Research Article
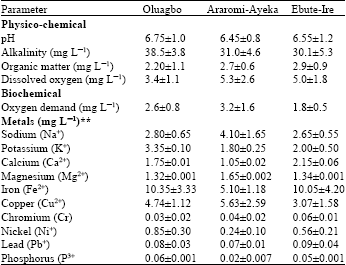
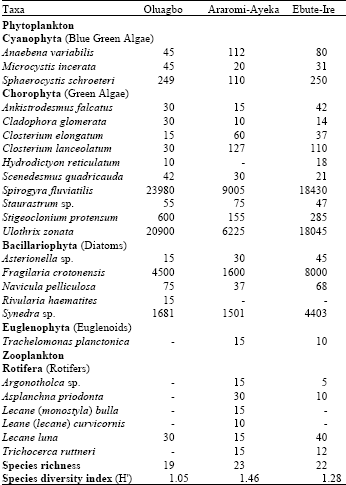
Water and Planktonic Quality of a Palm Oil Effluent Impacted River in Ondo State, Nigeria
Department of Zoology, Obafemi Awolowo University, Ile-lfe, Nigeria
A. A. Adedeji
Department of Zoology, Obafemi Awolowo University, Ile-lfe, Nigeria