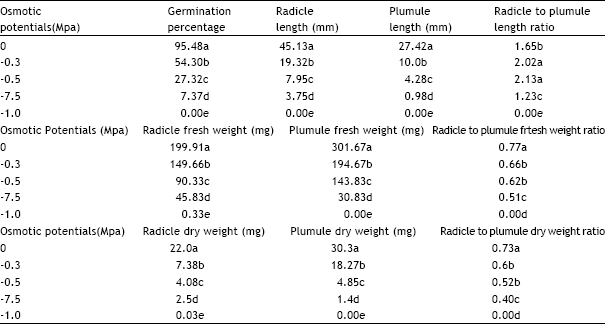
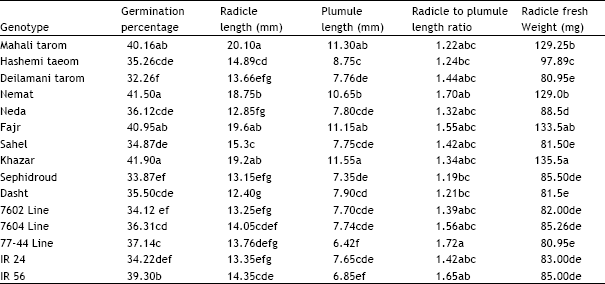
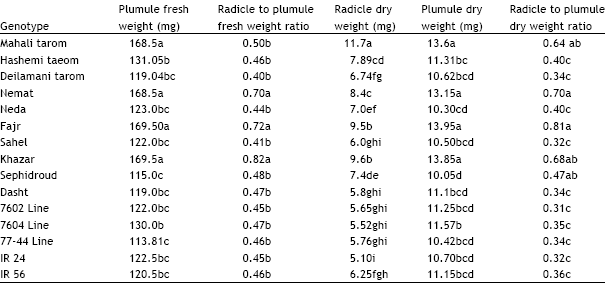
Research Article
Effect of Water Stress on Seed Germination and Seedling Growth of Rice (Oryza sativa L.) Genotypes
Faculty of Agriculture, University of Tarbiat Modarres, P.O. Box 14155-4838, Tehran, Iran Faculty of Agriculture, University of Mazandaran, Sari, Iran
Z. Tahmasebi Sarvestani
Faculty of Agriculture, University of Tarbiat Modarres, P.O. Box 14155-4838, Tehran, Iran
GH. Nematzadeh
Faculty of Agriculture, University of Mazandaran, Sari, Iran
A. Ismail
Crop, Soil and Water Science Division, International Rice Research Institute, DAPO P.O. Box 7777, Metro Manila, Philippines