Research Article
Effects of Captivity on Basal Metabolic Rate and Body Composition in Sanderling Bird Calidris alba
Department of Zoology, College of Science, King Saud University, P.O. Box 2455, Riyadh 11451, Saudi Arabia
Variation in basal metabolic rate has been reported to occur within individual captive shorebirds during their annual cycle[1-3]. There have been conflicting reports on effects of captivity on BMR when comparing captive individuals with wild conspecifics. Weathers et al.[4] reported that the BMR of four captive and four wild Apapanes bird Himatione sanguinea held in captivity for a period of one year did not differ significantly from those of freshly caught individuals. However, Warkentin and West[5] found out that Merlins Falco columbarius held in captivity for periods ranging from seven months to three years had significantly higher BMR and body temperatures than freshly caught birds. In their study however, only four captive birds of mixed sex and differing in age were used and they were compared with nine wild conspecifics. None of the captive birds were able to fly, due to wing fractures. Lean-mass-specific BMR increased significantly in long-term captive Knot between year one and year two[6]. However, long-term captive Knot had lower BMRs than those of wild conspecifics[7,8]. This was claimed to be due primarily to a decrease in mass of the digestive organs. The metabolic intensity of lean tissues is known to decrease with age[9], which may be another possible or additional mechanism involved in the decrease seen in BMR in captivity.
Many studies have also shown changes in body composition in captive birds when compared with wild conspecifics[6,10-12]. Scott et al.[10] and Mitchell[11], reported that there was a reduction in the digestive organ mass in captive Redshank when compared with wild conspecifics. Moreover, Piersma et al.[2] and Selman[6], have also shown a reduction in lean mass in captive Knot.
In this investigation, the effects of captivity on BMR and body composition in Sanderling have been studied primarily by comparing BMR of year one and year two of captive Sanderling of the same species and of similar mass, in conjunction with measurements of body composition and masses of various organs (liver, heart, gut and intestine length) of wild and captive conspecifics.
MATERIALS AND METHODS
Comparison of body composition of wild and captive Sanderling: Fifteen wild and seventeen captive Sanderling were killed under licence and dissected in order to do carcass analysis. Captive birds were held in captivity between September 1997 and June 1999. The 15 wild Sanderling were collected after they had been killed accidentally during cannon-netting at Teesmouth, north-east England and on the Severn estuary. All were sealed in plastic bags and frozen at -20°C until carcass analysis was carried out at later date.
The heart, liver, stomachs, intestines and pectoral muscle blocks (left pectoralis major and left supra-coracoideus) were dissected out. All organs were dried to constant dry mass at 40°C in a vacuum oven and then weighed to the nearest mg on a torsion balance. The bird was sexed by gonadal inspection, the gut length measured and four skeletal measurements were taken to the nearest 0.1 mm using vernier callipers, following the methods of Piersma et al.[13], in order to calculate a standard muscle volume SMV[14]. The mass of one lean dry pectoral muscle block was then expressed as a proportion of the SMV to produce an index of muscle size, independent of total body (skeletal) size, known as SMI or standard muscle index. Stored lipids (triglycerides) were extracted from the carcass and the dissected organs using a Soxhlet extractor with petroleum ether as a solvent. The carcass and organs were subsequently dried once again using constant mass at 40°C in a vacuum oven and the fat-free masses were then obtained.
Measurement of basal metabolic rate
Open-flow respirometry: The metabolic heat production of each bird was estimated by determination of rates of oxygen consumption using a paramagnetic oxygen analyser (Servomex plc, Crowborough, East Sussex, Model1111D/000) and carbon dioxide production using an infrared analyser (Lira 3000, Mine Safety Appliances Company, Pennsylvania, USA) in an open-circuit system described by Scott et al.[15]. For measuring BMR, each bird was removed from the aviary at 08:00 GMT and kept isolated in a box for a minimum of 1 h without food or water. Around 10:00 GMT the bird was weighed using a Pesola spring-balance to the nearest g and then its TOBEC measurement taken. The bird was placed in one of the two identical metabolic chambers measuring 24.5 cm (height) x 21cm (diameter).
BMR measurement started at or after 12:00 h, after a period of at least 2 h of acclimation by birds to the metabolic chamber and 4 h of fasting to ensure that the individual was post-absorptive. The chamber was in complete darkness and was placed in a controlled temperature cabinet (LMS, Sevenoaks, Kent) at a constant temperature of 25°C (within the thermoneutoral zone of Sanderling). Dry air was drawn through the chamber at a rate of 60 L h-1. Gas analyses were performed on samples taken from both the inlet and outlet gases via gas mass-flow controllers (Brooks Instruments, Netherlands, 5878 and 5850 TR series) at rates of 3.6 L h-1 for O2 and 4.8 L h-1 for CO2. Both inlet and outlet gases were dried prior to measurement by passing them over columns of dried coarse mesh silica gel. A measurement of BMR on a single individual was taken over a period of time ranging from between 90-120 min to ensure that a stable measurement of BMR was obtained for each run. If any periods of raised Metabolic Rate (MR) occurred during a measurement these were assumed to be periods of activity and the measurements were not used, although this was rare (Author, pers. Obs.). Under normal circumstances, birds remained at rest throughout the measurements, as proven by the lack of traces on activity-recording paper used to line the floor of the chamber. A period of 15-20 min was necessary between the first and second individual’s BMR measurements when measurements were carried out on two birds on the same day, to enable the levels of O2 and CO2 to return to the pre-measurement baseline levels. To avoid CO2 building up in the metabolic chamber not undergoing a BMR measurement, a constant flow of dry air was provided through this metabolic chamber using a simple diaphragm pump. Calibration was performed before each day’s measurements, using dry, oil-free 100% N2 and then a certified mixture of 21% O2, 0.03% CO2 in N2 (SIP Analytical Ltd.). During each analysis, measurements were taken every minute over a ten-minute period in which O2 consumption and CO2 production were appeared to be stabilised at a basal level. The means of O2 and CO2 levels over these ten-minute periods were used to calculate RQ (CO2 production/O2 consumption) and BMR expressed in W (using an energy value per litre O2 consumed appropriate for the RQ). The levels of O2 and CO2 were recorded onto a flatbed recorder (Kipp and Zonen, Delft, Netherlands, Model BD 112). Measurements of BMR were taken at least once every week.
Measurement of Total Body Electrical Conductivity (TOBEC) indices: Seventeen adult Sanderling, caught at Teesmouth (Northeast England) using cannon nets and mist nets and held in captivity thereafter, were used for studies of seasonal variation in body composition and basal metabolic rate and then used to find out the effects of captivity on basal metabolic rate and body composition in Sanderling.
Measurement of TOBEC is now a popular technique among ornithologists for estimating fat reserves in birds[16]. This method was developed in the early 1970’s and has recently been used widely in the agricultural and medical fields[17]. The machine must be calibrated for each species to be studied.
Predicted Total Lean Mass (PTLM) for Sanderling can be obtained using the equation below
PTLM = (0.49 * I) + 20.4 g (calibrated by the Author) for Sanderling Calidris alba.
Predicted Mass of Fat (PFM) is calculated by subtracting PTLM from Body Mass (BM);
PFM = BM – PTLM
TOBEC was measured using the EM-Scan (3420 Constitution Drive, Spring-field, Illinois 62707, USA) SA-1 Small Animal Body Composition Analyzer. The SA-1 was used in the laboratory and also in the field when it was powered by a 12 V battery via an Oertling PC-01 converter to provide 240 V, 50 cycles AC. Each of the captive birds was weighed using a Pesola spring-balance to the nearest g and its TOBEC index calculated at least once every week. Captive birds were isolated from the cage for 1 h before measuring TOBEC values.
To determine whether changes occurred in the BMR of an individual Sanderling during its time in captivity, when controlling for body mass, a comparison of mass-specific BMR and body composition predicted by TOBEC in individual Sanderling were compared between year one and year two of captivity was carried out with the results shown in Table 1. All BMR measurements taken were outside the pre-migratory period and did not include measurements taken during moult because the mean levels calculated for each year only included measurements taken in comparable months of year one and two (January-March). It can be seen that body mass deceased significantly in all captive Sanderling between year one and year two, except in the individuals AF (was not significant) and AO (decrease). PTLM also deceased significantly in all captive Sanderling between year one and two, except in the individuals AA, AB, AE and AF the decreases were not significant. However, the individual AO was the only increase in predicted lean mass seen in all captive Sanderling. PFAT was slightly more variable between years, with Sanderling AI showing a significant decrease in fat mass between year one and year two.
Mass-specific BMR decreased significantly in nearly all captive Sanderling between year one and year two. However, the individual AO was the only increase in mass-specific BMR and AB, AH remained stable between year one and year two.
Table 2 shows the comparative difference between fifteen wild and seventeen captive Sanderling in certain lean mass components. The mean liver mass was 39% lower in captive birds and the mean gut mass (stomach + intestine), had decreased by 55%. The mean length of the intestine and heart mass had also decreased by 30% in captive birds when compared to wild conspecifics. However, there was no significant difference between captive and wild Sanderling in the mean values of Standard Muscle Index (SMI).
Effects of captivity on BMR and body composition: The effect of captivity on BMR has been studied primarily by comparing wild and captive of the same species and of similar mass. In some, no difference has been reported[4,18]; in others, captives had an increased BMR[5,6] and yet others, captives had a decreased BMR compared to wild conspecifics[8]. It would appear from the literature that no study, has looked intensively[1,6] at how BMR changes within an individual bird over time in captivity.
Table 1 shows that BMR, hence average metabolic rate per gram of mass tissues (mass-specific BMR) in Sanderling, brought into captivity decreased significantly between the first year and second year of captivity. This decreased could be explained by the reduction of metabolically active lean tissues such as kidney, liver, heart and intestines (Table 2). It is not known whether the captive Merlins in Warkentn and West's[5] were undergoing moult or not during the period of measurements as the plumage state affect the thermal conductance and hence body temperature. They claimed that because of the significant atrophy of the pectoral muscle mass that had taken place in captivity, captive Merlines had higher body temperatures and BMR than wild conspecifics, although they did not measure this and total body masses were similar in captive and wild birds. They argued that muscle atrophy in captives had been compensated by growth of other components of highly metabolically active organs such as liver, leading to higher body temperature and BMR. This loss was probably due more to the disuse atrophy, as in their study, none of the captive birds were able to fly, due to wing fractures and data from only four captive birds out of six were compared with those from nine wild conspecifics. Selman[6] claimed that the increases in BMR that occurred in his captive Knots were due to the metabolic intensity of the lean tissues change with season rather than masses of metabolically active lean tissues[8,19]. However, Selman[6] did not actually measure the intensity of the lean tissues throughout annual cycle, but by showing that seasonal peaks in BMR in captive Knot did not coincide with peaks in body mass or predicted lean mass. It can be seen from this study that BMRs decreased significantly between year one and two in captivity and that could be due mainly to the decreases seen in mass of the digestive organs of captive Sanderling.
Effects of captivity on organ masses: The composition of organ masses between captive and wild Sanderling shows a significant reduction in organ masses in captive Sanderling. This result supports that of Piersma[7], Mitchell[11], Selman[6] and Almansour[12]. This shows that captive shorebirds are able to exhibit considerable flexibility in lean tissue in the face of altered living condition. The significant reduction in gut mass (stomach mass+ intestine mass) seen in this study is probably due to disuse atrophy, because captive Sanderling in this study fed on soft food pellets and therefore do not need a muscular gizzard to break up hard food[20]. The food in captivity is also less fibrous than that eaten in the wild and this reduction in the fiber content has been shown to lead to a decrease in gizzard size[21]. The significant decrease in intestine length in this study, Mitchell[11] and Selman[6] is likely also to be due to disuse atrophy.
| Table 1: | Changes in body mass, body composition and mass-specific BMR (Watts/kg) in 17 adult Sanderling between year one and year two of captivity. |
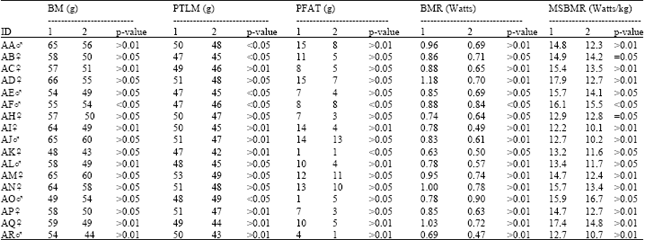 | |
| *All BMR measurements were taken outside the spring migratory period and do not include periods of moult. 1= Year one in captivity 2= Year two in captivity | |
| Table 2: | Comparisons of dry lean organ mass and gut length between 15 wild and 17 captive Sanderling. Values are means with standard errors in parentheses. |
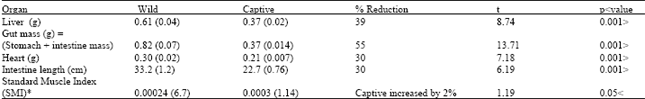 | |
| *SMI: Mass of left lean dry pectoral muscle mass/ standard muscle volume % Reduction: indicates the difference between mean organ mass of captive and wild Sanderling, as % of wild Sanderling mass | |
The assimilation through the gut of artificial, soft food pellets is probably easier and quicker than that of hard prey in the wild and this in turn causes a reduction in the surface area needed to adequately absorb food. The reason for the significant reduction in the mass of liver in captivity is more difficult to explain satisfactorily. However, this reduction could be due to the consistent food been taken by the captive birds comparing to the variety food available to the wild ones. More investigations need to be conducted on changes in enzyme activity during periods of captivity in birds.
The author of this paper is very grateful to the group of Lab 16 of Durham University for their help and support throughout this project.