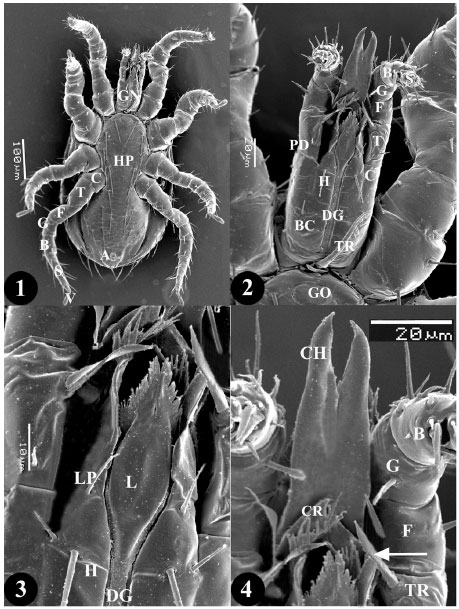
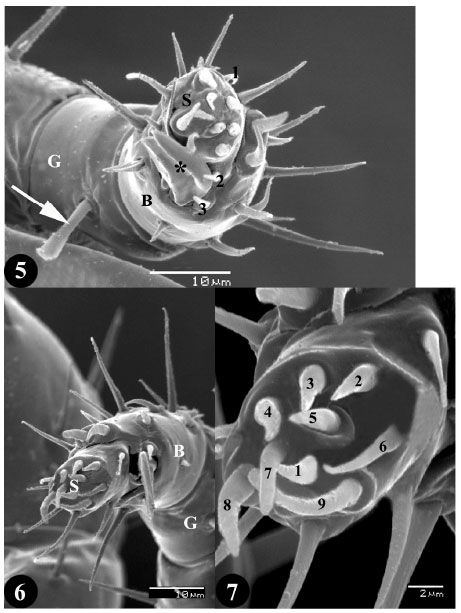
Research Article
Scanning Electron Microscopic Observations on Male Echinolaelaps echidninus (Berlese, 1887) (Dermanyssoidea: Laelapidae) with Emphasis on its Gnathosoma, Palpal Organ and Pulvilli
Department of Zoology, Faculty of Science, Ain Shams University, Cairo, Egypt