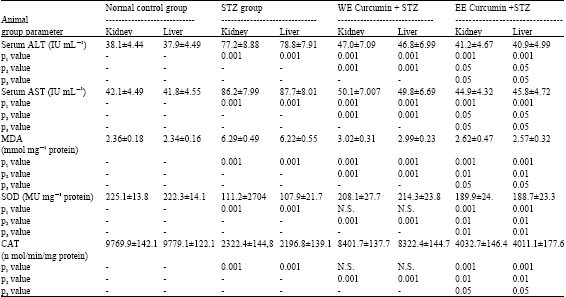
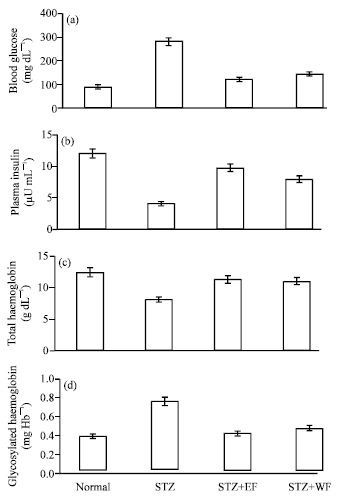
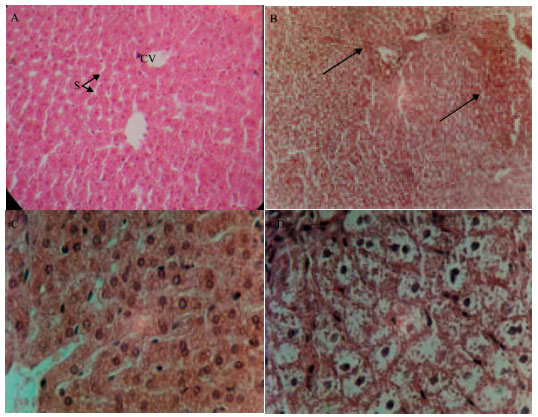
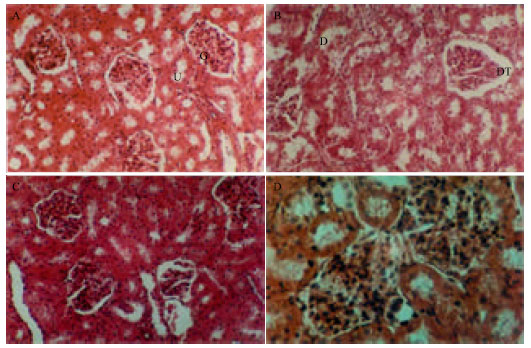
Research Article
Antioxidant Effect of Curcumin Extracts in Induced Diabetic Wister Rats
Department of Biological Sciences, Faculty of Science, King Abdul-Aziz University, Jeddah, Saudi Arabia
O. A. Abu-Zinadah
Department of Biological Sciences, Faculty of Science, King Abdul-Aziz University, Jeddah, Saudi Arabia