ABSTRACT
Sorption capacity of synthetically prepared amorphous Al and Fe hydroxides relative to heavy metal (Pb) was greatly affected by the presence of low (oxalate and citrate) or high (tannate) molecular weight organic ligands during their formation. Co-precipitation of tannate ligand with Al or Fe markedly enhanced the capacity of the precipitation products to sorb Pb. Regarding to oxalate and citrate ligands, their presence suppress the capacity of Al precipitates and enhanced that of Fe ones to sorb Pb. Although the capacity of all Al and Fe precipitates to sorb Pb significantly decreased as pH decreased from 7.0 to 5.5, Fe-Tan and Fe-Cit keep relatively high capacity. Lead sorption consists of fast initial reaction completed in few hours, followed by slow reaction conducted to the end of the experiment. The mols of H+ released per ion Pb sorbed (HR/Sorb.Pb) varied as the adsorbent and sorption density varied and ranged from 1:1 to higher than 2:1. Also, the ratios (HR/Sorb.Pb) increased with the reaction period and ranged from 0.3 to 2.2:1. These different stoichiometries and the delaying of H+ release, refer to formation of different complexes between Pb and adsorbents; an intermediate one {X-O(OH)-Pb2+} with no H+ release may be formed at the beginning; followed by formation of mono-dentate (X-OPb+ ) and bi-dentate (X-O2Pb) ones. Only small portions of Pb previously sorbed on Al and Fe precipitates at pH 7.0 (0 to 16 and 0 to 7.4%, respectively) are reversible. The reversibility increased as sorption density increased and pH decreased. As residence time increased, sorbed Pb, in particular at pH 7.0, was nearly irreversible over a desorption period of 24 h, which indicate the transformation of adsorbed Pb. Pb sorption/desorption characteristics on amorphous Al and Fe precipitates are highly dependent on surface properties, Point of Zero Salt Effect (PZSE) and remaining charge at the same pH (σp). Fe-Tan, Fe-Cit and Al-Tan had high ability to sorb and sequester sorbed Pb over a wide range of pH, which indicate that, these modified hydroxides could be produced and utilize for pollution prevention, remediation of the soil environment and decontamination of heavy metal polluted water.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/ja.2006.440.450
URL: https://scialert.net/abstract/?doi=ja.2006.440.450
INTRODUCTION
It is generally accepted that trace metal concentrations in soil solution are primarily controlled by sorption and desorption reactions at the particle solution interface (Glover et al., 2002). There has been a great deal of research examining trace metal adsorption reaction on soil and soil constituents (Muller and Sigg, 1992; Ainsworth et al., 1994; Gunneriusson et al., 1994). Trace metal adsorption on oxides of Fe and Al consists of two steps, an initial fast reaction followed by a slow reaction conducted to long period (Strawn et al., 1998; Eick et al., 1999; Strawn and Sparks, 2000). Adsorption of metals from solution is accompanied by the release of protons, typically between one and two protons released per cation adsorbed, the exact value depending on the adsorbing metal and the solution conditions (Kinniburgh and Jackson, 1982). Iron oxides have been shown to contain different surface functional groups with different affinities for trace elements (Benjamin and Leckie, 1981; Hiemstra et al., 1996). Fendorf et al. (1997), suggested that trace elements form different surface complexes on goethite depending on sorption density. Davis and Leckie (1978) found that, more than one stoichiometric surface reaction was required to describe the adsorption of metal on amorphous iron oxyhydroxide, which suggests that more than one species is being adsorbed, or more than one type of surface site is involved in the adsorption process, or both. Lead adsorption on goethite has been modeled successfully as both mono-dentate and bi-dentate surface complexes (Hayes 1986; Gunneriusson et al., 1994). Very recently, Xu et al. (2006), using EXAFS observed that Pb sorption on hydrous Fe oxide forms mononuclear bi-dentate surface complex. Unlike adsorption reactions, there are relatively little research dealt with desorption behavior of metals from soil and soil constituents. It is often observed that desorption reactions are slower than sorption ones (Smith and Comans, 1996; Scheidgger et al., 1996), which could be attributed to; 1) transformation of adsorbed ion from one state to another, for example: conversion from an adsorbed species to a surface precipitate; and 2) intra-particle diffusion of adsorbed ion along with micropore walls of oxide (Fan et al., 2004; Xu et al., 2006). In addition, desorption reactions often require large activation energies, which cause the reaction to proceed slowly (McBride, 1994).
Iron oxides have been recognized as playing a very important role in controlling the fate of heavy metals in soils and sediments and in the regulation of Pb in natural waters (Manceau et al., 1992). Hydrous amorphous Al and Fe oxides are ubiquitous in the surface as both discrete particles and coatings and exhibit a high affinity for heavy metal contamination. They have large surface areas and an abundance of binding sites; therefore, they have significant impact on contaminant mobility (Jenne, 1968; Fan et al., 2004). The high affinity of Fe oxides for certain trace elements, Pb in particular, makes them a likely long-term sink for cationic polyvalent metals (Sauve et al. 2000). Most Fe oxides form under the influence of pedogenic environments including the presence of organic ligands (Cornell and Schwertmann, 1979), which perturb the crystallization of mineral, i.e., modifies the crystalline structure of the resultant hydrolytic products (Krishnamurti and Huang, 1990, 1991, 1993). Short range ordered (noncrystalline) precipitation products of Al play a significant role in chemical adsorption (Hsu, 1977; Huang and Violante, 1986).
It is generally postulated that, the hazardous of heavy metal pollutants in the environment is primarily controlled by sorption/desorption reactions at solid phase/soil solution interface. Hence, assessment the capability of soil constituents to adsorb and sequester adsorbed pollutant is important to predict potential toxicity and mobility of contaminants. The objective of this research was to study sorption/desorption characteristics of heavy metal (Pb) on amorphous Al and Fe hydroxides synthetically prepared in absence and presence of low (oxalate and citrate) and high (tannate) molecular weight organic ligands. Kinetics of Pb sorption and its desorption were, also, studied. Surface properties of the prepared Al and Fe hydroxide, Point of Zero Salt Effect (PZSE) and remaining charge (σp) at the same pH and Cation Exchange Capacity (CEC) were used for interpretation of sorption/desorption characteristics. Pb/modified amorphous Al and Fe hydroxides system was chosen because, Pb is a common soil pollutant and the predominant Al and Fe oxides phases in soils are often the amorphous species, which form under the influence of pedogenic environments including the presence of organic ligands (Cornell and Schwertmann, 1979).
MATERIALS AND METHODS
Materials: Amorphous Al(III) and Fe(III) Hydroxides prepared, as described in the first part of this research (Helal, 2006), in absence or presence of low (oxalate and citrate) or high (tannate) molecular weight organic ligands (LMWL and HMWL), at ligand/(Al or Fe) Molar Ratio (MR) of 0.1, were used for the present study. The forms of Al precipitation products used included; nonmodified (Al-OH) and those modified using oxalate (Al-Oxal), citrate (Al-Cit) or tannate ligands (Al-Tan). Similar forms of Fe precipitates (Fe-OH, Fe-Oxal, Fe-Cit and Fe-Tan) were used. Surface properties, Point of Zero Salt Effect (PZSE) and Cation Exchange Capacity (CEC), of these precipitates as determined by Helal (2006), are given in Table 1. Al and Fe precipitates formed in absence of organic ligands (Al-OH and Fe-OH) had a remaining positive charge (σp) at PZSE. The remaining positive charge increased for the precipitates formed in the presence of oxalate ligand (Al-Oxal and Fe-Oxal). The remaining charge of the precipitates, Al-Cit, Fe-Cit, Al-Tan and Fe-Tan, formed in the presence of citrate or tannate ligands, inversed to be negative.
Methods:
Sorption and desorption of Pb: To 100 mg of Al or Fe precipitation products, 100 mL of 50 mM NaClO4 solution containing 0 to 0.8 mM Pb in the form of Pb(NO3)3, was added. The pH of NaClO4 and Pb(NO3)3 solutions were adjusted to 5.5 or 7.0 prior addition to the adsorbent using 0.1 N HClO4 or 0.103 N NaOH. The suspensions are then readjusted after addition, then shaked gently for 24 h at 23±0.5°C in a thermostatically controlled room. pH of the suspensions are periodically readjusted to pH 5.5 or 7.0 throughout the reaction period. The final suspension is centrifuged at 10.000 rpm for 30 minutes. Pb concentration is determined in the clear filtrate using an ICP-OES (GBC, Integra XL, Australia). The quantity of Pb sorbed was calculated as the difference between the determined initial and final concentration of Pb in the equilibrium solution. Number of mols of protons released during the reaction period was calculated using the volume of NaOH consumed in adjusting the pH of the equilibrium solution. To all suspensions, a few drops of toluene were initially added to prevent microbial activity. All experiments were done in two replicates.
A solution of 0.01M Ca(NO3)2 is used for the desorption of the previously sorbed Pb, at adsorbent: solution ratio equal to that used for sorption experiment. The suspension was shaked gently for reaction period of 24 h at 23±0.5°C. After centrifugation, Pb concentration is determined in the clear solution.
Kinetics of Pb sorption and its desorption: The kinetic experiment was conducted at pH 7.0 and 5.5 for two precipitates (Al-Tan and Fe-Tan). To 100 mg of Al-Tan or Fe-Tan, 100 mL of 50 mM NaClO4 containing 0.8 mM Pb in the form of Pb(NO3)3 was added. Both NaClO4 and Pb(NO3)3 solutions are adjusted to pH 7.0 or 5.5 prior addition to the adsorbent, then readjusted after addition. The suspension are gently shaked for different periods ranged from ¼ to 168 h at 23±0.5°C. The earlier procedure was adopted.
The earlier sorbed Pb was desorbed using 0.01M Ca(NO3)2 as the abovementioned.
Statistical analysis: Data of Pb sorption on Al and Fe precipitation products and its subsequent desorption was analyzed using linear model procedure of SAS program (SAS, 1996). The models consists of three factors including; 1) two types of metal hydroxides (Al and Fe), 2) four forms of Al precipitation products (Al-OH, Al-Oxal, Al-Cit and Al-Tan) as well as Fe ones, and 3) two pH values of 5.5 and 7.0. That is besides all possible interactions between the tested factors.
RESULTS AND DISCUSSION
Pb Sorption on Al and Fe precipitation products: The results showed that, at concentrations, up to 0.4 mmole Pb L-1, all added Pb was completely removed from the equilibrium solution. Co-precipitation of high molecular weight ligand, HMWL, (tannate) with Al considerably increased Pb sorption capacity of the precipitation products (Fig. 1a). The quantities of Pb sorbed on Al-Tan were much higher than those of nonmodified Al-OH. On the other hand, co-precipitation of low molecular weight ligands, LMWL (citrate or oxalate) with Al significantly (p = 0.001) decreases Pb sorption capacity. Sorption isotherms show that, although, Al-Cit and Al-Oxal are approaching maximum sorption capacity, Al-Tan and, to a lesser degree, Al-OH had high potential ability to sorb additional quantities of soluble Pb. Figure (1b) shows the high sorption capacity of Fe precipitates, in particular those modified with tannate and citrate ligands, to sorb Pb.
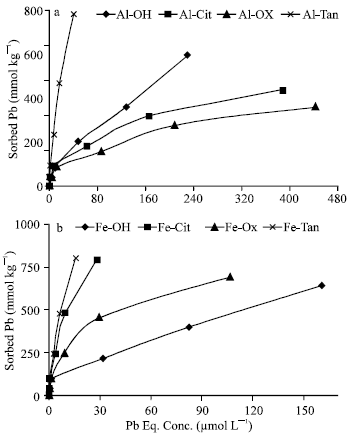 | |
| Fig. 1: | Sorption isotherms of Pb on Al (a) and Fe (b) precipitates at pH 7.0 |
The results showed that, dissimilar to Al precipitates, co-precipitation of LMWL (citrate and oxalate) with Fe enhanced the capacity of the precipitation products to sorb Pb. The quantities of Pb sorbed on Fe-Oxal, Fe-Cit and Fe-Tan (694, 791 and 796 mmol kg-1, respectively) are higher than (108, 123 and 124%, respectively) that (641 mmol kg-1) sorbed on nonmodified Fe precipitate (Fe-OH). Both Fe-Tan and Fe-Cit had high potential ability to sorb additional quantities of soluble Pb. The quantity of Pb sorbed on Fe-Cit is almost equal to that sorbed on Fe-Tan, which refers to differences in the behavior of citrate among Al and Fe precipitates. In general, the capacities of Fe precipitates to sorb Pb are higher than their correspondings of Al ones. Sorption isotherms of Pb on Fe-Tan, Al-Tan and Fe-Cit, are typical H-type isotherm which indicates the high affinity of Pb to the adsorbents and at the same time refers to that, the mechanism involves is complex formation. Eick et al. (1999), also, reported the high affinity of Pb to goethite. Amorphous Al and Fe hydroxides had considerably higher capacity to sorb Pb comparing with reactive components (hydroxyaluminum-and hydroxyaluminosilicate-montmorillonite complexes) synthetically prepared by Saha et al. (2002) which showed the capacity to sorb, approximately, 20 mmol Pb kg-1, at pH 7.0.
| Table 1: | Point of Zero Salt Effect (PZSE) and Cation Exchange Capacity (CEC, cmolc kg-1) of Al and Fe Precipitates formed at Ligand/Metal MR of 0.1 (Helal, 2006) |
 | |
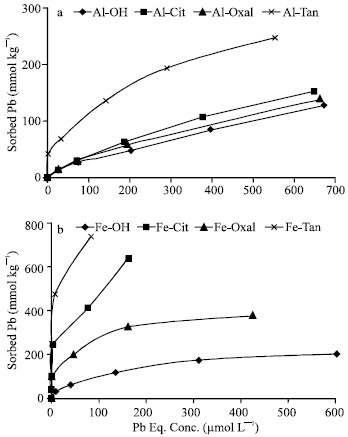 | |
| Fig. 2: | Sorption isotherms of Pb on Al (a) and Fe (b) precipitates at pH 5.5 |
Also, the quantity of Pb sorbed on Fe-Tan, Al-Tan and Fe-Cit exceeded those sorbed by binary and ternary model particles containing soil humic acid and Wyoming montmorillonite (approximately 700 and 200 mmol kg-1, respectively) prepared by Cruz-Guzman et al. (2003). These results proved that, Fe and Al precipitates, in particular those modified by tannate, are more efficient than other reactive components in removing soluble heavy metal pollutants. Thus they could be produced and utilize for pollution prevention and for remediation of the soil environment.
Effect of pH on Pb sorption: In general, the results show significant decreases (p = 0.001) in Pb sorbed on Al and Fe precipitates at pH 5.5, comparing with their correspondings at pH 7.0. The quantities of Pb (128 to 247 mmol kg-1) sorbed on Al precipitates at pH 5.5 (Fig. 2a) represented 22 to 38% of those sorbed on their correspondings at pH 7.0. The highest reduction (78%) in Pb sorption capacity is observed for nonmodified Al precipitate (Al-OH). However, relatively low reductions (62 to 68%) are recorded for Al-Oxal, Al-Cit and Al-Tan. All Al precipitates had limited potential ability to sorb additional quantities of Pb at pH 5.5. Their isotherms approached maximum sorption capacity. Fig. (2b) shows that, the quantities of Pb sorbed on Fe precipitates at pH 5.5 varied widely and ranged from 202 to 739 mmol kg-1. These quantities are generally lower than (representing 32 to 93% of) those sorbed at pH 7.0. Fe-Tan and Fe-Cit had the highest capacity to sorb Pb (739 and 639 mmol kg-1, respectively) at pH 5.5, whereas Fe-OH and Fe-Oxal had the lowest ones (202 and 369 mmol kg-1, respectively). That is besides, Fe-Tan and Fe-Cit had high potential ability to sorb additional quantities of Pb at pH 5.5. These results proved that, co-precipitation of tannate and citrate ligands with Fe markedly enhanced the capacity of the precipitates to sorb Pb even at low pH values, comparing with Fe-OH and Fe-Oxal. In comparison with Al precipitates, Fe ones had higher capacity (158 to 420%) to sorb Pb at pH 5.5. The highest orders of magnitude of increment are recorded for Fe-Tan, Fe-Cit, whereas the lowest ones are recorded for Fe-OH and Fe-Oxal. In general, the results showed that, Fe precipitates, particularly those formed in the presence of tannate and citrate, had high capacity to sorb and to remove soluble Pb from solution, at wide range of pH.
Surface properties and Pb sorption: Pb sorption on Al and Fe precipitates could be discussed with surface properties, PZSE values and the remaining charge at the same pH (σp) and Cation Exchange Capacity (CEC). In general, the results showed the ability of Al-Oxal, Al-OH, Al-Cit, Fe-OH and Fe-Oxal to sorb Pb at pH (5.5) lower than or equal to their PZSE values (7.1, 5.65, 5.45, 6.4 and 6.1, respectively, Table 1). These findings indicated that, specific adsorption is a mechanism involved in Pb sorption process. However, at pH values higher than PZSE, Pb sorption capacity of Al and Fe precipitates markedly enhanced, which could be attributed to electrostatic forces between the surface of negative charge and metal cation of Pb. An inverse relationship was observed between Pb sorbed at pH 5.5 and 7.0 and PZSE (Fig. 3). The results showed that, as PZSE values of Al-Tan, Al-Cit and Al-Oxal, increased from 4.85, 5.45 to 7.1, respectively, quantities of Pb sorbed, at pH 7.0 and 5.5 significantly (r = 0.937, p = 0.05, pH 7.0) decreased from 781, 432 to 359 and from 247, 152 to 138 mmol kg-1, respectively.
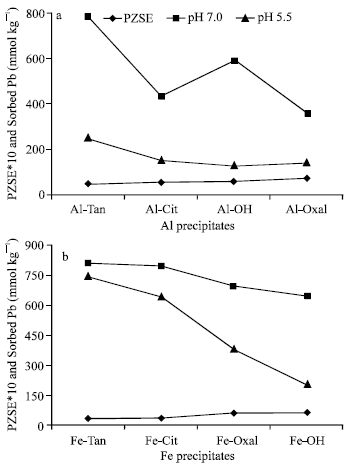 | |
| Fig. 3: | Point of Zero Salt Effect (PZSE) and Pb sorption on Al (a) and Fe (b) precipitates at pH 7.0 and 5.5 |
(Al-OH showed a little deviation.) Highly significant correlations (r = 0.975 and 0.976, p = 0.01) are obtained between PZSE values and Pb sorbed on Fe precipitates at pH 7.0 and 5.5, respectively (Fig. 3b). As PZSE values of Fe precipitates (Fe-Tan, Fe-Cit, Fe-Oxal and Fe-OH) increased from 3.2, 3.7, 6.1 to 6.4, respectively, the quantities of Pb sorbed at pH 7.0 and 5.5, markedly decreased from 796, 791, 694, to 641 mmol kg-1 and from 739, 639, 377 to 202 mmol kg-1, respectively. Shifting PZSE to higher pH values indicated the decrease in the acidity of surface OH and COOH groups (Mustafa et al., 2004), which lead to decreases in metal sorption capacity of the surface.
Also, the quantities of Pb sorbed on Al and Fe precipitates (Fig. 2) could be discussed with the remaining charge (σp). The data showed that, the precipitates had positive σp, had lower Pb sorption capacity than those had negative σp. The quantities of Pb sorbed at pH 5.5 on Al-Cit and Al-Tan which had negative σp, are significantly (p = 0.001) higher than those sorbed on Al-OH and Al-Oxal, which had positive σp. Also, the quantities of Pb sorbed on Al-Tan at pH 7.0 was much higher than those sorbed on Al-OH and Al-Oxal. (Little deviation is shown for Al-Cit). This trend was rather clear with Fe precipitates, the quantities of Pb sorbed on Fe-Cit and Fe-Tan, at pH 5.5 and 7.0, which had negative σp, were significantly (p = 0.01) higher than those sorbed on Fe-OH and Fe-Oxal which had positive σp.
Regarding to Cation Exchange Capacity (CEC) of Al and Fe precipitates, the results showed that, all precipitates had high capacity to sorb Pb in quantities much higher than their CEC values. The quantities of Pb sorbed on Al precipitates (Al-OH, Al-Oxal, Al-Cit and Al-Tan, Fig. 1a) at pH 7.0, are 1.5 to 3 times higher than CEC values (Table 1). Also, the quantities of Pb sorbed on Fe precipitates (Fe-OH, Fe-Oxal, Fe-Cit and Fe-Tan, Fig. 1b) at pH 7.0, are, roughly, two times higher than CEC values. These results proved that, specific adsorption rather than nonspecific one is responsible for sorption of Pb on Al and Fe precipitates at pH 7.0. At pH 5.5, quantities of Pb sorbed on Al precipitates markedly decreased. The quantities of Pb sorbed on all Al precipitates, are much lower than CEC values (Table 1). Unlike Al precipitates, Fe ones had the capacity to sorb quantities of Pb, at pH 5.5, much higher than CEC values. Except for Fe-OH, the quantities of Pb sorbed on Fe precipitates are 1.2 to 2.2 times higher than CEC values. However, the quantity (202 mmol kg-1) of Pb sorbed on Fe-OH is much lower than CEC value of 312 mmol kg-1. These results suggested that, specific adsorption is, also, involved in the removal of Pb from solution at pH 5.5, particularly for Fe precipitates.
Release of H+ against Pb sorption: The quantities of H+ released are generally higher than sorbed Pb and increased as sorbed Pb increased (Fig. 4a). The mols of H+ released per Pb sorbed (HR/Sorb.Pb) varied widely as both quantity of sorbed Pb and form of Al precipitates varied. Generally, HR/Sorb.Pb ratios are higher than 1:1. At the lowest sorbed quantity, the ratio calculated for Al-OH was approximately 6:1. As sorbed Pb increased, the ratio decreased gradually to be 1:1, in particular at the highest quantities of Pb sorbed, which could be attributed to the formation of mono-dentate surface complex. In agreement with Mckenzie (1980); Honeyman and Leckie (1986), the wide ratio (6:1) could be attributed to the hydrolysis of Pb (in solution or at the surface) during the adsorption reaction, particularly at low surface coverage, exchange reactions at the surface, or a combination of these. However, the ratios obtained for Al-Cit and Al-Tan were little higher than 1:1 (ranging from 1.1 to 1.5:1). i.e., approximately one proton released per one Pb ion sorbed to Al-Cit and Al-Tan, which refers to the formation of mono-dentate surface complex.
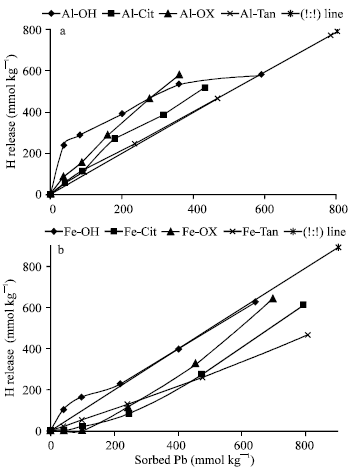 | |
| Fig. 4: | Mols of H+ released against Pb sorption on Al (a) and Fe (b) precipitates at pH 7.0 |
The additional 0.1 to 0.5 mols of H+ released may result from a change in the adsorbent surface charge attributable to the specifically adsorbed Pb (resulting in the repulsion of protons into the bulk solution), the formation of mixed surface complexes, or a more complex adsorption mechanism (Eick et al., 1999). For Al-Oxal, the ratios are approximately 2:1 (ranging from 2.2 to 1.6:1), which could be attributed to the formation of bi-dentate surface complex, or mixture of mono-dentate and bi-dentate complexes. Generally, these results indicated that, more than one mechanism are involved in Pb adsorption on Al precipitates. Fendorf et al. (1997) suggested that trace elements form different surface complexes on goethite depending on sorption density.
Figure (4b) shows the quantities of H+ released from Fe precipitates against Pb sorbed. The results showed that, the quantity of H+ released from Fe-Tan is much lower than those released from Fe-OH, Fe-Cit and Fe-Oxal, in despite of Fe-Tan had the highest Pb sorption capacity. The mols of H+ released per Pb sorbed (HR/Sorb.Pb), varied widely among Fe precipitates. Generally, the ratios (HR/Sorb.Pb) obtained for Fe precipitates are much lower than their correspondings of Al precipitates. The ratios obtained for Fe-OH decreased from 2.5 to 1:1, as sorption density increased. Macroscopic studies examining Pb sorption on goethite have measured proton stoichiometries ranging from 1 to 2 (Gunneruiusson et al., 1994). The release stochiometries were ascribed to the formation of mono-dentate surface complexes, bi-dentate surface complexes and the hydrolysis of Pb during adsorption. Unlike Fe-OH, the results showed the delaying of H+ release from other Fe precipitates. The ratios obtained for Fe-Oxal and Fe-Cit are much lower than 1:1, particularly at low sorbed Pb. They increased from 0:1 to 0.9:1 as Pb sorption increased. On the other hand, the ratios recorded for Fe-Tan are constant at 0.5:1, i.e., the ratios are independent on sorption density. The ratios lower than 1:1 verify that Pb is initially adsorbed to the external sites forming outer sphere surface complex {X-O(OH)-Pb2+}with no H+ released. The narrow ratios obtained for Fe-OH, comparing with wide ones reported for Al-OH proved that, metal hydrolysis at Al precipitate surfaces, not in solution, is responsible for the wide ratios (6:1) obtained at low sorption density. Various ratios recorded for mols of H+ released against Pb sorption verified that, many processes are involved in binding Pb with Al and Fe precipitates. These processes varied as co-precipitated ligand and sorption density varied. Pb binding has been found to change with increasing sorption density (Templeton et al., 2003).
Pb desorption: The results of Fig. 5 showed that, desorption of Pb increased as the previously sorbed increased. The total quantities of Pb desorbed from Al precipitates (pH 7.0) during desorption period of 24 h, varied from 0 to 66 mmol kg-1 (Fig. 5a). The desorbed quantities showed that, only small percentages (0 to 16%) of the previously sorbed Pb {(Des.Pb/Sorb.Pb)*100} are reversible. The highest reversibility are recorded for Al-Oxal and Al-OH, whereas the lowest ones are recorded for Al-Tan and Al-Cit. Figure (5b) shows the quantities (0 to 57 mmol kg-1) of Pb desorbed from Al precipitates at pH 5.5. These quantities represented 0 to 41% of the previously sorbed Pb. Thus the reversibility of Pb sorbed at pH 5.5 is much higher than that at pH 7.0. Al-Tan had the highest ability to retain sorbed Pb at pH 5.5, whereas the other precipitates had low ability in this connection. Desorption of Pb could be discussed with surface characteristics. As PZSE of Al-Oxal, Al-Cit and Al-Tan decreased from 7.1, 5.45 to 4.85, respectively, the percentages of Pb desorbed from previously sorbed at pH 7.0, decreased from 16, 7.3 to 3.3%, respectively. (A little deviation is observed for Pb desorbed from Al-OH). Also, as PZSE values of Al-Oxal, Al-OH and Al-Tan decreased, percentages of Pb desorbed from previously sorbed at pH 5.5 decreased from 41, 36 to 3.5%, respectively. (A little deviation is observed for Al-Cit).
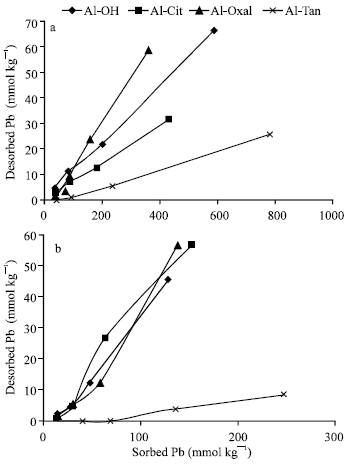 | |
| Fig. 5: | Pb desorption as a function of the previously sorbed on Al precipitates at pH 7.0 (a) and 5.5 (b) |
Also, the ability of Al precipitates to retain sorbed Pb had a good harmony with the remaining charge (σp). The precipitates of Al-Oxal and Al-OH which had positive σp, had lower ability than those (Al-Cit and Al-Tan) had negative ones to retain sorbed Pb at pH 7.0 and 5.5.
Quantities of Pb desorbed from previously sorbed on Fe precipitates (Fig. 6a), at pH 7.0, increased as sorption density increased and ranged from 0 to 47.5 mmol kg-1. These results showed that, only small portions of Pb sorbed on Fe precipitates are reversible (0 to 7.4%). In agreement with these results, Glover et al. (2002) reported that, Pb removal from goethite surface was not completely reversible during an 8 h desorption period for all of the experiments. The highest Pb reversibility are recorded for Fe-OH, whereas, the other three precipitates (Fe-Tan, Fe-Oxal and Fe-Cit) had relatively low ones. However, the quantities (0 to 68 mml/kg) of Pb desorbed from previously sorbed on Fe precipitates at pH 5.5 (Fig. 6b) are higher than those at pH 7.0 and represented 0 to 15% of the previously sorbed. Among all Fe precipitates, Fe-OH had the highest percentages of desorbed Pb, whereas the other three forms (Al-Tan, Al-Oxal and Al-Cit) had lower ones.
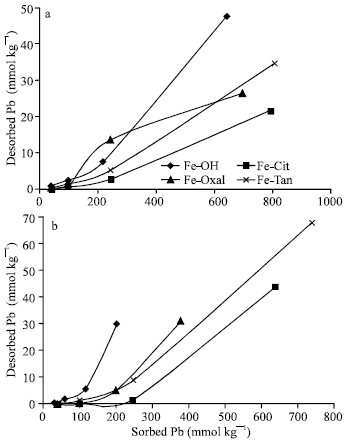 | |
| Fig. 6: | Pb desorption as a function of the previously sorbed on Fe precipitates at pH 7.0 (a) and 5.5 (b) |
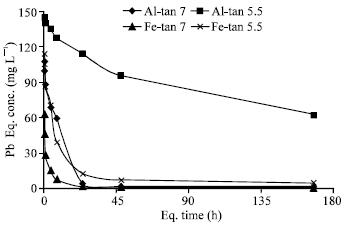 | |
| Fig. 7: | Pb concentration in solutions equilibrated with Al-Tan and Fe-Tan at pH 7.0 and 5.5, as a function of time |
In general, the reversibility of Pb sorbed on Fe precipitates at pH 7.0 and 5.5 are significantly (p = 0.002) lower than those of Al precipitates. These findings indicated that, Fe precipitates had higher ability than Al ones to retain sorbed Pb over a wide range of pH. Among all Al and Fe precipitates, Fe-Tan and Fe-Cit, as well as Al-Tan, had the highest capacity to sorb and to sequester sorbed Pb over a wide range of pH.
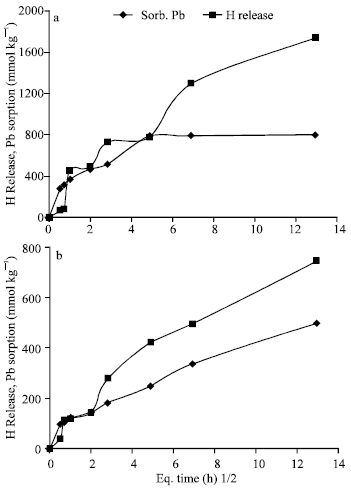 | |
| Fig. 8: | Pattern of H+ released with time against Pb sorption on Al-Tan at pH 7.0 (a) and 5.5 (b) |
Thereby, Fe-Tan, Fe-Cit and Al-Tan could be considered as a sink of Pb in soil and they could be produced and utilized for stabilization of heavy metal (Pb) in polluted soil. The other forms of Al and Fe precipitates are effective only at higher pH values of 7.0.
Desorption of Pb from Fe precipitates, could be discussed with surface properties. As PZSE of Fe precipitates decreased from 6.4, 6.1 to 3.7 for Fe-OH, Fe-Oxal and Fe-Cit, respectively, percentages of desorbed Pb (pH 7.0) decreased from 7.4, 3.8 to 2.7%, respectively. Also, as PZSE of Fe-OH, Fe-Oxal, Fe-Cit and Fe-Tan decreased from 6.4, 6.1, 3.7 to 3.2, respectively, percentages of desorbed Pb (pH 5.5) decreased from 15, 8.2, 7.0 to 4.9%, respectively. Furthermore, Pb desorption are in a good harmony with the remaining charge (σp) of Fe precipitates. The precipitates of Al-Ox and Al-OH, which had positive σp, had lower ability than those (Al-Cit and Al-Tan) had negative σp, to retain Pb sorbed at pH 7.0 and 5.5. The irreversibility of sorbed Pb could be attributed to transformation of sorbed Pb, formation of inner sphere complex with surface functional groups of co-precipitated ligands. In despite of the statement of Ainsworth et al. (1994) that, inter-particle diffusion is not expected due to the difference in ionic radii between Pb and central cations of Fe or Al, Xu et al. (2006) stated that, slow intra-particle diffusion of adsorbed Pb resulting in a surface diffusivity of 2.5X10-15 Cm2 S-1 could be a reason for the irreversibility of adsorbed Pb. However, formation of surface precipitates is not expected due to the short reaction period.
Kinetics of Pb sorption and its desorption: Figure (7) shows two steps of changes of Pb concentrations in solution equilibrated with Al-Tan and Fe-Tan at pH 7.0 and 5.5, with time. The first step shows sharp decrease in Pb concentration completed in approximately 4 h. The second step shows slow decrease in Pb concentration conducted to the end of the reaction period (168 h). In agreement with the statements of Strawn et al. (1998); Eick et al. (1999); Glover et al. (2002), these results verify that, two different mechanisms are involved in Pb sorption. The initial fast one is exchange reaction in which Pb is attracted to the negative charge sites and/or formation of outer-sphere complex. However, the second one (slow reaction mechanism) is not as well understood and has been attributed to intra-particle diffusion (Xu et al., 2006), formation of surface precipitate and/or adsorption processes. The patterns of H+ released from Al-Tan and Fe-Tan against Pb sorption are shown in Fig. (8 and 9). The results showed a delaying of H+ release at the beginning of the reaction. As the reaction period increased, the quantity of H+ released increased gradually and exceeded Pb sorption. The mols of H+ released from Al-Tan per Pb sorbed (HR/Sorb.Pb) at pH 7.0 and 5.5 increased with time and ranged from 0.3 to 2.2:1 and 0.4 to 1.5:1, respectively. The corresponding ratios obtained for Fe-Tan, also, increased from 0.4 to 1.9:1 and 0.4 to 1.1:1, respectively, as the reaction period increased. The gradual increases in HR/Sorb.Pb ratios could be attributed to that, the initial sorbed Pb form intermediate complex {Al-O(H)-Pb2+} with no release of H+ (Hachiya et al., 1979; Liu and Huang, 2001). As residence time increased the sorbed ions form mono-dentate (Al-O-Pb+), then bi-dentate (Al-O2-Pb) complexes, with release of one or two protons per ion Pb sorbed. The gradual increases in HR/Sorb.Pb ratios with time, is a good explanation for the low ratio (0.5:1) obtained for sorption of Pb on Fe-Tan at a reaction period of 24 h (Fig. 4b). Based on the obtained results, the mechanism of Pb sorption on Al-Tan and Fe-Tan including; exchange reaction and/or formation of outer sphere surface complex at the beginning of the reaction, followed by specific adsorption through replacement of one proton to form mono-dentate, then the adsorbed Pb replaces one more proton to form bi-dentate complex.
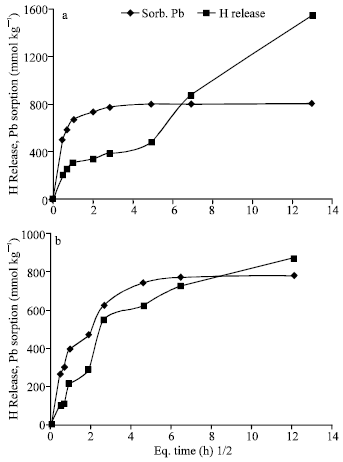 | |
| Fig. 9: | Pattern of H+ released with time against Pb sorption on Fe-Tan at pH 7.0 (a) and 5.5 (b) |
The quantities of Pb desorbed from previously sorbed on Al-Tan and Fe-Tan varied as the residence time, sorption density and pH, varied (Fig. 10). Two different trends are observed for the changes of Pb desorption with residence time. The first one showed gradual increases in the desorbed quantities as the residence time increased to 4 and 8 h for Al-Tan and Fe-Tan, respectively. The desorbed quantities represented 10 and 5% of Pb earlier sorbed on Al-Tan and Fe-Tan (pH 7.0), respectively. The second one, which observed for longer residence periods showed gradual decreases in the quantity of Pb desorbed, in which the reversible portion decreased with time to be 1.1 and 1.7%, respectively. Regarding to the desorption of Pb earlier sorbed at pH 5.5, at shorter residence period, 20 and 5% of Pb earlier sorbed on Al-Tan and Fe-Tan, respectively, are reversible. At longer residence period, the reversibility decreased to be 3.3 and 5%, respectively. These results proved that, as residence time increased, Pb sorbed on Al-Tan and Fe-Tan, are nearly irreversible, in particular at pH 7.0. In agreement with these results, McLaren et al. (1998) Eick et al. (1999) Glover et al. (2002) reported that, Pb desorption kinetics was slow compared with sorption reaction and not completely reversible during 6-8 h desorption experiment.
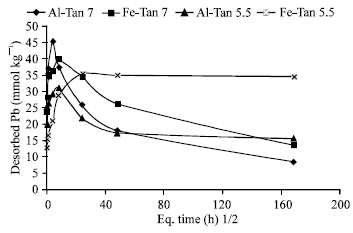 | |
| Fig. 10: | Pb desorption as a function of residence time |
Also, Eick et al. (1999), stated that, desorption rate coefficients and the quantity of Pb desorbed were greater for the short-term experiment. However, irreversibility of Pb with ageing could be attributed to the transformation of the adsorbed Pb, formation of inner-sphere complex, formation of surface precipitates and/or intra-particle diffusion.
In conclusion amorphous Al and Fe hydroxides synthetically prepared in the presence of low organic ligand, in particular oxalate, act as efficient sorbents for anions (Helal, 2006), whereas those formed in the presence of high molecular weight ligand (tannate) act as efficient sorbent for metal cation. Therefore, these precipitates play a vital role in the transformations of nutrients and pollutants in soils and associated environments. Moreover, these precipitates could be produced and utilize for pollution prevention, chemi-immobilization of cationic and anionic pollutants in soils and decontamination of polluted water.
REFERENCES
- Ainsworth, C.C., J.L. Pilon, P.L. Gassman and W.G. Van Der Sluys, 1994. Cobalt cadmium and lead sorption to hydrous iron oxide residence time effect. Soil Sci. Soc. Am. J., 58: 1615-1623.
Direct Link - Cruz-Guzman, M., R. Celis, M.C. Hermosin, P. Leone, M. Negre and J. Cornejo, 2003. Sorption-Desorption of lead (II) and Mercury (II) by model association of soil colloids. Soil Sci. Soc. Am. J., 67: 1378-1387.
Direct Link - Eick, M.J., J.D. Peak, P.V. Brady and J.D. Pesek, 1999. Kinetics of lead adsorption/desorption on goethite: Residence time effect. Soil Sci., 164: 28-39.
Direct Link - Fan, M., T. Boonfueng, Y. Xu, L. Axe and T.A. Tyson, 2005. Modeling Pb sorption to microporous amorphous oxides as discrete particles and coatings. J. Colloid Interface Sci., 281: 39-48.
CrossRefDirect Link - Glover, L.G., M.J. Eick and B.V. Drady, 2002. Desorption kinetics of cadmium2+ and Lead2+ from goethite: Influence of time and organic acids. Soil Sci. Soc. Am. J., 66: 797-804.
Direct Link - Helal, M.I.D., 2006. Surface properties of amorphous al and fe hydroxides formed in the presence of oxalate, citrate and tannate ligands. J. Agron., 5: 406-417.
CrossRefDirect Link - Hiemstra, T., P. Venema and W.H. Van Riemsdijk, 1996. Intrinsic proton affinity of reactive surface groups of metal (hydr)oxides the bond valence principle. J. Colloid Interface Sci., 184: 680-692.
Direct Link - Liu, C. and P.M. Huang 2001. Pressure-Jump relaxation studies on kinetics of lead sorption by iron oxides formed under the influence of citric acid. Geoderma, 102: 1-25.
Direct Link - Xu, Y., T. Boonfueng, L. Axe, S. Maeng and T. Tyson, 2006. Surface complexation of Pb (II) on amphorous iron oxide and manganese oxide: Spectroscopic and time studies. J. Colloid Interface Sci., 299: 28-40.
Direct Link - Strawn, D.G., A.M. Scheidegger and D.L. Sparks, 1998. Kinetics and mechanisms of Pb(II) sorption and desorption at the aluminum oxide-water interface. Environ. Sci. Technol., 32: 2596-2601.
Direct Link








