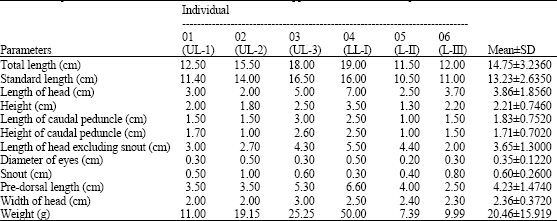
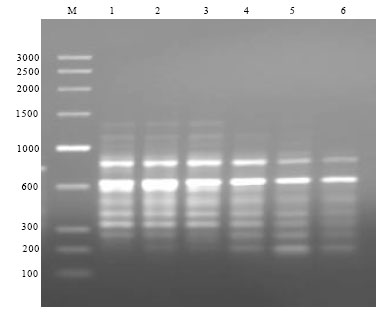
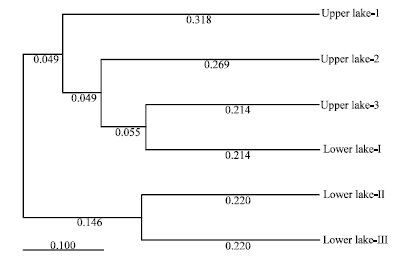
Research Article
Genetic Diversity Between Two Populations of Heteropneustes fossilis (Bloch) Using RAPD Profile
Centre of Excellence in Biotechnology, M.P. Council of Science and Technology (MPCST), Vigyan Bhawan, Nehru Nagar, Bhopal-462003 (M.P.), India
P. Sairkar
Centre of Excellence in Biotechnology, M.P. Council of Science and Technology (MPCST), Vigyan Bhawan, Nehru Nagar, Bhopal-462003 (M.P.), India
N. Silawat
Centre of Excellence in Biotechnology, M.P. Council of Science and Technology (MPCST), Vigyan Bhawan, Nehru Nagar, Bhopal-462003 (M.P.), India
N. Vijay
Centre of Excellence in Biotechnology, M.P. Council of Science and Technology (MPCST), Vigyan Bhawan, Nehru Nagar, Bhopal-462003 (M.P.), India
N. Batav
Centre of Excellence in Biotechnology, M.P. Council of Science and Technology (MPCST), Vigyan Bhawan, Nehru Nagar, Bhopal-462003 (M.P.), India
N.N. Mehrotra
Central Drug Research Institute (CDRI), Lucknow-226001 (U.P.), India