Research Article
Soybean Extract Antioxidant Protective Activity Against Copper-Induced Cytotoxicity
Department of Zoology, College of Science, King Saud University, P.O. Box 2455, Riyadh 11451, Saudi Arabia
Copper is an essential micronutrient for plants that is a component of several electron transport enzymes and is involved in catalyzing the redox reactions in mitochondria and chloroplasts (Marschner, 1995). Copper is a cofactor and an integral part of at least 13 enzymes involved in a number of vital biological processes and stimulates the immune system to fight infections (Scheinberg and Sternleib, 1976). Copper is also included in the list of heavy metals causing environmental pollution (Shaikh and Smith, 1984). Oxidative tissue damage has been linked to chronic copper exposure caused by accidents, occupational hazards and environmental pollution. Both cupric and cuprous ions can participate in redox reactions. In the presence of super oxide or reducing agents such as ascorbic acid, cupric ions can be reduced to cuprous ones that catalyze the formation of hydroxyl radicals (Abuja and Albertini, 2001; Raveh et al., 2001). Abnormal copper metabolism induces highly reactive hydroxyl radicals, oxidative damage and interfere with cellular metabolism (Abuja and Albertini, 2001; Codinach-Huix and Pamias, 2002; Diniz et al., 2004). Copper is capable of producing Reactive Oxygen Species (ROS) and impairing DNA synthesis by oxidation of nitrogenous bases (Stoewe and Prutz, 1987). It is also believed that High Density Lipoprotein (HDL) may be more susceptible to copper induced oxidation than LDL at low copper concentrations. At high copper concentrations HDL has a higher affinity to copper and increases its oxidation ability and malondialdehyde accumulation (Kadiiska et al., 1992; Bremner, 1998; Campos et al., 2003).
Flavonoids are low molecular weight polyphenolic substances based on the flavan nucleus (Cotelle et al., 1996), which found in most plant species specially fruits, vegetables and beverages. Thousands of flavonoids have been identified and classified into several groups where flavones and flavonols that occur either as aglycons or glycosides top the most common flavonoids. Many reports have shown that these compounds have potential anti-inflammatory and also suggest that certain members of this group of compounds significantly affect the function of the immune system and inflammatory cells, antineoplastic, anti-thrombotic and antioxidant activities, as well as, Flavonoids are oxidized by radicals, rendering them more stable and less reactive (Korkina and Afanasev, 1997). Cells and tissues are continuously threatened by the damage caused by free radicals and Reactive Oxygen Species (ROS) produced during normal oxygen metabolism or induced by exogenous damage (Groot, 1994). Flavones and catechines are considered to be effective for protecting the body against ROS which has a pivotal role in the action of xenobiotics associated with tissue injury and disease progression (Halliwell et al., 1992). Flavonoids α G -rutin is effective against ferric nitrilotriacetate that induces renal damage, renal lipid peroxidation and increases incidence of renal carcinoma. Investigations showed that flavonoids are capable of scavenging hydroxyl radicals, super oxide anions and lipid proxy radicals which have been implicated in a number of disease process and tissue damage (Halliwell, 1995).
Under highly oxidative stress induced Malonaldehyde accumulation (MDA) causing cellular membrane damage. Macrophages can recognize oxidized LDL in vitro and can induce oxidation catalyzed by copper ions. Also, the increase in the activities of functional enzymes in the blood serum of animals exposed to sub acute copper levels could be attributed to cell membranous system damage leading to changes in membrane permeability and intercellular metabolism (Harris, 1983; Ettinger et al., 1986). In vitro cell culture studies showed that phenolic compounds aggravate copper induced oxidative damage. Laboratory studies conducted on copper oxidative stress can be evidenced by these criteria. Oxygen-derived radicals are known to be cytotoxic to cells. During oxidative stress, reactive oxygen species, superoxide anion, hydroxyl radicals and hydrogen peroxide (H2O2) can elicit widespread damage to cell constituents such as membrane lipids. Lipid peroxidation is a free radical-mediated chain reaction, since it is self-perpetuating. The length of the propagation depends upon chain breaking antioxidant, such as the enzymes superoxide, dismutase, catalase and glutathione peroxidase (Diniz et al., 2003).
The present study was undertaken to investigate the complementary implementation of administrated natural soybean extract compounds in addition to the endogenous enzymatically and non-enzymatically defense systems to work sufficiently against copper ions induced oxidative damage associated with the overproduction of reactive oxygen species in quails Coturnix coturnix.
Animals and Housing
A total of 48 adult male quails Coturnix coturnix of the same age, weighing 190-200 g were used and housed between August and September 2005, in the animal house of the Faculty of Science, King Saud University in Riyadh city. Birds were randomly divided into six groups of 8 quails each, kept at room temperature, under simulated photoperiod regimes close to external conditions, received food and had free access to tap water for 14 days before carrying out the experiments. After that the first group received normal diet, while the diets of the second group was supplemented with soybean ethanloic extract (100 mg kg-1 diet) alone. The third and the fourth groups received copper sulphate (500 and 1000 ppm), respectively with soy extracts (100 μg mL-1), the fifth and the sixth groups supplemented copper sulphate (500 and 1000 ppm) only. After 30 days, birds were killed by decapitation, blood samples were collected and kept in plain tube to clot and serum was prepared by centrifugation and liver samples were collected from all treated animals, weighed and homogenized to be used in the biochemical tests.
Soybean Extract Chemical Constituent`s Determination
Soybean (Glycine max L.) samples were collected, washed and dried in shade at room temperature for 5-7 days before being reduced to fine powder. Fifty grams of the air-dried powdered of soybean sample were extracted with ethanol 70% (Et.1), filtered, reduced under vacuum and finally freeze dried. Total contents of tannins, saponins and flavonoids were determined according to the conventional analytical methods (Burns, 1971; Ebrahimzadeh and Niknam, 1998; Zhuang et al., 1992).
Free Radical-Scavenging Method
The antioxidant activity of soybean extract was determined on the bases of hydrogen donating and radical scavenging ability using the stable radical 2, 2-diphenyl-1-picrylhydryzyl (DPPH) (Brand-Williams et al., 1995). The inhibition percent of the DPPH by each concentration of the extract was calculated according to the formula of Yen and Duh (1994) as follows:
Inhibition (%) = [(Ac (0)-Ac (t))/Ac (0)] x 100 |
Liver Cell Culture Preparation for Viability Test
The effects of various concentration of soybean extract on the viability of quails Coturnix coturnix prepared hepatocytes were assessed in cultured cells (Moldeus et al., 1978). Cells were grown in Dulbecco`s modified Eagle`s medium (DMEM) with glucose content of 4.5 g L-1 and supplemented with the followings: inactivated fetal calf serum 10% (v/v), non-essential amino acids (1%), glutamine (1%), penicillin (100 U mL-1) and streptomycin (10 mg mL-1). The medium was adjusted at pH 7.4 and maintained in a humidified atmosphere of O2 (95%) and CO2 (5%) at 37°C. At 70-80% confluence, cells were trypsinized, centrifuged (250xg for 5 min at 4°C), re-suspended in fresh medium and plated in micro titer wells (2x104 cells/well). After attachment, they were incubated in serum-free medium containing various concentrations of soybean extract alone or together with various concentrations of copper sulphate (100, 250, 500, 750 and 1000 ppm), respectively for 24 h at 37°C.
Methylthiazolyl Tetrazolium (MTT) Assay
The metabolic competence in viable cells was relied on the conversion of yellow MTT to the purple formazan derivative by mitochondrial succinate dehydrogenase (Heras et al., 2001). Cells (2x104 cells/well) were incubated with soybean extract (1 g mL-1) for 24 h at 37°C before being washed in phosphate buffered saline and incubated in serum-free medium to which MTT (0.5 mg mL-1, 100 μL) was added. Following 4 h incubation, the medium was removed and 100 μL of acidic isopropanol (0.08 N HCl) was added to dissolve the formazan crystals. The absorbance was determined spectrophotometer at 570 nm optical density. Viability was defined as the ratio (expressed as a percentage) of absorbance of treated cells to untreated cells that served as control.
Biochemical Analysis
The following biochemical tests were carried out on the serum and/or the aliquots of the liver homogenate supernatants: Fasting Blood Glucose (FBG) (Mulec et al., 1990), Total Protein (TP) (Lall et al., 1999), Super-oxide dismutase activity (Madamanchi et al., 1994), alkaline phosphatase (ALP) (McComb and Bowers, 1972), glutamate oxaloacetate transaminase (AST) (Bergmeyer et al., 1976) and glutamate pyruvate transaminase (ALT) (Bergmeyer et al., 1976). Lipid peroxidation parameters including MDA content by thiobarbituric acid method (Buege and Aust, 1978) and glutathione peroxidase (GSH-PX) activity by using 1-chloro-2, 4 dinitrobene (CDNB) as substrate (Friedewald et al., 1972) were measured.
Serum contents of total lipids (T.L) (Burneiko et al., 2004), total cholesterol (TC.) (Aydin et al., 2001), low density lipoprotein (LDL-c) (Friedewald et al., 1972), high density lipoprotein (HDL-c) (Lopes-Virela et al., 1977), uric acid (Ihler et al., 1975) and activities of lactate dehydrogenase (LDH) (Bass et al., 1969) as indices of hepatic cell damage were assayed.
Statistical Analysis
Statistical analyses were done using SPSS (version 10) program. Mean and standard error were descriptive measures of quantitative data using the analysis of variance test (ANOVA) for independent samples using Snedecor and Cochran (1982) method. p-values <0.05 were considered significant. The difference between means of the different groups studied was tested for significant against the Least Significant Difference (LSD) (Waller and Duncan, 1969).
RESULTS
The Chemical analysis showed that soybean extracts in dry weight bases contains the following: Total tannins (2.34%), saponins (0.12%) and total flavonoids (2.54%).
The antioxidant activity of different concentrations of soybean extract according to the free radical-scavenging method is shown in Table 1 which revealed that, the highest antioxidant activity was observed with 100 ppm soybean extract on the other hand the lowest anti oxidant activity has been observed with 10 ppm.
The protective activity of soybean extract against oxidative stress initiated via copper sulphate toxicity is shown in Fig. 1, which showed a highly toxic effect on the viable liver cells which have been realized with the highest copper sulphate concentrations 1000 ppm on the other hand, the protective activity of soybean ethanloic, extract 100 ppm against copper toxicity have been observed in all copper sulphate concentrations throw the highly viability of the treated cells with soybean extracts against the non treated cells.
Table 2 indicates that, the administration of high doses of copper sulphate significantly increased the net gain, feed intake and feed efficiency, on the other hand, the addition of soybean crude extract alone significantly affected net gain and feed efficiency. On the other hand, treatment with soy extract and various copper sulphate concentrations significantly and positively affected net gain, feed intake and feed efficiency. This may be due to the synergistic effects of both copper ions as a cofactors and phenolic compounds as antioxidant natural supplementations.
The obtained data revealed a significant increase in the liver weight, which may be due to the liver dysfunction under toxic oxidative compounds during its detoxification activity.
Table 3 shows a protective activity of soybean crude extracts against liver damage initiated by copper sulphate, in addition to a recovery in the liver function tests of SGOT, SGPT and LDH enzymes in the soy extract treatment under highly copper sulphate concentrations, on the other hand there was a significant increase in the total lipids, cholesterol, HDL and LDL under copper sulphate administration alone without the soy protective extract. On the other hand, the administration of soybean extracts alone showed no significant changes in total lipids, total cholesterol and LDL contents compared with the negative control.
Table 4 shows that, no significant change in alkaline phosphatase activity, uric acid, total protein, hemoglobin, hematocrit, blood glucose concentrations were observed under soybean extract administration alone. On the other hand, a slight change has been observed with copper sulphate administration with soy extracts. While administration of copper sulphate alone significantly increase alkaline phosphatase activity and uric acid content, the total protein, hemoglobin and hematocrit were also significantly affected under the highest copper sulphate treatments 1000 ppm. The obtained data revealed that the addition of such natural compounds significantly reduced the affected kidney function and blood profile parameters and glucose levels.
| Table 1: | DPPH radical scavenging activity of various concentrations of soybean extracts and rutin standard |
 | |
| All values are mean of 3 replicates | |
| Table 2: | Feed efficiency in quails fed soybean extract supplementation with different copper sulphate concentrations |
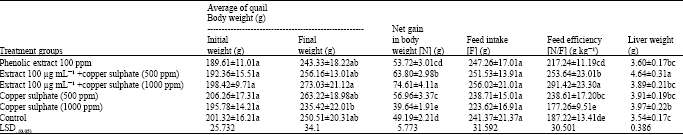 | |
| Each value is the mean of 5 replicates; Means±Standard deviation; Means in the same column with the same letter are not significantly at p<0.05 | |
| Table 3: | Effect of soybean extract against copper sulphate toxicity on liver function parameters and lipid profile |
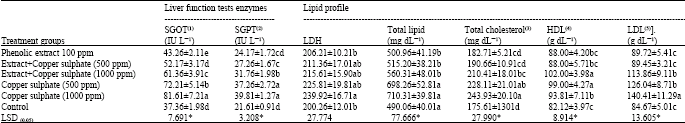 | |
| Each value is the mean of 5 replicates; Means±Standard deviation; Means in the same column with the same letter are not significantly at p<0.05 | |
| Table 4: | Protective activity of soybean extract against the change on kidney function parameters and hematologic indices induced by copper sulphate |
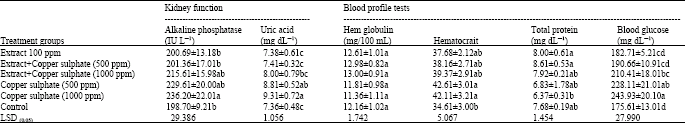 | |
| Each value is the mean of 5 replicates; Means±Standard deviation; Means in the same column with the same letter are not significantly at p<0.05 | |
| Table 5: | Effect of soybean extract against copper phosphate effect on glutathione peroxidase, supper oxide dismutase (SOD), malonaldhyde and liver of treated quails |
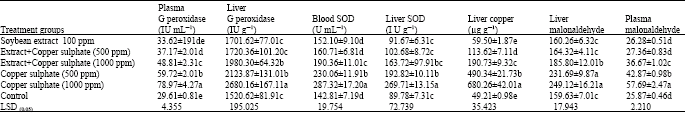 | |
| Each value is the mean of 5 replicates; Means±Standard deviation; Means in the same column marked with the same letter are not significantly different at p<0.05 | |
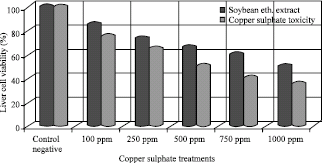 | |
| Fig. 1: | Soybean extract protection activity against copper sulphate toxicity in vitro studies |
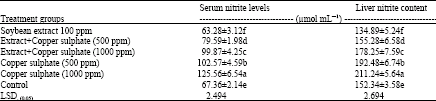
| Table 6: | Effect of soybean extract against copper phosphate on Serum and liver nitrite levels in treated quails |
 | |
| Each value is the mean of 5 replicates; Means±Standard diffusion; Means in the same column marked with the same letter are not significantly different at p<0.05 | |
On the other hand, the toxic effect of high levels of copper ions administration have been suppressed by natural antioxidants soybean extract which scavenge the free radicals initiated under copper oxidative damage.
Table 5 shows no significant effect of the only administrated soybean extract on GPX and SOD activity compared with control negative. On the other hand, the addition of these soy extracts showed a significant protection against oxidative damage and lipid peroxidation (MDA) induced by copper sulphate administration. But highly concentrations of copper sulphate alone significantly increased GPX and SOD activity and lipid peroxidation (MDA) content and a reported significant increase in liver copper content.
Table 6 shows that there was a significant decrease in the liver nitrite content in response to the administration of soybean extracts. On the other hand, the lowest decrease in the nitrite levels was observed with soybean extract against the positive control but, the administration of copper sulphate alone significantly increased the nitrite levels against the control negative and the administration of soybean extract significantly reduced the nitrite levels compared to the corresponded copper sulphate only administration.
The results of the present study revealed the presence of tannins, saponins and flavonoids in the soybean extract. Soybean extract exhibits anti-inflammatory and anti-oxidant effects due to its phenolic compounds content (Robert et al., 2001). The results of soybean chemical analysis in the present work indicated high content of natural flavonoids which are known to have significant biological activities including antiradical and antioxidant properties (Anders, 2002). Soybean extract supplementation significantly and positively affected net gain, feed intake and feed efficiency at low level of copper sulphate. This may be due to the synergistic effects of both copper ions at low concentration that act as cofactors for specific antioxidant defense enzymes.
The obtained results suggest that soybean addition possesses highly free radical- scavenging activity. This activity may be attributed to the soybean contents of natural flavonoids and other phenolic compounds in addition to the synergistic effect of ascorbic acid. Halliwell (1995) concluded that Flavonoids may enhance the free radical scavengers system.
Various studies showed that antioxidant activity against free radicals has a protective role against liver lesions induced by heavy metals (Johnson and Kroening, 1998). Hepatocytes enzymes leakage specially AST and ALT into serum is considered as an indicator of liver damage (Zhang et al., 2002). The results of the present investigation showed that high concentration of copper sulphate significantly elevated the levels of G-PX, SOD, AST and ALT indicating hepatocytes damage. Also, the results of the present investigation indicated that soybean extract significantly compensated the increase in the activity of AST, ALT and alkaline phsophatase. This may indicate a protective activity for soybean flavonoids against copper toxicity.
Also, administration of soybean extract reduced the activities of SOD and ALP enzymes in the blood and lowered the activity of G-PX and MDA content in the liver in comparison with that of the corresponded controls. This may indicate that soybean extract improves liver and kidney functions, in addition to its ability to chelate ions that promote lipid peroxidation.
These results suggest that, the treatment with soybean extracts with copper treatments offered considerable protection to liver as evidenced from the levels of biochemical parameters Levels of AST and ALT that used as an indicators for the chemically induced hepatic injury. The results of the present work are in harmony with those obtained by Vitaglione et al. (2005) who found that flavonoids administration reduces liver injury as shown by the slight increase in plasma AST, ALT and ALP. Glutathione-S-transferase, a phase II enzyme, confers protection against toxic chemicals through metabolizing them into less toxic compounds (Li et al., 1994).
The high copper sulphate doses significantly increased the MDA content (Spatari et al., 2002). This may indicate that the prevention role of the complementary defense system failed to stand against the oxidative damage resulted from free radicals that initiate lipid peroxidation. The obtained data in the present work indicated a considerable antihyperlipidemic activity of soybean supplementation. This may be due to the protective activity of soybean extract against the induced hepatotoxicity that could modulate lipid metabolism and maintain lipid profile within the normal levels.
Necrotizing agents including copper ions produce sufficient injury to hepatic parenchyma and cause disturbance in the level of serum biochemical parameters (Chevion, 1988; Sutton and Winterbourn, 1989). In the present study there was a significantly reduction in the level of MDA compared to other copper treatments. This may indicate that soybean extract probably acts as an antioxidant by preventing peroxidation damage caused by oxidative stress under higher concentration of copper ions. This effect could be also due to the soybean high content of flavonoids that may inhibit lipid peroxidation (Jitoe et al., 1992). At the same time soybean may also possess antioxidant activity that inhibits the deleterious effect of free radicals generated by these ions and thus the membranes rigidity by preventing membrane peroxidation. Oxidative stress, which involves excess accumulation of reactive oxygen species, such as superoxide anion, hydrogen peroxide and hydroxyl radical, can damage cells through lipid peroxidation and alteration of protein and nucleic acid structure (Goldstein and Czapski, 1990).
Nitrite content was increased under the oxidative stress conditions as shown in the present investigation on the other hand, the nitrite levels decreased in response to the administration of soybean extracts. This may be due to the inhibitory effect of soybean flavonoids on the inducible nitric oxide synthetase. This inhibitory activity against nitric oxide formation was probably achieved through scavenging peroxynitrite formation especially in the presence of superoxide anion which is very toxic and responsible for liver and kidney dysfunction.
The results of the present study showed that the administration of copper sulphate induced toxic effects with highest concentrations compared with the negative control. The induced hepatotoxicity under oxidative stress initiated by various copper sulphate concentrations is indicated by the significant increase in the serum ALT, AST, LDH and alkaline phosphatase enzymes. On the other hand, the lipid profile and the lipid peroxidation were indicated via the disturbance in the levels of lipids biochemical parameters represented in the significant increase in the LDL, total lipids and total cholesterol contents under copper toxicity in high doses. High copper doses also significantly affected the kidney function that can be concluded from the elevated content of uric acid (Buege and Aust, 1978) The noticeable influence of copper toxicity was reflected as significant increase in activity of anti-oxidant enzymes (SOD and GPx). The induction of defense enzymes activity did not sufficiently protect the biological systems against copper toxicity especially with high doses. This conclusion is in line with the obtained high serum and liver content of MDA, which indicates lipid peroxidation induction.
On the other hand, the administration of soybean extract did not provoke any toxicity on treated quails Coturnix coturnix. Moreover it offered protective activity against oxidative stress initiated by high doses of copper sulphate.
It is concluded that the complementary implementation of endogenous enzymatically and non-enzymatically defense systems reported to be in need to the administration of such external natural compounds e.g., 100 ppm of soybean extract to work sufficiently under acute oxidative stress conditions to suppress the development of tissue damage associated with the overproduction of reactive oxygen species and maintain the less induced damage.
The author of the present study is very grateful to Dr. Osama Knsoa for his help and support throughout this project. This project was supported by the Research Center, College of Science, King Saud University (Project No. Zoo/2008). The author gratefully acknowledges this support.