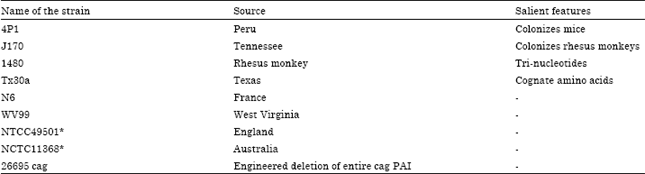
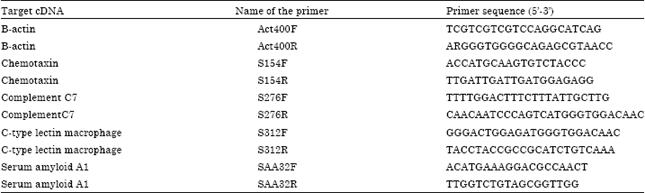
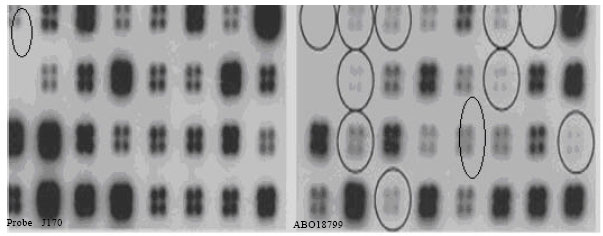
Research Article
Biotechnological Based Subtractive Hybridization and Variations of Gene Content in an Organ of Pathogenic Shrimp
St. Xaviers College (Autonomous), CPRC, Palayamkottai, Tirunelveli, India
S. Jeyaparvathy
St. Xaviers College (Autonomous), CPRC, Palayamkottai, Tirunelveli, India
M. Jasmine Priya
St. Xaviers College (Autonomous), CPRC, Palayamkottai, Tirunelveli, India