Research Article
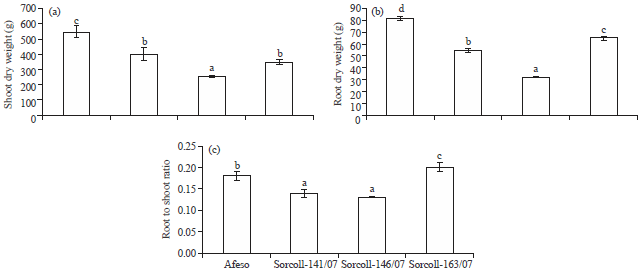
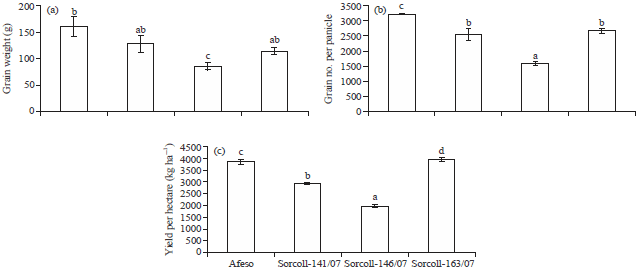
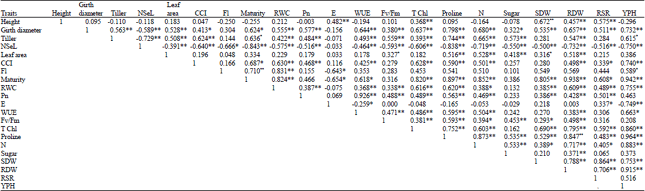
Growth, Water Status, Physiological, Biochemical and Yield Response of Stay Green Sorghum (Sorghum bicolor (L.) Moench) Varieties-A Field Trial Under Drought-Prone Area in Amhara Regional State, Ethiopia
Department of Biology, College of Natural and Computational Sciences, University of Gondar, P.O. Box 196, Gondar, Ethiopia
Azamal Husen
Department of Biology, College of Natural and Computational Sciences, University of Gondar, P.O. Box 196, Gondar, Ethiopia
Masresha Fetene
Department of Plant Biology and Biodiversity Management, College of Natural Sciences, Addis Ababa University, P.O. Box 3434, Addis Ababa, Ethiopia
Gietahun Yemata
Department of Plant Biology and Biodiversity Management, College of Natural Sciences, Addis Ababa University, P.O. Box 3434, Addis Ababa, Ethiopia