Research Article
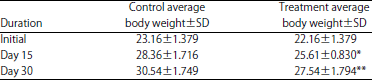
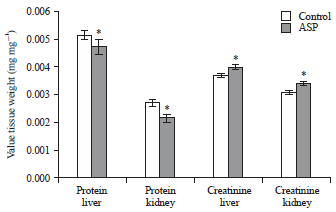
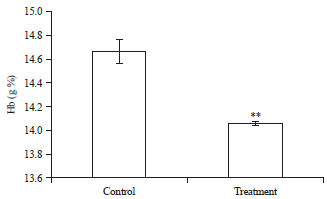
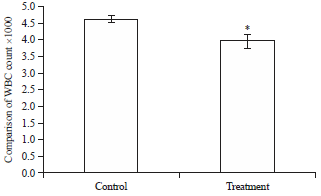
Effects of Short-term Consumption of Aspartame on Some Biochemical and Hematological Parameters in Female Swiss Albino Mice
Laboratory of Endocrinology, Department of Biosciences, Faculty of Life Sciences, Barkatullah University, Bhopal-462026 (M.P.), India
LiveDNA: 91.23608
Vinoy Kumar Shrivastava
Laboratory of Endocrinology, Department of Biosciences, Faculty of Life Sciences, Barkatullah University, Bhopal-462026 (M.P.), India
LiveDNA: 91.28873