin insecticide should be used with caution as it can be hazardous to both humans and domestic animals.
REFERENCES
- Abou-Egla, M.H., F.A. Shaheen and M.M. Kady, 1988. Skeletal anomalies and liver malformations induced in chick embryos by some pesticides. J. Agric. Res. Tanta Univ., 14: 1369-1375.
Direct Link - Cabaud, P.G. and F. Wroblewski, 1958. Colorimetric measurement of lactic dehydrogenase activity of body fluids. Am. J. Clin. Pathol., 30: 234-236.
PubMed - Clemedson, C., B. Schmid and E. Walum, 1989. Effects of carbon tetrachloride on embryonic development studied in the post-implantation rat embryo culture system and in chick embryos in ovo. Toxicol. In Vitro, 3: 271-275.
CrossRefPubMedDirect Link - Das, B.K. and S.C. Mukherjee, 2003. Toxicity of cypermethrin in Labeo rohita fingerlings: Biochemical, enzymatic and haematological consequences. Comp. Biochem. Physiol. Part C: Toxicol. Pharmacol., 134: 109-121.
CrossRefPubMedDirect Link - Eells, J.T., P.A. Bandettini, P.A. Holman and J.M. Propp, 1992. Pyrethroid insecticide-induced alteration in mammalian synaptic membrane potential. J. Pharmacol. Exp. Ther., 262: 1173-1181.
Direct Link - Elbetieha, A., S.I. Daas, W. Khamas and H. Darmani, 2001. Evaluation of the toxic potential of cypermethrin pesticide on some reproductive and fertility parameters in the male rats. Arch. Environ. Contam. Toxicol., 41: 522-528.
CrossRefDirect Link - El-Demerdash, F.M., M.I. Yousef and K.S. Al-Salhen, 2003. Protective effects of isoflavone on some biochemical parameters affected by cypermethrin in male rabbits. J. Environ. Sci. Health Part B: Pestic. Food Contam. Agric. Wastes, 38: 365-378.
CrossRefPubMedDirect Link - El-Tawil, O.S. and M.S. Abdel-Rahman, 2001. The role of enzyme induction and inhibition on cypermethrin hepatotoxicity. Pharmacol. Res., 44: 33-40.
CrossRefPubMedDirect Link - Giray, B., A. Gurbay and F. Hincal, 2001. Cypermethrin-induced oxidative stress in rat brain and liver is prevented by vitamin E or allopurinol. Toxicol. Lett., 118: 139-146.
CrossRefPubMedDirect Link - Giri, S., A. Giri, G.D. Sharma and S.B. Parsed, 2003. Induction of sister chromatid exchanges by cypermethrin and carbosulfan in bone marrow cells of mice in vivo. Mutagenesis, 18: 53-58.
PubMedDirect Link - Gowland, B.T.G., C.F. Moffat, R.M. Stagg, D.F. Houlihan and I.M. Davies, 2002. Cypermethrin induces glutathione S-transferase activity in the shore crab, Carcinus maenas. Mar. Environ. Res., 54: 169-177.
CrossRefDirect Link - Hamburger, V. and H.L. Hamilton, 1951. A series of normal stages in the development of chick embryo. J. Morphol., 88: 49-92.
CrossRefPubMedDirect Link - Haratym-Maj, A., 2002. Hematological alternations after pyrethroids poisoning in mice. Ann. Agric. Environ. Med., 9: 199-206.
PubMedDirect Link - Heder, A.F., K.I. Hirsch-Ernst, D. Bauer, G.F. Kahland and H. Desel, 2001. Induction of cytochrome P450 2B1 by pyrethroids in primary rat hepatocyte cultures. Biochem. Pharmacol., 62: 71-79.
CrossRefPubMedDirect Link - Imamura, L., H. Hasegawa, K. Kurashina, A. Hamanishi, A. Tabuchi and M. Tsuda, 2000. Repression of activity-dependent c-fos and brain-derived neurotrophic factor mRNA expression by pyrethroid insecticides accompanying a decrease in Ca2+ influx into neurons. J. Pharmacol. Exp. Ther., 295: 1175- 1182.
Direct Link - Izaguirre, M.F., R.C. Lajmanovich, P.M. Peltzer, A.P. Solar and V.H. Casco, 2000. Cypermethrin-induced apoptosis in the telencephalon of Physalaemus biligonigerus tadpoles (Anura: Leptodactylidae). Bull. Environ. Contam. Toxicol., 65: 501-507.
CrossRefDirect Link - Kapoor, R.K., S.S. Chauhan, N. Sing and U.K. Misra, 1988. Induction of hepatic mixed-function oxidases by permethrin and cypermethrin in chicks fed vitamin A-deficient diet. Pestic. Biochem. Physiol., 32: 205-211.
CrossRefDirect Link - Kind, P.R.N. and E.J. King, 1954. Estimation of plasma phosphatase by determination of hydrolysed phenol with amino-antipyrine. J. Clin. Pathol., 7: 322-326.
CrossRefPubMedDirect Link - Lowry, O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall, 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem., 193: 265-275.
CrossRefPubMedDirect Link - Misawa, M., J. Doul, P.A. Kitose and E.M. Uyeky, 1981. Teratogenic effects of cholinergic insecticides in chick embryos I. Diazinon treatment on acetylcholinesterase and choline acetyltransferase activities. Toxicol. Applied Pharmacol., 57: 20-29.
CrossRefDirect Link - Moore, S. and W.H. Stein, 1954. A modified ninhydrin reagent for the photometric determination of amino acids and related compounds. J. Biol. Chem., 211: 907-913.
CrossRefDirect Link - Natelson, S., M.L. Scott and C. Beffa, 1951. A rapid method for the estimation of urea in biologic fluids. Am. J. Chem. Pathol., 21: 275-281.
PubMedDirect Link - Rashatwar, S.S. and F. Matsumura, 1985. Interaction of DDT and Pyrethroids with calmodulin and its significance in the expression of enzyme activities of phosphodiesterase. Biochem. Pharmacol., 34: 1689-1694.
Direct Link - Reddy, A.T. and K. Yellamma, 1991. Perturbations in carbohydrate metabolism during cypermethrin toxicity in fish, Tilapia mossambica. Biochem. Int., 23: 633-638.
PubMedDirect Link - Reddy, A.T.V. and K. Yellamma, 1991. Cypermethrin induced changes in nitrogen metabolism of fish, Tilapia mossambica. Biochem. Int., 23: 649-654.
Direct Link - Reitman, S. and S. Frankel, 1957. A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am. J. Clin. Pathol., 28: 56-63.
CrossRefPubMedDirect Link - Santoni, G., F. Cantalamessa, L. Mazzucca, S. Romagnoli and M. Piccoli, 1997. Prenatal exposure to cypermethrin modulates rat NK cell cytotoxic functions. Toxicology, 120: 231-242.
CrossRefDirect Link - Sarafian, T.A. and D.E. Bredesen, 1994. Invited commentary is apoptosis mediated by reactive oxygen species? Free Radical Res., 21: 1-8.
CrossRefDirect Link - Schom, C.B., U.K. Abbott and N.E. Walker, 1979. Adult and embryo responses to organophosphate pesticide: Azodrin. Poult. Sci., 58: 60-66.
Direct Link - Sheets, L.P., 2000. A consideration of age-dependent differences in susceptibility to organophosphorus and pyrethroid insecticides. Neurotoxicology, 21: 57-63.
PubMedDirect Link - Usmani, K.A and C.O. Knowles, 2001. Toxicity of pyrethroids and effect of synergists to larval and adult Helicoverpa zea, Spodoptera frugiperda and Agrotis ipsilon (Lepidoptera: Noctuidae). J. Econ. Entomol., 94: 868-873.
CrossRefPubMedDirect Link - Walker, N.E., 1971. The effect of malathion and malaoxon on esterases and gross development of the chick embryo. Toxicol. Applied Pharmacol., 19: 590-601.
CrossRefPubMedDirect Link - Schneider, W.C., 1957. Determination of Nucleic Acids in Tissues by Pentose Analysis. In: Methods in Enzymology, Colowick, S.P. and N.O. Kaplan (Eds.). Vol. 3, Academic Press, New York, USA., pp: 680-684.
Direct Link








