Research Article
Assessment of Effectiveness of the Imidacloprid and Azadirachtin on the Black Watermelon Bug
Department of Biology, Faculty of Science, University of Jeddah, Jeddah, Saudi Arabia
LiveDNA: 966.24834
Demand for food has been increasing; a more comprehensive understanding of the impact of landscapes on sustainable agriculture production is required to meet the continual increase in human demand. This includes how chemical are used but also how cultivated and surrounding landscapes are managed to maximize ecosystem services. Watermelon Citrulus lanatus (Thuab.) is considered as the second most-produced vegetable, with regards to production quantity, with 89.1 million t produced in 2010 and a total worldwide production area1,2 of 3.2 million ha. It was reported that the genetic origin of the watermelon is Africa but the exact geographical origin and domestication process of the crop is not clear. One possible genetic center is in the Kalahari Desert region where various wild forms of the species can still be found but it has also been suggested that the origin is in the Sahel Region in Northern Africa3. Watermelon has been grown in Africa and the Middle East for thousands of years. Throughout history, watermelon was distributed throughout the world as trade and knowledge4.
There are several insects causing damage on important watermelon crop species, one of which is the black watermelon bug (Coridius viduatus) Fabricicus (F.) (Heteroptera: Dinidoridae). The genus, Coridius, including this species5. Afterward, this species was represented in Aspongopus genus by Stal6 but current research classifies this species under Coridius genus7. The black watermelon bug (Cordius viduatus) is one of the most destructive pests threatening the watermelon and is widely distributed in the Near East and Africa. It is present in almost all of Africa, the Arabian Peninsula, Egypt and Turkey. Optimum temperature and humidity, adequate rainfall and good vegetation cover create suitable conditions for the insect survival1,8-10 female black watermelon bug deposits approximately 287-295 eggs, sometimes up to 741 eggs, over a 150 day period. Eggs are laid individually or in clusters or rows on the main stems or lower parts of leaves, of the host plants8. Also, the insect has the ability to paste eggs together so that they can be lifted as one long stick of eggs1,8,9. Oviposition period was long (64 days), which may be due to the long rainy season and good climatic conditions that influence the fecundity and which resulted in the increase in the number of eggs and spontaneous population increases. Nevertheless, hatchability, nymphal mortality, predators, parasites, pathogens and the climate are the important factors influencing population density and should be considered in the strategy of the pest management1. A survey of arthropods associated with cucurbit crops during 2011 and 2012 at the New Valley in Egypt found the existence of 28 insect species belonging to 25 genera under 20 families of 9 orders. The important piercing-sucking arthropod pests were the black melon bug, Coridius viduatus F. on the melon1,11. Previous studies showed that C. viduatus is considered to be the most important piercing-sucking arthropod pest in cucurbit plants, causing very serious yield losses if no chemical control is applied11.
Imidacloprid is a synthetic chemical insecticide widely used for controlling insect pests. In turfgrass and ornamental settings, Imidacloprid has been shown to effectively control adelgids, aphids, lace bugs, leafminers, mealybugs, scales, thrips, whiteflies, elm leaf beetles, leafhoppers and Japanese beetles12. This insecticide is used less often because of its highly negative effect on non-target organismss13 but such organophosphates also have poor environmental and toxicological profiles. The unique mode of action of Imidacloprid, the degree of systemic and contact activity, a variety of application methods, low application rates, extended residual control, resilient binding to soil organic matter and good environmental and toxicological profiles result in this being one of the most widely used agents for chemical control14. Several previous studies have documented that insect susceptibility to neonicotinoids is dependent upon dosage, exposure route and species. Exposure to neonicotinoids may kill insects outright or may cause altered behaviors via sublethal effects15,16.
The problems caused by organo synthetic insecticides on the environment and non-target organisms17,18 has stimulated the use of natural products as an alternative pest control strategy, mainly in developing countries19. In general, these natural products have a lower environmental persistence and therefore, are considered environmentally and toxicologically safer than several of the currently used organo synthetic pesticides19. In addition to lethal effects20, natural products, such as botanical insecticides, might have repellent effects, inhibit oviposition and change the feeding and hormonal systems of several arthropod pests21.
These characteristics made the use of these biopesticides attractive for pest management. Plant-based insecticides such as Neem (i.e., Azadirachtin) have a range of commercial formulations that exhibit good efficacy against more than 400 insect species22. In fact, many scientific studies and have shown that Neem are a successful model in the natural control of many agricultural pests. The possibility of using nontoxic deterrents and repellents as crop protectants and the concept of using insect antifeedants such as Azadirachtin on a large number of pest species is intuitively attractive23. The remarkable bioactivity of Azadirachtin from the Indian Neem tree (Azadirachta indica) led to the search for natural insecticides that can be used in the natural control of many agricultural pests. Botanical insecticides can be obtained from seeds of the Indian Neem tree, Azadirachta indica24. Neem oil could be effective against insects and mites and is useful in the management of phytopathogens. Neem seeds contain more than a dozen Azadirachtin analogs but the major form is Azadirachtin and the remaining minor analogs likely contribute little to the overall efficacy of the extract. The role of these other natural substances has been controversial but most evidence points to Azadirachtin as the most important active ingredient21. However, the authorization to use these products remains controversial worldwide and although the activity of these botanical insecticides on agricultural pests is well established, their effects on other organisms need to be studied more comprehensively25.
Due to the economic benefits of watermelon and the effects of the exposure to several insect pests, specifically, the black watermelon bug Coridius viduatus (F.), it is from very harmful agricultural pests. So, in this study the exposure of some insecticides on this insect was determined and the acute toxicity and sublethal effects of some insecticides was assessed including Imidacloprid and Azadirachtin, which that is of the most used insecticides in agriculture and determine the effect of the use of different concentrations and periods different from it. Also, the concentration of lethal concentration (LC50) and lethal time (LT50) was studied.
Study samples: This study considered an insect Coridius viduatus (F.) (Heteroptera:Dinidoridae) as a harmful pest of the melon plant because it feeds on the juice of the cell, affecting its productivity. It is a light black insect with medium size, the mouth is a piercing pipette which is used to absorb the cellular juice from the leaves of the plant. Its front wings are semi-sheath and the membrane background is used for flight (Fig. 1).
Insecticide used in the study: Firstly, Imidacloprid insecticide; Trade name: Imidaclorin, soluble insecticide active ingredient: Imidacloprid (20% SL), the general name of pesticide: Imidacloprid 20 SL, chemical group: Neonicotinoid, based on available information, is a pesticide that works by contact. Second: Azadirachtin insecticide: trade name: Amen, which is a pesticide of plant origin, chemical group: From the botanical insecticide group. Insecticide: An insecticide of plant origin in the form of an Emulsifying Center (EC). Active ingredient: Azadirachtin 1% (EC). The properties of the insecticide: A selective insecticide, it is effective on all larval stages and pupae, they reduce crop damage by repelling and deterring feeding of all stages of insects. It is a pesticide commonly used in the control of agricultural pests (Fig. 2).
 | |
| Fig. 1: | A black watermelon bug (Coridius viduatus (F.)) (12MP-10X) |
Concentrations used: Different concentrations (0, 0.2, 1, 5, 6) were used as parts per billion (ppb)14.
Procedures of the experiment: The study samples were collected in the summer of 2018 (July) from farms located south of Jeddah (21°15'00.7"N 39°17'27.0"E) that are famous for the cultivation of watermelon. Samples were collected and transported directly to the laboratory. Laboratory experiments were carried out under the influence of the laboratory where the temperature was 25±5°C and the humidity was 65±10% relative humidity.
The samples were placed at a rate of 15 insects in a container. They were supplied with watermelon leaves for feeding and a small container with water. The container was covered with a piece of gauze to help it ventilate and prevent the insects from escaping. The insecticides of Imidacloprid and Azadirachtin were applied in five groups per insecticide (dosages of 0, 0.2, 1, 5, 6 ppb), with each group placed in a separate container and to calculate the mortality of individuals.
Divisions study groups
First: Imidacloprid insecticide:
| • | Treatment group with Imidacloprid with a concentration of 0 ppb (control group) |
| • | Imidacloprid treatment group with a concentration of 0.2 ppb |
| • | A treatment group with Imidacloprid with a concentration of 1 ppb |
| • | Treatment group with Imidacloprid pesticide with a concentration of 5 ppb |
| • | Treatment group with Imidacloprid pesticide with a concentration of 6 ppb |
 | |
| Fig. 2(a-d): | (a) Imidacloprid insecticide, (b) Azadirachtin insecticide and (c, d) Insect black watermelon bug in the experimental bowl |
Second: Azadirachtin insecticide:
| • | Azadirachtin treatment group with a concentration of 0 ppb (control group) |
| • | Azadirachtin treatment group with a concentration of 0.2 ppb |
| • | Azadirachtin treatment group with a concentration of 1 ppb |
| • | Azadirachtin treatment group with a concentration of 5 ppb |
| • | Azadirachtin treatment group with a concentration of 6 ppb |
Statistical analysis: Percentage of mortalities and longevity of black watermelon bug was corrected for control mortalities using Abbott’s formula26 and the LC50 value was determined after 12 h using27,28. The lethal halftime, LT50 was determined using LC50 concentration. Statistical analysis was conducted by a program (Ldp line). Also, the confidence limits of the LC50 and LT50 value were calculated as described by Litchfield and Wilcoxon29.
The results of the current study, which evaluated the toxicity of four different concentrations (0, 0.2, 1, 5, 6 ppb) of Imidacloprid and Azadirachtin insecticides on the black watermelon bug after 24 h of treatment. The lethal concentration of individuals (LC50) is the concentration that caused in the death of half of the individuals after 24 h. The lethal halftime (LT50) was defined as the time at which the highest death rate for individuals is achieved when exposed to the LC50 dosage, with observations conducted at 1, 3, 6, 10, 12 and 24 h. The group that was not treated with any type of insecticide (control group, 0 ppb) was fed naturally and did not show any type of changes nor was any deaths observed in the control group. Groups exposed to insecticide have the following effects.
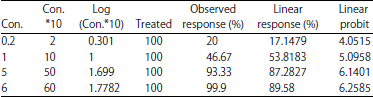
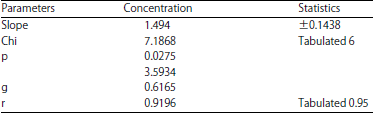
Imidacloprid: Mortality at a lethal concentration (LC50): Of the obtained results in Imidacloprid, observed response rate was 20-99.9% at the concentration of 0.2 and 6 ppb, respectively, while the linear response rate was the lowest at the concentration of 0.2 ppb, the highest percentage of 89.58% at the concentration 6 ppb. The linear probity range ranged from 4.0515-6.2585 at the concentration of 0.2 and 6 ppb, respectively (Table 1). The slope of the regression line was (1.494±0.1438). The Chi and tabular values were calculated and the value of Chi (computed) (7.1868) and tabulated (6) were found at degrees of freedom (n-2). A strong relationship between the concentration and the death rate (r) was observed, with correlation coefficients of 0.9196 (Table 2). The concentration LC25 = 0.305 ppb resulted in 25% death, whereas LC50 = 0.8627 ppb in the death of 50%. Based upon these statistics, 99% death is at a concentration of LC99 = 31.1137 ppb. The regression line, which represents the relationship between the death ratios of the black watermelon bug and the concentrations of the Imidacloprid were plotted. From the Ldp line, the value of the concentration that kills 50% of the LC50 was determined. It was 0.8627 ppb as in Table 3.
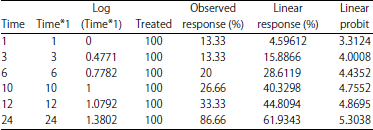
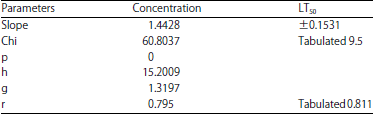
Lethal time (LT50): In the current study, lethal concentration (LC50) was calculated from an Imidacloprid at which there was 50% mortality of the black watermelon bug after 24 h (LC50 = 0.8627 ppb) (Table 3). It was exposed to this concentration of the insecticide and the death rate was evaluated at times 1, 3, 6, 10, 12, 24 h. The lethal half time, LT50 was calculated as the time at the highest death rate. The results obtained from Table 4 showed that the observed response rate ranged from 13.33-86.66% at 1 and 24 h, respectively. The linear response rate was 4.59612% at 1 h it is less valuable and the highest ratio its 61.9343% at 24 h. Linear probity ranged from 3.3124-5.3038 between 1 and 24 h, respectively (Table 4). The slope of the regression line was calculated (0.1531±1.4428). The Chi and tabular values were also calculated and the calculated Chi-value (60.8037) and the scale (9.5). The table shows a strong relationship between the concentration and the death rate (r) was observed, with a correlation coefficient of 0.795 and tabular scale value of 0.811 (Table 5).
| Table 1: | Median lethal concentration (LC50) of Imidacloprid on black watermelon bug and risk assessment after 24 h of exposure |
 | |
| Table 2: | Statistics from the analysis of the capacities of the black watermelon bug that treated by different concentrations of Imidacloprid |
 | |
| r: Death rate | |
| Table 3: | Lethal Concentration (LC) values of Imidacloprid after exposure of black watermelon bug to different concentrations of it |
 | |
| Table 4: | Lethal Time (LT) of Imidacloprid after exposure to lethal concentration (LC50) on the black watermelon bug |
 | |
| Table 5: | Statistics from the analysis of the capacities of the black watermelon bug that treated with Imidacloprid at a lethal time |
 | |
| r: Death rate | |
The confidence limits of the LT50 value were calculated according to the Litchfield and Wilcoxon29 methods for statistical analysis and LT time was calculated at confidence intervals of 25, 75, 90, 95 and 99% (Table 6).
Azadirachtin: Mortality at a lethal concentration (LC50): When these insects were exposed to Azadirachtin, the observed response rate was 6.66-66.66% at the concentration of 0.2 and 6 ppb, respectively, while the linear response rate was the lowest in concentration 0.2 ppb, with the highest concentration at 6 ppb.
| Table 6: | Lethal Time (LT) values of Imidacloprid after exposure of black watermelon bug to different concentrations of it |
 | |
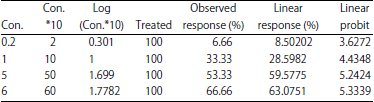
| Table 7: | Median lethal concentration (LC50) of Azadirachtin on black watermelon bug and risk assessment after 24 h of exposure |
 | |
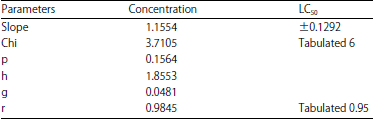
| Table 8: | Statistics from the analysis of the capacities of the black watermelon bug that treated by different concentrations of Azadirachtin |
 | |
| r: Death rate | |
| Table 9: | Lethal concentration (LC) values of Azadirachtin after exposure of black watermelon bug to different concentrations of it |
 | |
Linear probity ranged between 3.6272-5.3339 at the concentration of 0.2 and 6 ppb, respectively (Table 7). The slope of the regression line was determined as 0.1292±1.1554. The Chi-values of the data (Chi) calculated to (3.7105) and the tabular value of (6) was calculated at the degrees of freedom (n-2). There was a strong correlation between the concentration and the death rate (r), with a correlation coefficient of (0.95) (Table 8). The concentration of LC25 = 0.8043 ppb was found to result in 25% death, while LC50 = 3.0846 ppb lead to 50% death of and the 99% death rate would occur at a concentration of LC99 = 318.165 ppb.
| Table 10: | Lethal Time (LT) of Azadirachtin after exposure to lethal concentration (LC50) on the black watermelon bug |
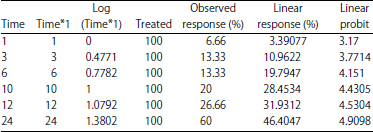 | |
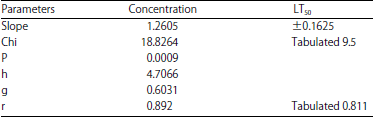
Table 11: | Statistics from the analysis of the capacities of the black watermelon bug that treated with Azadirachtin at a lethal time |
 | |
| r: Death rate | |
| Table 12: | Lethal Time (LT) values of Azadirachtin after exposure of black watermelon bug to different concentrations of it |
 | |
The regression line, which represents the relationship between the death rates of the black bug and the concentrations of Azadirachtin was drawn. From the Ldp line, the value of the concentration that death 50% of the LC50 was determined. It was 3.0846 ppb as in Table 9.
Lethal Time (LT50): A sample of black bugs was exposed to the LC50 concentration of Azadirachtin, LC50 (3.0846 ppb). Follow-up measurements were conducted at 1, 3, 6, 10, 12, 24 h after exposure to determine the fatal half time LT50. The results in Table 10 showed that the observed response rate ranged from 6.66-60% at 1 and 24 h, respectively. The linear response rate was less than 3.39077% at 1 h and the highest rate of 46.4047% at 24 h. Linear probity ranged from 3.17-4.9098 between 1 and 24 h, respectively in Table 10. The slope of the regression line (0.1625±1.2605) was calculated. The Chi and tabular values were also calculated and the value of Chi (calculated 18.8264) and the scale (9.5) was found at the degrees of freedom (n-2) (Table 11). The confidence limits of the LT50 value were calculated according to the Litchfield and Wilcoxon 29 methods of statistical analysis and the LT time ratio was calculated for confidence intervals of 25, 75, 90, 95 and 99% (Table 12).
| Table 13: | Comparison of the LC50 mortality rate of the effect of Imidacloprid and Azadirachtin on the black watermelon bug after 24 h of exposure |
| Index compared with Imidacloprid, Resistance Ratio (RR) compared with Imidacloprid | |
| Table 14: | Comparison of the half-lethal time of (LT50) for the effect of Imidacloprid and Azadirachtin on the black watermelon bug after 24 h of exposure |
| Index compared with Imidacloprid, Resistance Ratio (RR) compared with Imidacloprid | |
Comparison of the effects of Imidacloprid and Azadirachtin on the black bug
According to the mortality LC50: The results of the current study showed that Imidacloprid (LC50 of 1.658 ppb) has a stronger effect in the elimination and mortality of black bugs compared to Azadirachtin (LC50 of 3.085 ppb) based upon a lower dosage that leads to 50% mortality (Table 13).
According to longevity and the lethal half time LT50: The results of the current study showed that the Imidacloprid pesticide needs less time in the elimination and death of the black bug population when using the half-lethal concentration. At the LC50 dosage, it takes 14.779 h to kill half of the individuals using Imidacloprid compared to 28.3 h using Azadirachtin (Table 14).
Insecticide treatments are under scrutiny because of their variable efficacy against crop pests and for their potential impacts on organisms in agroecosystems. In this study, the group that was not treated with any type of insecticide (control group) and were fed naturally did not show any changes, continued to the end of the experiment and no deaths were observed30.
In the obtained results for black watermelon bug exposure to Imidacloprid, the concentration LC25 = 0.305 ppb resulted in 25% death, whereas LC50 = 0.8627 ppb in the death of 50%. The 99% death concentration was calculated as LC99 = 31.1137 ppb. Majority of researchers have documented that insect susceptibility to neonicotinoids is dependent upon dosage, exposure route and species. Exposure to neonicotinoids may kill insects outright or may cause altered behaviors via sublethal effects15,16. From the current study, Imidacloprid had an observed response rate of 20% at a concentration of 0.2 ppb. This low concentration caused a high rate of mortality, consistent with prior results that observed impaired mobility in ants treated with low dosages of Imidacloprid and fungicide treated seeds31. A study by Dilling14 revealed that the differences between soil drenched and soil injection Imidacloprid concentrations and respective effect on non-target canopy insects may represent a threshold of tolerance; however, the correlation between Imidacloprid concentration and LC50 of non-target insects is not known and the LC50 of Imidacloprid on hemlock wooly adelgid is loosely correlated with existing estimates varying from 120-300 ppb. This is an area in need of future research. Additionally, species-level interaction differences with Imidacloprid were seen in how ant species’ abundances responded to seed dressing with this neonicotinoid. Although, some species were not affected, pitfall trap captures of others increased or decreased with the addition of Imidacloprid to the system. However, some insects such as aphids were not negatively impacted by the Imidacloprid seed dressing despite aphids being among the targeted pests of this insecticide30. This study showed when a black watermelon bug is exposed to an LC50 concentration of Imidacloprid, the lethal time calculated as LT50 = 14.7794 h and LT99 = 605.2896 h. The sublethal effects from Imidacloprid might impact in other ways such as their ability to reproduce, longevity or aggression of a colony31. One potential cause is that the mechanism of neuro-toxic insecticides, such as Imidacloprid, was related to its nicotinic acetylcholine receptor affinity15.
On the one hand, when the black watermelon bug was exposed to Azadirachtin the concentration of found to result in 25% death was LC25 = 0.8043 ppb, while 50% death was LC50 = 3.0846 and a calculated 99% death rate concentration was LC99 = 318.165 ppb. Measurements of death rate made at the LC50 dosage at multiple times following treatment during time periods set to determine the time at 50% mortality, LT50 determined as LT50 = 28.2999 h. These results were in agreement with Omar and El-Kholy32 who found that the reduction in the population size of Coccinella undecimpunctata ranged between 12.7 and 14.2% when azadirachtins (Neemazal 5% EC) was applied against Thrips tabaci (Lind.) on onion crop. Meanwhile, Abdel-Aziz et al.33 found that the highest effect of Neem oil (Neemix) on the population of cereal aphids was observed 3 days after application. The residual effect extended for about 21 days post application with medium selective effect against total numbers of natural enemies after 3 days and good selective after 21 days. Rembold and Czoppelt34 studied the effects of Azadirachtin on honey bee larvae (Third instar larvae) by topical application. Larvae were fed with a royal jelly and yeast mixture and reared in the incubator. The lowest dose causing observable effects was 0.25 μ larva. Also, Naumann and Isman35 used an emulsifiable concentrate with an undiluted Azadirachtin content of 46000 mg kg–1. Oral application of increasing doses of Azadirachtin on first and fourth instar larvae resulted in larval ejection by nurse bees in a dose-dependent manner. The LD50 for both instars was 37 and 61 μg g–1 b.wt.
On the other hand, Neem seed oil was tested at concentrations of 5.0, 2.5, 1.2, 0.6 and 0.3% for oviposition deterrence, by Raguraman and Singh36 feeding deterrence, toxicity, sterility and insect growth regulator effects against Trichogramma chilonis Ishii. Neem seed oil at 0.3% deterred oviposition (parasitization) by the parasitoid. Neem seed oil also deterred feeding at or above a 1.2% concentration, seed oil at 5% concentration caused <50% mortality to both males and females. In contact toxicity tests, females were affected, while males did not seem to be affected. In view of the sensitivity of the parasitoid adults to oviposition deterrent and (mild) toxic effects of Neem seed oil, it is suggested that for the inundative release of the parasitoid T. chilonis pretreatment of the crop with Neem extracts should be avoided. Other research37 concluded that the effect of Neemix (Azadirachtin) pesticide against the adult and nymph stages of Bemisia tabaci and its natural enemies inhibiting cucumber plants during summer. The tested compound was more effective against the adult stage of the whitefly than the nymph stage, during the summer season. The initial mortality rates were 93.10 and 73.80%, respectively. The high effects 93.10 and 75.63% against the adult and nymph stages were recorded after one day and 11 days of application, respectively. Neem seeds typically contain 0.2-0.6% Azadirachtin by weight, so solvent partitions or other chemical processes are required to concentrate this active ingredient to a range of 10-50% in the technical grade materials used to produce commercial products21.
Azadirachtin had two significant impacts on insects. At the physiological level, Azadirachtin hinders the union and arrival of shedding hormones (ecdysteroids) prompting inadequate ecdysis in immature insects. In adult female insects, a similar mechanism of action leads to sterility. In addition, Azadirachtin is a strong antifeedant to numerous insects. The discovery of Neem by western science is credited to Heinrich Schmutterer, who saw that swarming desert beetles in Sudan defoliated all neighborhood greenery with the exception of some nearby Neem trees. Without a doubt, Azadirachtin was first disconnected due to its effective antifeedant action in the desert beetle and this substance remains the most powerful insect antifeedant found to-date. On the other hand, Azadirachtin has defied total synthesis to this point. Advanced in the Unified States by Robert Larson (with assistance from the U.S. Department of Agriculture), Neem rapidly became the modern paradigm for the development of botanical insecticides21,38.
In this study, the concentration LC50 = 1.658 ppb of Imidacloprid leads to 50% mortality, compared to Azadirachtin at a higher concentration of LC50 = 3.085 ppb. Also, it takes LT50 = 14.779 h to reach 50% mortality, compared to Azadirachtin which takes approximately LT50 = 28.3 h to reach 50% mortality. This is consistent with the study conducted by Aljedani39 where Imidacloprid and Azadirachtin at concentrations of 0.5 and 7.5 ppm on the honeybee were treated. Treatment was found most effective at 0.5 ppm, the lethal time LT50 = 10.348 and 47.535 h in Imidacloprid and Azadirachtin, respectively. Whereas at 7.5 ppm, the LT50 = 2.714 and 8.687 h in Imidacloprid and Azadirachtin, respectively. The control group showed the longest age of the honey bee workers having an LT50 = 1749.421 h.
The present study concluded that the Imidacloprid had a stronger effect on the elimination and mortality of the black watermelon bug (Coridius viduatus) compared to Azadirachtin. Also, the Imidacloprid pesticide needs less time in the elimination and death of the black bug population when using the half-lethal concentration. Finally, the study concludes that the Imidacloprid was very toxic against the black bugs, in terms of both mortality and longevity. In addition, the botanical insecticide group such as Azadirachtin had a moderate toxic effect on the population density of the black bug compared to the Imidacloprid.
The present investigation has proven the different concentrations of Imidacloprid that produce toxic effects on the black watermelon bug. The natural-origin insecticides (Azadirachtin) has also been shown as effective. Therefore, this study helps to advance the fight against this insect by using natural-origin insecticides that are considered as environmentally friendly. This can lead to the rational use of the insecticides, which reduces the cost of control and reduces environmental pollution. The motivation in assessing effects of insecticides on a black bug is that they are a common pest in cucurbits crops. Furthermore, this insect will attract the attention of scientists in the future due to its utilization as food in the human diet in some countries.