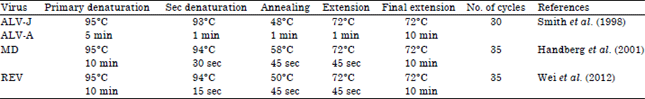
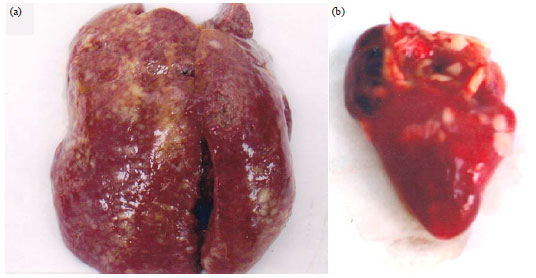
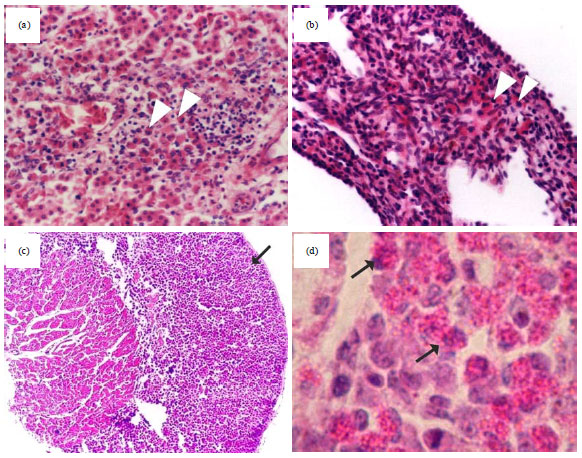
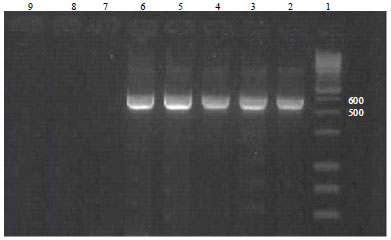
Short Communication
Detection of Avian Leukosis Virus Subgroup J from Commercial Peking Duck Breeder Farm in Egypt
National Laboratory for Veterinary Quality Control on Poultry Production (NLQP), Animal Health Research Institute, P.O. Box 264, Giza, Egypt
Mohamed A. Soliman
National Laboratory for Veterinary Quality Control on Poultry Production (NLQP), Animal Health Research Institute, P.O. Box 264, Giza, Egypt
Marwa A. Safwat
National Laboratory for Veterinary Quality Control on Poultry Production (NLQP), Animal Health Research Institute, P.O. Box 264, Giza, Egypt
Osama Mehana
National Laboratory for Veterinary Quality Control on Poultry Production (NLQP), Animal Health Research Institute, P.O. Box 264, Giza, Egypt
Marwa A. El- Magid
National Laboratory for Veterinary Quality Control on Poultry Production (NLQP), Animal Health Research Institute, P.O. Box 264, Giza, Egypt
Mohamed K. Hassan
National Laboratory for Veterinary Quality Control on Poultry Production (NLQP), Animal Health Research Institute, P.O. Box 264, Giza, Egypt
Soad A. Nasif
National Laboratory for Veterinary Quality Control on Poultry Production (NLQP), Animal Health Research Institute, P.O. Box 264, Giza, Egypt