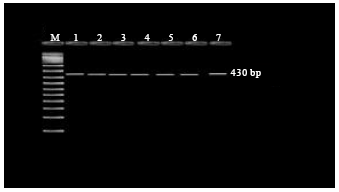
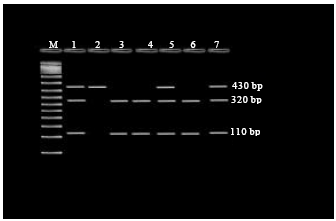
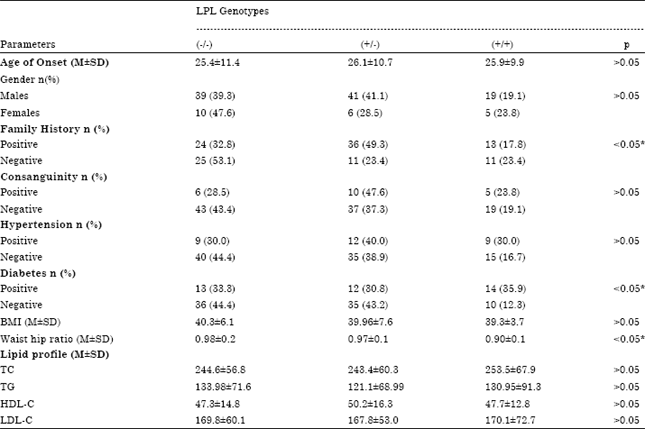
Research Article
Physiological Study of Lipoprotein Lipase Gene Pvu II Polymorphism in Cases of Obesity in Egypt
Department of Zoology, Faculty of Science, Zagazig University, Egypt
Mansour Hasan
Department of Zoology, Faculty of Science, Zagazig University, Egypt
Rizk Elbaz
Department of Genetics, Faculty of Medicine, Mansoura University, Egypt
Ghada El-Kannishy
Department of Internal Medicine, Faculty of Medicine, Mansoura University, Egypt
Saad Elshaer
Department of Zoology, Faculty of Science, Zagazig University, Egypt
Ahmed Settin
Department of Genetics, Faculty of Medicine, Mansoura University, Egypt