Research Article
Prevalence of Intestinal Parasites in Leafy Vegetables in Riyadh, Saudi Arabia
Department of Biology, College of Science, Princess Nora Bint Abdul Rahman University, P.O. Box 25701, Riyadh 11476, Saudi Arabia
Fresh vegetables are regarded as important part of a healthy diet. In many countries such leafy plants are eaten raw, or lightly cooked to preserve taste and this practice may also favors the likelihood of food-borne parasitic infections (Ozlem and Sener, 2005). Food normally becomes a potential source of human infection by contamination, during production, collection, transport and preparation or during processing and the sources of zoonotic contamination are usually faeces, faecally contaminated soil or water (Slifko et al., 2000; Daryani et al., 2008; Damen et al., 2007).
In recent years, there has been an increase in the number of reported cases of food-borne illnesses linked to consuming fresh vegetables. The consumption of raw vegetables plays a major epidemiological role in the transmission of parasitic food-borne diseases. Intestinal parasites are widely prevalent in developing countries, probably due to poor sanitation and inadequate personal hygiene (Kang et al., 1998). Several surveys in different parts of the world showed that the vegetables can be agent for transmission of protozoan cysts and oocysts (Giardia, Entamoeba, Cryptospordium, Cyclospora, Toxoplasma and Isospora) and helminths eggs and larvae (Hymenolepis, Taenia, Fasciola, Toxocara, Ascaris, Trichostrongylus, Strongyloides and Hookworms) (Vuong et al., 2007; Darchenkova et al., 2006; De Oliveira and Germano, 1992a,b).
Intestinal parasitic infections are among the most common infections worldwide (Pozio, 2008). Various epidemiological studies indicated that the prevalence of intestinal parasites was high especially in developing countries, although, in many of these, the environmental risk factors have not been clearly elucidated (Nyarango et al., 2008).
The increasing consumption of leafy vegetable plants by many people in Saudi Arabia has urged undertaking this study to investigate the parasites stages carried by these plants in the Riyadh City.
A total of 470 leafy vegetable samples from the following vegetables, were collected for the present investigation: spinach, radish, leek, parsley, basil, green onion, dill, lettuce, cabbage, watercress, coriander, mint (during period from April 2007 to March 2008). These vegetables were collected randomly from vegetable markets and bought from retail sellers at different times of the year (123 in Summer, 113 in Autumn, 91 in Winter and 143 in Spring) from Riyadh city.
A subset of each sample (250 g) was chopped into small pieces, put in a plastic bag and washed with physiological saline solution (0.95% NaCl) and the washing water/saline was left overnight. After removing bits of leaves, a volume of 300 mL of supernatant was removed and the remainder was transferred to 5 mL test tubes and centrifuged at 2000 g for 20 min. Following centrifugation, the supernatant was removed and the sediment was then examined under a light microscope (x100-400) for parasites stages (cysts, oocysts, eggs or larvae) after adding a drop of lugol iodine (Bailenger, 1962) and the parasites stages were identified according to Soulsby (1982). If the sample was positive for parasites stages, the number was counted and the developmental stage recorded.
Statistical Analysis
Comparisons between different groups was performed using the statistical software SPSS. Chi square test was used for multiple comparisons between rates of parasites recovery in different seasons of the year. p-values less than 0.05 were considered significant (Greenwood and Nikulin, 1996).
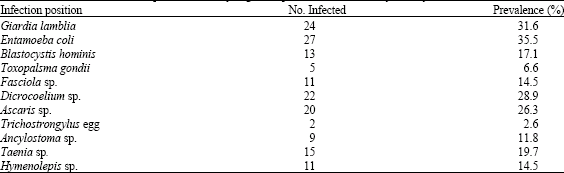
The results of the parasitic stages recovered from different leafy plants and the different parasitic stages from these plants are shown in Table 1 and 2, respectively. Out of 470 samples collected from leafy vegetables examined, 76 (16.2%) were positive for parasites stages. The highest rate of parasite stages contamination was reported from the lettuce (27.8%) and only samples collected from cabbage did not reveal any parasites. The predominant type of parasite stage was found to be cyst of Entamoeba coli which has been reported in 27 (35.5%) samples out of 76 positive samples showed parasites stages. Whereas, the lowest frequent parasite stage was found to be Trichostrongylus sp. eggs which were detected in 2 (2.6%) of the samples showed parasites stages. There was no significant difference in the frequency of reporting parasites stages from different leafy vegetables investigated (χ2 = 19.41, p>0.05).Table 3 shows the seasonal differences in the parasites stages detected in leafy vegetable plants investigated in the present study.
| Table 1: | Distribution of intestinal parasites in different leafy vegetable plants consumed in the Riyadh City |
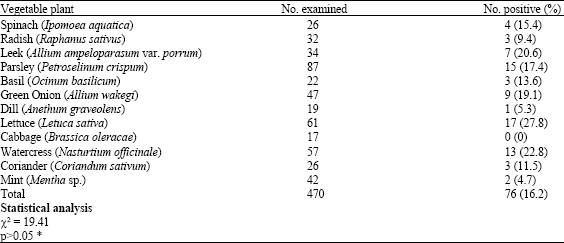 | |
| *p>0.05 no significant difference | |
| Table 2: | Prevalence of intestinal parasites in leafy vegetable plants consumed in Riyadh city |
 | |
| Table 3: | Seasonal prevalence of intestinal parasites in leafy vegetable plants consumed in the Riyadh City |
 | |
| *p<0.05 significant difference between different seasons | |
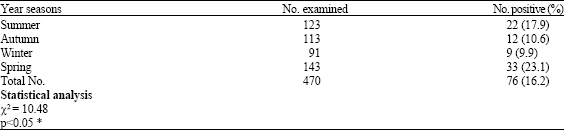
The highest rate of parasites stages was detected during the spring (23.1%) while, the lowest rate was detected during the Winter (9.9%). There was significant difference in the prevalence of parasites stages in different leafy vegetables examined in different season (χ2 = 10.48, p<0.05).
The present study has shown that investigation of 12 leafy vegetable plants used routinely for human consumption in the Riyadh city resulted in several parasitic stages carried by these plants. The consumption of raw vegetables plays an important role in the transmission of human parasitic infections (Anuar, 1977). Reporting of parasites stages from vegetables consumed as the source of contamination may be helpful in indicating the incidence of intestinal parasites among a given community. The consumption of poorly washed vegetables is regarded as a major way for transmission of parasitic contamination. Several surveys in different parts of the world showed that the vegetables can be agents for transmission of protozoan cysts and oocysts (Giardia, Entamoeba, Toxoplasma, Cryptospordium, Cyclospora and Isospora) and helminths eggs and larvae (Hymenolepis, Taenia, Fasciola, Toxocara, Ascaris, Trichostrongylus, Strongyloides and Hookworms) (Mesquita et al., 1999; De Silva et al., 1995; Vuong et al., 2007; Darchenkova et al., 2006; De Oliveira and Germano, 1992a, b). Several studies dealing with the recovery of parasites from leafy vegetables have been conducted in different parts of the world and the prevalence was high in the vegetables examined with parasite stages such as those of E. coli and G. lamblia have been reported by Robertson and Gjerde (2001), Daryani et al. (2008), Monge and Arias (1996) and Vuong et al. (2007).
A previous study carried out by Al-Binali et al. (2006) in South Western Saudi Arabia has demonstrated that eggs of Ancylostoma and Ascaris together with cysts of Entamoeba coli and Blastocystis homonis are the most common parasites stages found in the 5 leafy vegetable plants investigated. Although, Al-Binali et al. (2006) have used a method which they claimed giving good recovery, however, in the present study, a high rate of parasitic contamination in the lettuce (27.8%) compared to 17% reported by Al-Binali et al. (2006). This could be attributed to the fact that lettuce used in the present study is highly contaminated with parasitic stages. This finding is supported by the use of wastewater for cultivating crops around Riyadh city whereas, the agricultural schemes in South Western Saudi Arabia is different to this and such schemes seldom use wastewater. Previous studies have supported the findings of the present study where, the presence of intestinal parasites in vegetables may have resulted from the use of wastewater to irrigate vegetables (Kozan et al., 2007). However, Kozan et al. (2007) found out that no helminthes or developmental parasitic stages in the treated water as opposed to untreated water where, they found stages of helminthes eggs in untreated water. The presence of parasitic stages of different parasites is dependent on the parasitic fauna carried by the community and it is likely that the parasites reported from the leafy vegetables in this investigation and those of Al-Binali et al. (2006) are prevalent in the community so it contaminated the sewage. Also some studies indicated that agricultural use of untreated wastewater was the major cause of the increase in intestinal parasites (Srikanth and Naik, 2004).
The use of sewage water plays an important role in the epidemiology of transmission of parasitic diseases to human through consuming such vegetables (Gupta et al., 2009) as, it has been demonstrated in the present study.
Seasonal variation in the prevalence of parasites stages was noticed in the present study. This study shows incidence of present intestinal parasites in leafy vegetables was higher in Spring and lower in Winter as shown in Table 3 (Clavo et al., 2004; Vuong et al., 2007; Nimri, 2003). Post-harvest faecal contamination of vegetable may also occur during handling and transport of vegetables and this occur through splashing the vegetables with contaminated water in order to keep vegetable fresh and that was done through dirty vegetable containers or unhygienic handling. In which case, some of the viable parasitic cysts or oocysts will be easily transmitted to humans.
Also, many epidemiological studies have relied on excess of parasitic contamination associated with raw water reuse in irrigation (Gupta et al., 2009).
The findings of seasonal variation in parasites stages found in leafy vegetables coincided with what has been reported earlier by Nimri (2003) who found high prevalence of natural infection with cryptosporidiosis (caused by Cryptosporidium sp.) and cyclosporiasis (caused by Cyclospora cayetanensis) in the Spring compared to other seasons. Similarly, (Calvo et al., 2004; Vuong et al., 2007) found the same pattern in the leafy plants they investigated.
There is a high risk of infection with intestinal parasites in the sampled markets. The high level of leafy vegetables contamination with parasitic stages is significant hence, the inhabitants of the Riyadh City should be informed and educated with regard to food safety, good distribution practices and improvement on sanitary conditions in the local vegetable markets. People must also avoid using of untreated sewage for the irrigation of vegetables as this constitutes an important route of intestinal parasites transmission.
Dr. Yomn Mobarak Reply
How come? As cited in the fourth line of abstract the study was done during the period April and March 2008 i.e. two months. and at the end of Abstract there was seasonal variations of parasite prevalences.
Dr. Wafa A.I. Al-Megrin
This Study was done during April 2007 to March 2008 (one year)