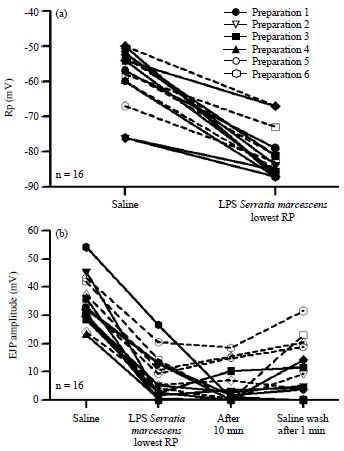
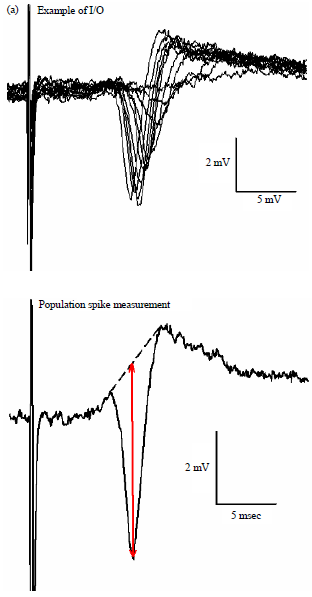
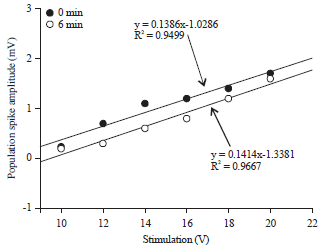
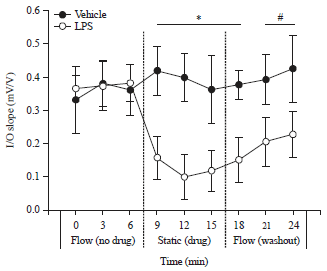
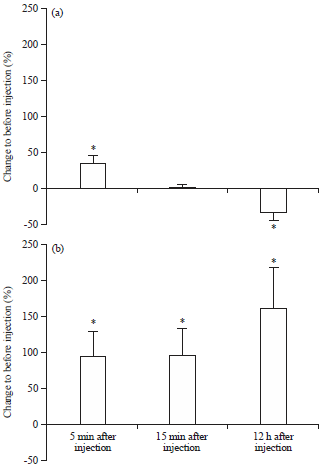
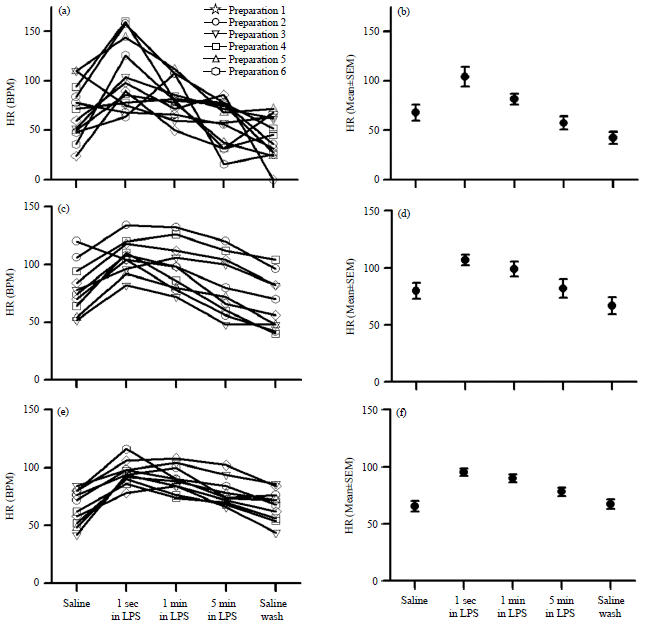
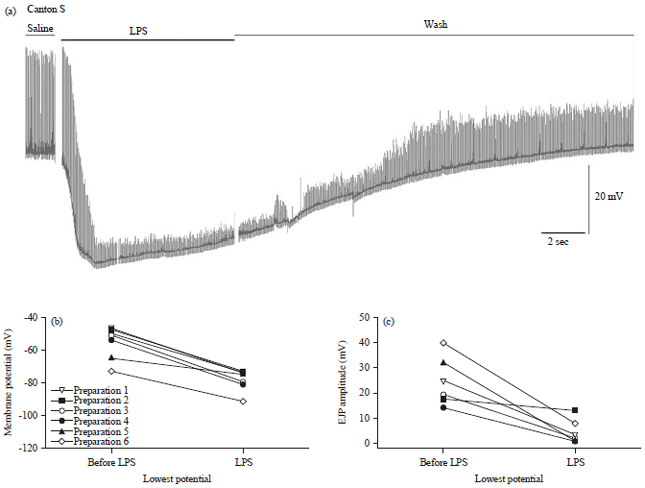
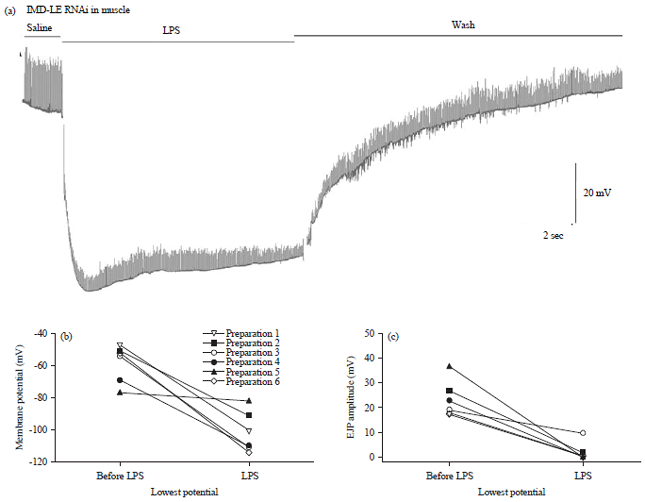
Review Article
Effects of Bacterial Endotoxin (LPS) on Cardiac and Synaptic Function in Various Animal Models: Larval Drosophila, Crayfish, Crab and Rodent
Department of Biology, University of Kentucky, Kentucky, United State of America
Ogechi Anyagaligbo
Department of Biology, University of Kentucky, Kentucky, United State of America
Jate Bernard
University of Virginia, Charlottesville, Virginia, United State of America
Sonya M. Bierbower
Department of Biology, William Paterson University, Wayne, New Jersey, United State of America
Esther E. Dupont-Versteegden
Division of Physical Therapy, Department Rehabilitation Sciences, College of Health Sciences, University of Kentucky, Kentucky, United State of America
Adam Ghoweri
Department of Pharmacology and Nutritional Sciences, University of Kentucky, Kentucky, United State of America
Abigail Greenhalgh
Department of Biology, University of Kentucky, Kentucky, United State of America
Doug Harrison
Department of Biology, University of Kentucky, Kentucky, United State of America
Oscar Istas
Department of Biology, University of Kentucky, Kentucky, United State of America
Micaiah McNabb
Department of Biology, University of Kentucky, Kentucky, United State of America
Christa Saelinger
Department of Biology, University of Kentucky, Kentucky, United State of America
Alex Stanback
Department of Biology, University of Kentucky, Kentucky, United State of America
Maddie Stanback
Department of Biology, University of Kentucky, Kentucky, United State of America
Olivier Thibault
Department of Pharmacology and Nutritional Sciences, University of Kentucky, Kentucky, United State of America
Robin L. Cooper
Department of Biology, University of Kentucky, Kentucky, United State of America
LiveDNA: 1.523