Research Article
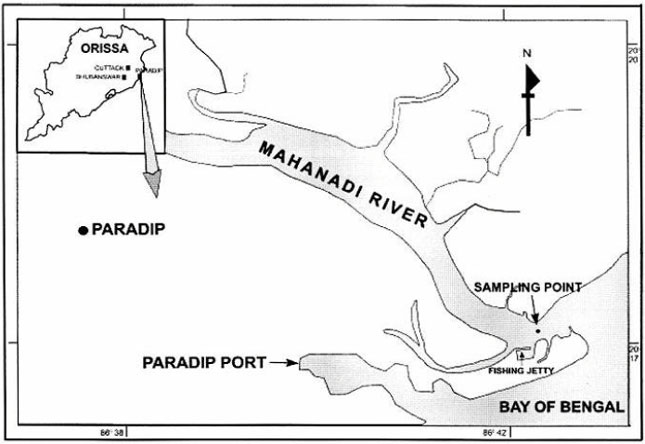
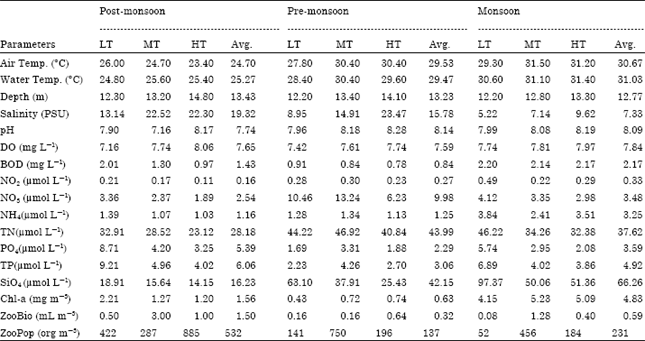
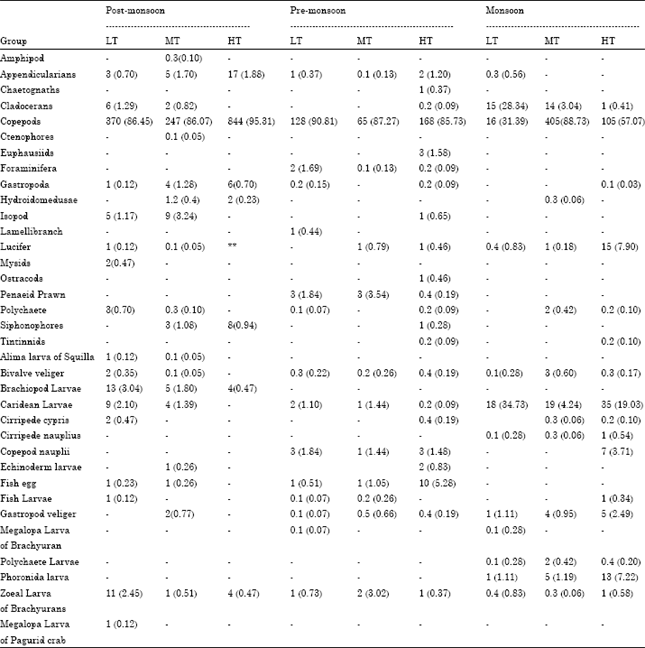
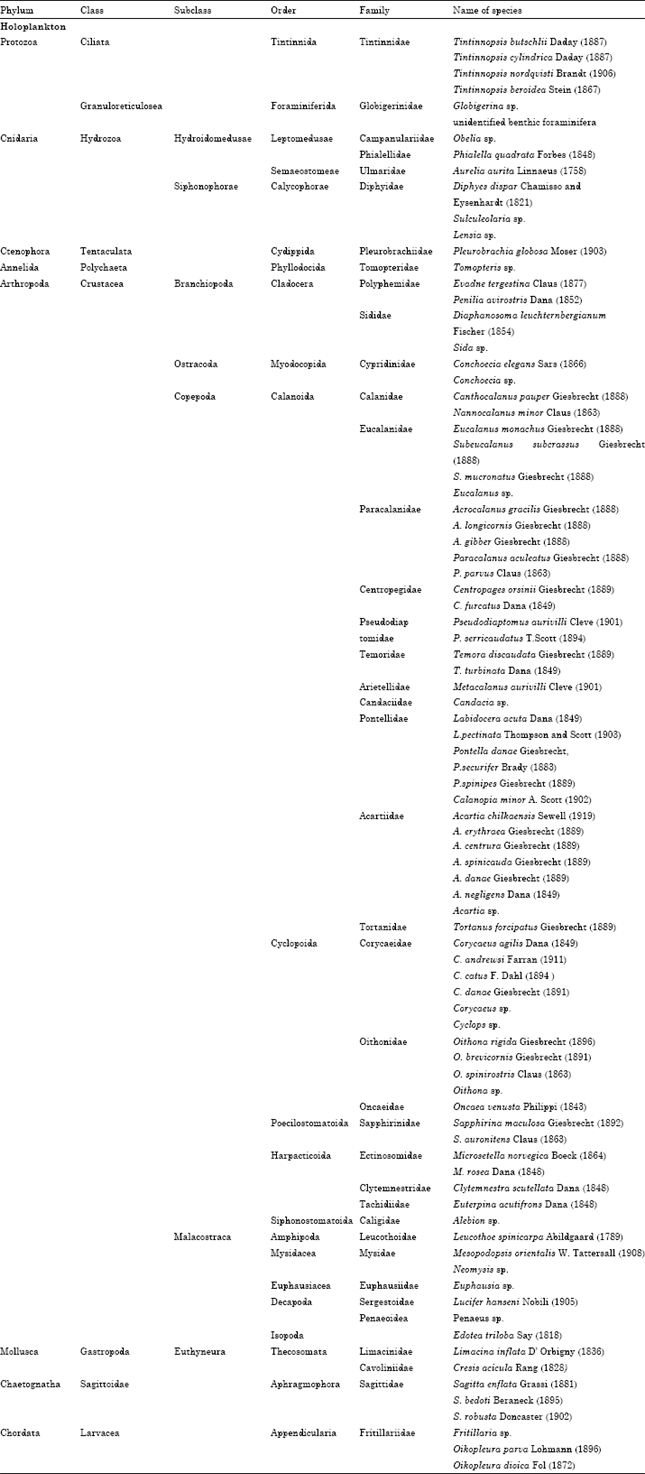
Seasonal Distribution of Zooplankton in Mahanadi Estuary (Odisha), East Coast of India: A Taxonomical Approach
Institute of Minerals and Materials Technology (CSIR), Bhubaneswar-751013, India
C. R. Panda
Institute of Minerals and Materials Technology (CSIR), Bhubaneswar-751013, India
N. C. Rout
Institute of Minerals and Materials Technology (CSIR), Bhubaneswar-751013, India