Research Article
Occurrence of Helminth Parasites in Schizothorax plagiostomus and Cyprinus carpio communis from Nallah Sukhnag, Kashmir
Department of Zoology, University of Kashmir, Jammu and Kashmir, India
LiveDNA: 91.20065
Syed Tanveer
Department of Zoology, University of Kashmir, Jammu and Kashmir, India
LiveDNA: 91.22736
Showkat Ahmad Ganie
Department of Clinical Biochemistry, University of Kashmir, Jammu and Kashmir, India
LiveDNA: 91.10673









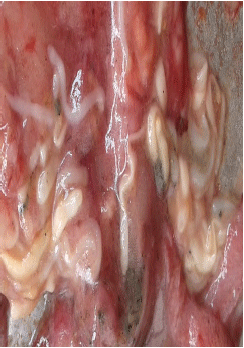
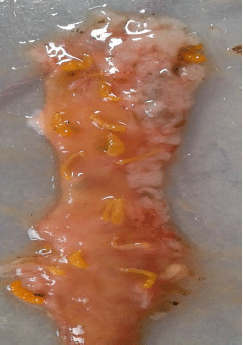
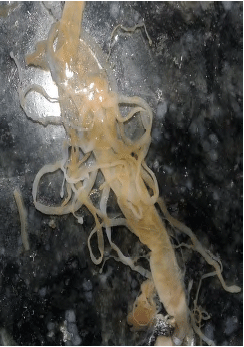

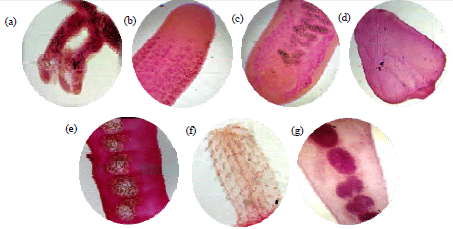




Rashaid Ali Mustafa Reply
Need copyright of Fig. 1 and Fig.2
Editor
We would be happy to grant permission for the use of the figures, provided that proper attribution is given to the original source. Please provide us with the details of the intended use of the figures and we will review your request and provide you with further information.