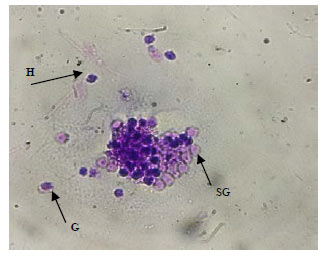
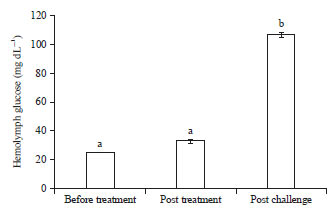
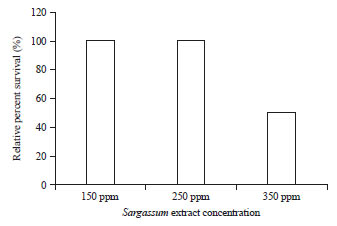
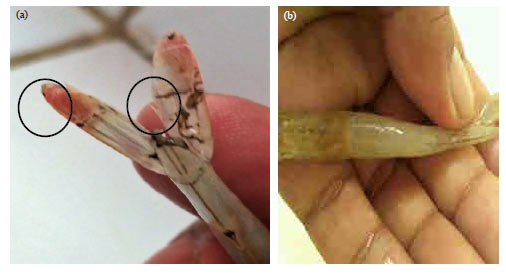
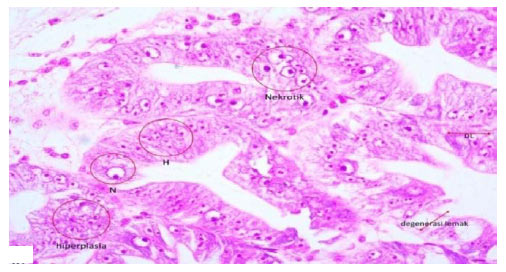
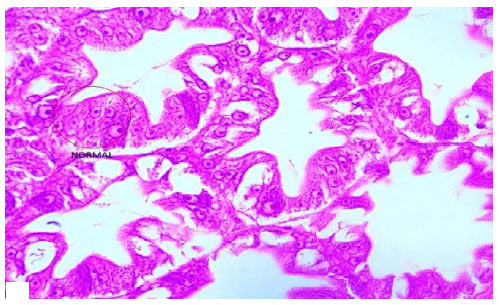
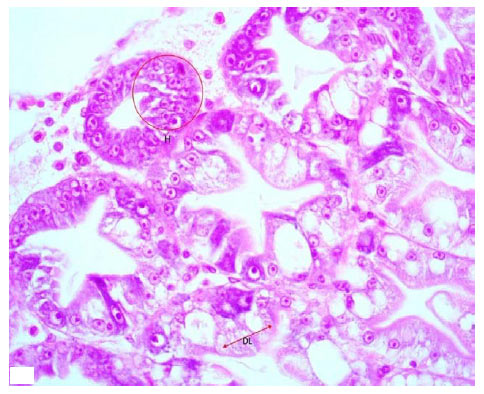
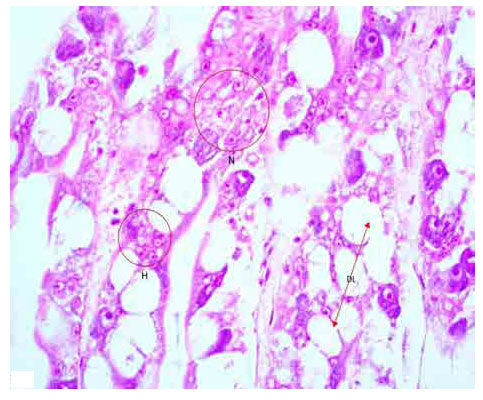
Research Article
Efficacy of Seaweed (Sargassum sp.) Extract to Prevent Vibriosis in White Shrimp (Litopenaeus vannamei) Juvenile
Department of Aquaculture, Faculty of Fisheries and Marine Science, Halu Oleo University, 93232 Kendari, JL. HEA Mokodompit, Anduonohu, South East Sulawesi, Indonesia
Indriyani Nur
Department of Aquaculture, Faculty of Fisheries and Marine Science, Halu Oleo University, 93232 Kendari, JL. HEA Mokodompit, Anduonohu, South East Sulawesi, Indonesia
LiveDNA: 62.27287
Wa Iba
Department of Aquaculture, Faculty of Fisheries and Marine Science, Halu Oleo University, 93232 Kendari, JL. HEA Mokodompit, Anduonohu, South East Sulawesi, Indonesia