Research Article
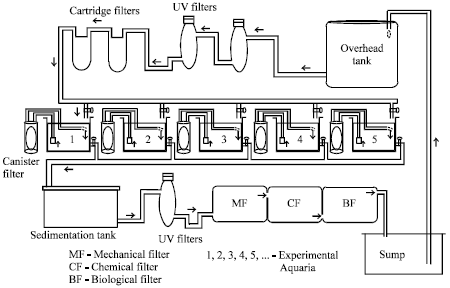
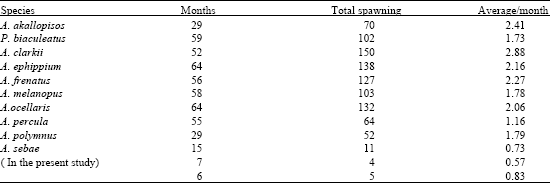
Studies on Spawning in Clownfish Amphiprion sebae with VariousFeed Combinations under Recirculating Aquarium Conditions
Marine Research Aquarium, CAS in Marine Biology, Annamalai Nagar-608 (102, India
K. Raja
Marine Research Aquarium, CAS in Marine Biology, Annamalai Nagar-608 (102, India
T. Balasubramanian
Marine Research Aquarium, CAS in Marine Biology, Annamalai Nagar-608 (102, India