ABSTRACT
In this study seasonal effect of Ligula intestinalis of plerocercoids in Alburnus alburnus (bleak) have been investigated. Out of 244 A. alburnus examined and 40 specimens were found to be heavily infected with parasites mentioned above (16%). The highest infection rate has been found in July and August (36 and 44%). In this study among the all fish species A. alburnus was only infected fish by this parasite in Enne Dam Lake. According to ages and seasonally the ratio of infection were calculated.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/ijzr.2006.73.76
URL: https://scialert.net/abstract/?doi=ijzr.2006.73.76
INTRODUCTION
The pathogenic parasite is commonly known much more as a tapeworm. Plerocercoids, large, smooth, unsegmented and with a distinct longitudinal groove, scolex is laterally compressed, it’s not bisected when viewed apically (Cubb et al., 1987). This larval cestode generally is known as an endoparasite in inner cavity of cyprinid fish. The first intermediate hosts are copepods (Cyclops strennus, Acanthocyclops bicusbidatus, A. viridis, Eucyclops serrulatus and Diaptomus gracilis) and definitive hosts are piscivoraus birds (sea-gull, duck, lards, anus, govia, anser) (Bychovskaya and Pavlovskaya, 1962; Markevic, 1951; Mitchell and Hoffman, 1980). Taxonomic categories of Ligula sp., is very new. Before Dubinnina all species of Ligula were known as L. intestinalis, after Dubinnina this parasites distinguished in to four species; these are L. intestinalis, L. colymbi, Digramma interrupta and Digramma nemachili (Dubinina, 1964; Dubinina, 1966).
Also Ligulosis and L. intestinalis are reported from different places in Turkey. The first one is from Egirdir Lake (Güralp, 1968). Distribution of Ligula intestinalis some freshwater fish living on Devegeçidi Dam Lake (Basaran and Kelle, 1976). Ligulosis on some freshwater fishes (Kelle, 1978). Ligulosis in freshwater fish from Elazig region (Cantorary and Ozcan, 1975). Ligulosis in freshwater of Turkey (Keskin and Erk’akan, 1987). Parasites of freshwater fishes in some areas of central Anatolia (Burgu et al., 1988). Some parasites of Barbus capito pectoralis from Keban Dam Lake (Ozdemir and Sarıeyuboglu, 1993). Ligulosis in tenc (Tinca tinca) (Oge and Aydin, 1995). Ligulosis in Porsuk basein-Kutahya (Yilmaz et al., 1996). From Iznik lake (Aydogdu, 1997). Helminthofauna of Manyas Lake fishes (Ozturk, 2000 and Koyun, 2001). And also recorded from several countries; some of them (Sweeting, 1977; Richards, 1981; Hoole and Arme, 1986; Taylor and Hoole, 1989; Arme and Owen, 1968; Dabrowski and Spilewski, 1980). The present study aims, to increase our knowledge about L. intestinalis and its seasonal effects of bleak.
MATERIALS AND METHODS
In the period of one-year survey fish specimens were captured by net from Enne Dam Lake. Samples were being taken at monthly. Fish samples were placed in a plastic tank and transferred to the research laboratory with local lake water, each of them measured, weighed, sexed and dissected. L. intestinalis plerocercoids were collected numbered and recorded all specimens killed and fixed in AFA and later stored in 70% ethanol. The ages of fishes were determined from their scales. Identification of parasite species was done according to Bychovskaya and Pavlovskaya (1962) and fish species according to Geldiay and Balık (1988). Between December and May L. intestinalis has not been seen, so this period was not be evaluated.
The seasonal effect of L. intestinalis, the rate of infection and correlation between length and weight of infected and uninfected fish were calculated.
RESULTS
Out of 244 A. alburnus examined, 40 specimens were found to be infected with L. intestinalis. Throughout a year research parasites have been seen from early June to end of November. In this period infected fish and recovered a number of parasites were given on Table 1. For example number of totally infected fish are 40 and number of totally parasites are 75. So the ratios of infected fish to number of plerocercoids were 23. The percentage of infections were recorded as in June is 27%, in July is 36%, in August is 44%, in September is 14%, in October 11% and in November is 1%. The highest infection was in July and August (Table 1). Of the infected to examine in the two year-old fish is 27%, three year-old fish is 36%, four year-old fish is 21% and five-year old fish is 1% (Table 2).
| Table 1: | The seasonal effects of Ligula intestinalis plerocercoids on A. alburnus |
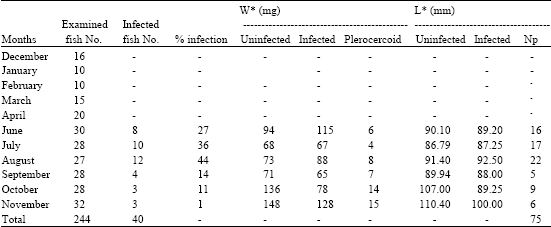 | |
| W* = Weight of fish and plerocercoid, L* = Lenght of fish, Np= Number of plerocercoid | |
| Table 2: | Infection of L. intestinalis plerocercoids according to ages in A. alburnus |
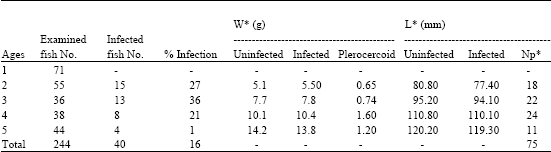 | |
| W* = Weight of fish and plerocercoid, L* = Lenght of fish, Np*= Number of plerocercoid | |
Internal organs of infected fishes are completely destroyed particularly sexual organs are totally spoiled by L. intestinalis plerocercoids. At the end of heavily infected, belly of fish is burst and plerocercoids are released out, the infected fish can be caught with the parasite by piscivarous birds.
DISCUSSION
This study was carried out the effect and seasonal period of Ligula intestinalis in A. Alburnus. L. intestinalis is well known as a belt-worm in common. This was the first study in this area, but Ligulosis or L. intestinalis were recorded from different parts of Turkey.
The important thing is that for this area L. intestinalis could not be found in fish, live in this lake but A. alburnus. Property of this subjecst should be studied in many respects, especially in food conceptions of fishes of Enne Dam Lake.
The highest infection is in July-August (36 and 44%). During the spring in June when the water is getting warmer, the plerocercoid will come into view from June until late October. Parasite specimens were recorded rare in November, but later they could not be seen. The ratio of infection is 23% in all fish population, which originate serious problem in this lake.
Statistically comparing of infected and uninfected fish, there is positive correlation among two years old fishes p< 0.01. Having infection fish, when we evaluating them monthly and yearly two years and three years old fishes can grow more then the others. It has been shown that plerocercoids are high, according to weight and numbers compare to the four years old fishes (Table 2).
Infected fish moves slowly during the heavily infection at the late summer, so they can be caught by piscivarous birds. Parasite may grow so large, that they rupture the belly of the second intermediate host and are left free in the water (Mitchell and Hoffman, 1980). Then free worms are eaten by certain birds soon after they are released and the cycle continuous as it normally does in the final host. As a result we can say that the controlling of Ligulosis is rather difficult or impossible because of piscivarous birds which definitive host (Yilmaz et al., 1996).
REFERENCES
- Arme, C. and R.W. Owen, 1968. Occurrence and pathology of Ligula intestinalis infections in british fishes. J. Parasitol., 54: 272-280.
Direct Link








