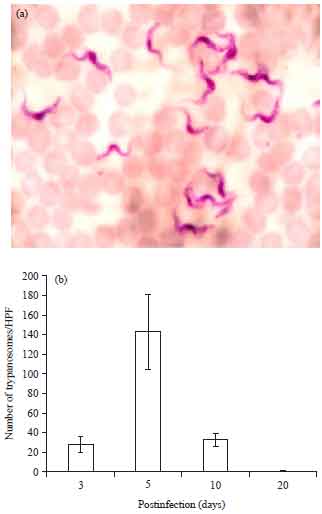
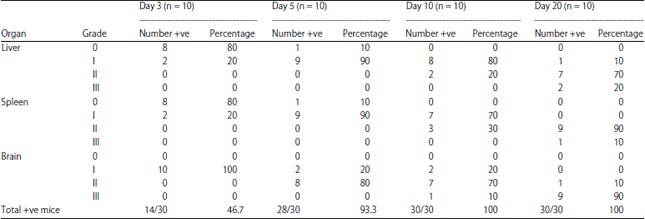
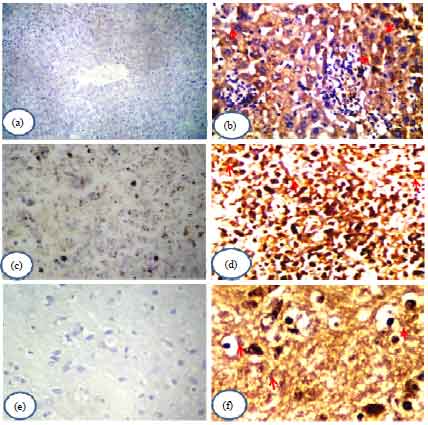
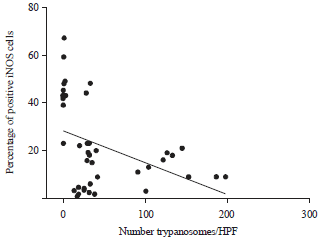
Research Article
Roles of Inducible Nitric Oxide Synthase and Interleukin-17 in Mice Experimentally Infected with Trypanosoma evansi
Department of Medical Parasitology, Faculty of Medicine, Menoufia University, Egypt
Amira Fathy Abd El-Atty
Department of Medical Parasitology, Faculty of Medicine, Menoufia University, Egypt
Manal Ahmed El-Melegy
Department of Medical Parasitology, Faculty of Medicine, Menoufia University, Egypt
Raafat Mohamed Shaapan
Department of Zoonotic Diseases, Veterinary Research Division, National Research Centre, Giza, Egypt
LiveDNA: 20.659
Amany Fawzy Atia
Department of Medical Parasitology, Faculty of Medicine, Menoufia University, Egypt
Noha Ahmed El Kaddy
Department of Pathology, Faculty of Medicine, Menoufia University, Egypt
Mohamed Mohamed Abd El-Ghaffar
Department of Medical Parasitology, Faculty of Medicine, Menoufia University, Egypt