ABSTRACT
Potato leaf roll virus (PLRV) is a serious disease of potato causing decline in potato production worldwide. Symptoms of PLRV like leafroll and stunting were observed during survey in potato fields of Punjab. Tubers and leaves of symptomatic potato plants were indexed for PLRV using test plants, DAS-ELISA and RT-PCR. Seedlings of Phaseolus floridana and Datura stramonium were used as test plants and they showed stunted growth and chlorotic spots after 10-12 days of inoculation feeding by aphid vector (Myzus persiace). P. floridana developed more visible symptoms of PLRV infection as compared to D. stramonium. Sample collected during survey from potato growing areas and test plants showing symptoms of virus after aphid transmission were indexed by DAS-ELISA. All test plants showing symptoms of virus and 78.57% of leaves and 74.41% of tuber samples colleted during survey were detected positive for virus in this serological method. The RT-PCR assay of ELISA positive samples yielded amplification of 336 bp corresponding to partial coat protein gene of PLRV. The present study reports the prevalence of PLRV in potato growing areas of Punjab.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijv.2014.272.279
URL: https://scialert.net/abstract/?doi=ijv.2014.272.279
INTRODUCTION
Potato (Solanum tuberosum L.) is one of the important vegetable and fourth most important food crops in India (Saxena and Mathur, 2013). India ranks second in total world potato production after China (Guenthner, 2010). Punjab is the third largest potato growing state in India and it has contributed considerably to the development of Indian potato processing industry (Rana, 2011). Potato is widely cultivated in two to three crops per year in Punjab because of suitable agroclimatic conditions. It is vegetatively propagated by using seed tubers and saving of so-called seed tubers transmit diseases to the next season crop and cause a progressive decline in potato production (Tiwari et al., 2012). Potato is vulnerable to wide range of viral pathogens and most of the viruses move easily and survives in tubers and reduces yield and quality of next season crop (Halterman et al., 2012).
Potato leaf roll virus (PLRV) is one of the most important and damaging viral diseases of potato distributed widely in potato-growing areas of different parts of the world (Gillen and Novy, 2007). PLRV is a single-stranded positive-sense RNA virus which belongs to the genus Polerovirus of family Luteoviridae (Mayo et al., 1989). The PLRV is circulative, non-propagative virus transmitted exclusively by aphid and is capable of causing severe yield and quality losses in potato (Flanders et al., 1990). Banttari et al. (1993) reported that planting of PLRV infected seed tubers can cause yield loss of 80%. Infection of PLRV results into small size and less number of marketable tubers per plant (Hamm and Hane, 1999). The PLRV also causes tuber necrosis in susceptible potato cultivars and these symptoms can further intensify during prolonged storage and thus limits marketability (Manzer et al., 1982).
Characteristic symptoms of PLRV like rolling of leaves, chlorosis and stunting were commonly observed on potato plants in fields of Punjab. Damage from PLRV is one of the important reasons for variability in yield and quality of potatoes in Punjab. Kang et al. (2009) surveyed potato growing areas in different districts of the Punjab and identified symptoms of PLRV on potato plants. Study on indexing of PLRV from potato growing areas of Punjab is required to develop strategies for management of this virus. In present study, survey was conducted in potato fields of Punjab and incidence of PLRV was reported using test plants, ELISA and RT-PCR method. Routine screening of seed tubers by different virus indexing methods and control of aphids is required to prevent further spread of this virus in potato growing areas of Punjab.
MATERIALS AND METHODS
Survey and sample collection: Survey was conducted in potato growing areas of Amritsar, Punjab, India during growing season from October 2012 to February 2014 to study status of PLRV in this region of the country. Plants showing symptoms of PLRV such as leaf roll and stunting were selected and surveyed regularly during growth and harvesting stage to study effect of symptoms on plants and tuber growth. Tubers and leaf samples were collected from these selected plants for indexing of virus.
Virus transmission test: The PLRV can not be transmitted mechanically by sap inoculation so aphid vector, Myzus persiace (Sulzer), was used in transmission of this virus on test plants as described by McCarthy (1954). Myzus persiace is the most important aphid transmission vector of potato viruses and was common in potato growing areas of Punjab. Seedlings of Datura stramonium and Physalis floridana were raised in pots in net house as test plants to study aphid transmission. Brassica juncea (Indian mustard) seedlings were also raised in pots to maintain stock colonies of aphid as an immune host plant. Wingless adult aphids were collected and transferred to leaves of mustard plant and were allowed to develop colonies in insect free cage. Nymphs from these leaves were then transferred on healthy young plantlets of mustard to maintain virus-free colonies of aphids in separate cage. Non-infective aphids were given an acquisition access of 2 days on potato samples showing symptoms of virus. After acquisition feeding, eight aphids were then placed on seedlings of each indicator plant (at 2-3 leaf stage) in cage and were labeled. The aphids were touched with brush before few moments they were lifted, in order to allow them to withdraw their stylets. After infection access of 3 days, the test plants were fumigated with aphicide to kill the aphids. Ten indicator plants of each species were labeled as control and were fed with non-viruliferous aphids maintained on mustard plant. Each experiment consists of 12 replicates and was repeated thrice and observations on appearance of visual symptoms on the indicator plants were recorded daily.
Double Antibody Sandwich-ELISA (DAS-ELISA): The DAS-ELISA was performed on sap extracted from (i) Leaves and tubers of potato plants showing symptoms of PLRV and (ii) Leaves of indicator plant showing symptoms after aphid transmission. Anti PLRV IgGs, enzyme conjugate, positive and negative controls of Agdia, Inc (Elkhart, Indiana) were used in the present study. Leaf and tuber tissue were grounded and extracted at a sample to buffer ratio of 1:10 in Agdia’s General Extract Buffer (GEB 1X) using mortar and pestle. The detailed method as described by Clark and Adams (1977) was followed for DAS-ELISA. Ninety wells were used on each plate for sample analysis and remaining six wells were used for positive and negative controls. Absorbance values were read at 405 nm on Multiskan EX plate reader (Thermo Fisher Scientific, Vantaa, Finland). The reaction was considered positive only when the mean absorbance value was more than two times of negative control.
RT-PCR: Total RNA was extracted from frozen leaf and tuber samples using TRIzol reagent (Invitrogen, USA). Each sample was homogenized in liquid nitrogen using pre-chilled pestle and mortar. About 100 mg of ground sample was taken in 2 mL eppendorf tube and vortexed with 1 mL of TRIzol reagent. The homogenate was centrifuged in microcentrifuge (Eppendorf, Hamburg, Germany) at 12,000 rpm for 10 min to remove insoluble material. About 200 μL of chloroform was added to supernatant and the mixture was vortexed vigorously for 15 sec and then allowed to stand for 10 min at room temperature. The resulting mixture was centrifuged at 12,000 rpm for 15 min. Upper aqueous phase was taken in a fresh 2 mL eppendorf tube and 500 μL of isopropanol was added and mixed properly. The mixture was incubated at room temperature for 10 min and centrifuged at 12,500 rpm for 20 min. The RNA pellet was washed with 75% ethanol by centrifuging it at 12,500 rpm for 5 min. Pellet was air dried for 5-10 min and dissolved in 50 μL of RNase and DNase free sterile water (Sigma Aldrich, USA) at room temperature for 10-15 min. All the centrifugation steps were performed at 4°C. RNA was quantified and stored at -20°C for further use.
Reverse Transcription (RT) reaction was carried out in a total reaction volume of 50 μL in 0.2 mL thin walled tube using following reaction components: 5X reverse transcriptase buffer, 10 mM of dNTPs mix, 25 units of RNase inhibitor, 200 units of M-MLV reverse transcriptase (G-Biosciences, India), 200 ng down stream primer and 10 μL total RNA as template. The RT reaction was carried out at 37°C for 1.15 h followed by incubation at 70°C for 5 min and then immediate transfer to ice.
The upstream (5'CGCGCTAACAGAGTTCAGCC3') and downstream (5'GCAATGGGGGTCCAACTCAT3') primers for PLRV were designed and used to yield amplification of 336 base pairs in RT-PCR assay (Singh et al., 1995). Complementary DNA (cDNA) produced in RT reaction was amplified using following reaction mixture: 5 μL of 10X Taq polymerase buffer, 10 μL cDNA, 1 μL (0.2 μg) of each upstream (U) and downstream (D) primer, 0.5 μL (5 units μL-1) of Taq DNA polymerase enzyme (G-Biosciences, India), 1.5 μL of 10 mM dNTP mix. The contents were thoroughly mixed after making final reaction volume of 50 μL with RNase and DNase free sterile water (Sigma Aldrich, USA). Reaction mixture was incubated in a gradient thermal cycler (Geno Technology, USA) in 0.2 mL thin walled PCR tube. Cycling conditions were optimized by initially heating reaction mixture at 92°C for 5 min and then 28 cycles of amplification with each consisting of denaturation at 92°C for 1 min, annealing at 59°C for 1 min and extension at 72°C followed by final extension at 72°C for 10 min. About 10 μL of PCR product were electrophoresed in a 1.5% agarose gel containing 0.5 μg mL-1 ethidium bromide and visualized and photographed under UV illumination.
RESULTS AND DISCUSSION
Survey: Symptoms of PLRV such as inward rolling of upper leaves with upright growth habit were observed in potato fields of Punjab. Rolled leaves become thickened and brittle with leathery texture and such plants when shaken produced rustling noise (Fig. 1a).
 | |
| Fig. 1(a-c): | Potato plant showing symptoms of PLRV in potato growing areas of Punjab, (a) Inward rolling of leaflets with stunted growth, (b) Rolling along with purple discoloration of affected leaflets and (c) Aphids on lower side of potato leaves |
Leaves of some plants become chlorotic and showed purple discoloration (Fig. 1b). Reductions in number and size of tubers were also observed in severely infected plants. Earlier, Hamm and Hane (1999) also reported that PLRV-infected plants produce fewer and smaller size tubers than healthy plants. Such small size infected tubers are more likely to be selected as seed tubers for next season crop. They produce more sprouts per unit weight as compared to large size tubers and are mainly used by farmers. This might be the possible reason for prevalence of PLRV in Punjab. Njukeng et al. (2013) reported that potato viruses are more prevalent in small size tubers and least in large size potato tubers. They further suggested that tuber size can serve as a guide to identify healthy tubers and farmers should always use large size potato tubers for planting.
Aphids might be playing a key role in transmission and spread of PLRV in Punjab during the months of October and February because of warm climatic conditions (18-24°C). It was observed during survey that foliage symptoms of both primary and secondary infection of PLRV were common in early and late season potato crop due to warm agro-climatic conditions and high aphid population (Fig. 1c). Syller (1994) reported that Myzus persiace is more efficient in acquisition and transmission of PLRV while feeding at elevated temperatures. Warm humid conditions (18-24°C) are favorable for aphid survival and development of colonies along with movement of aphid populations. Syller (1996) reported high rate of PLRV transmission with rise in temperature in potato growing regions due to greater amount of virus accumulation in aphids fed at warm temperatures. It was also found during survey that potato growers were not applying regular aphicide sprays in early and late season crop resulting into rapid spread of this virus. Control of aphid population is very important in order to minimize the spread of PLRV.
Aphid transmission study: Aphid transmission is the most widely used approach in indexing of viruses because specialized skills and equipments are not required to perform the experiments. The technique involves the use of aphids to transfer the virus to the indicator plant which then develops visible symptoms.
 | |
| Fig. 2: | Chlorotic spots on leaf of Physalis floridiana after 10 days of inoculation feeding |
Physalis floridana and Datura stramonium were used in present study as they are known to be the most suitable indicator plants for indexing of PLRV (Syller, 1996). As PLRV is localized in the phloem of infected plants, acquisition feeding of 2 days and inoculation feeding of 3 days were used for efficient transmission of virus to test plant. Seedlings of P. floridana showed chlorotic spots on leaves, epinasty and stunted growth after 10 days of inoculation feeding (Fig. 2). Chlorotic spots on leaves and stunted upright growth were also observed after 12 days of virus transfer by viruliferous aphids on D. stramonium. About 75% of P. floridana and 66.6% of D. stramonium plants developed symptoms of chlorosis and showed stunted growth after 15 days of virus transmission and no such symptoms were observed on control seedlings fed with non-viruliferous aphids. Among two test plant, P. floridana developed more visible symptoms of PLRV infection as compared to D. stramonium. Earlier, Kotzampigikis et al. (2010) have also reported superiority of P. floridana as test plant to detect PLRV by vector transmission. Dhawan and Rishi (1990) have successfully isolated and purified PLRV by aphid transmission using P. floridiana as indicator plant. They have also used P. floridiana as differential host for identification of different strains of PLRV in northern India (Dhawan and Rishi, 1992).
DAS-ELISA: Direct double antibody sandwich format ELISA in microtitre plates as described by Clark and Adams (1977) was used. It is the most reliable and common serological method for routine diagnosis of plant viruses in large number of samples in a relatively short period of time. The advantage of this assay is that the only virus particles are concentrated from plant extracts by the specific antibody coated in wells and other components are removed by rinsing before addition of detecting antibody and enzyme substrate (Khan et al., 2003). In this study, 112 samples of potato leaves with leafroll symptoms and 43 samples of tubers from potato plants showing both primary and secondary foliage symptoms of PLRV were collected and indexed by DAS-ELISA. Fifteen samples of each indicator plants after 20 days of aphid transmission were also analyzed in the present study to confirm the test results. The mean absorbance values observed at 405 nm for negative and positive controls were 0.027 and 0.432, respectively. The absorbance values for 57 potato leaf samples ranged between 0.319-0.463 and 31 samples showed absorbance value in the range of 0.210-0.298 and all these were considered ELISA positive. Twenty four leaf samples were considered negative in ELISA as they showed absorbance in the range of 0.28-0.76. Twenty five out of 43 tuber samples showed absorbance in the range of 0.220-0.300 and 7 samples showed absorbance in between 0.109-0.187.
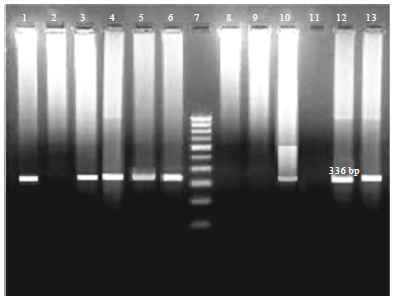 | |
| Fig. 3: | Agarose gel electrophoresis of RT-PCR products of PLRV. Lanes 1 and 2: Positive and Negative controls respectively, Lanes 3-4: Leaf samples from potato plant detected positive in DAS-ELISA, Lane 5 and 6: Leaf samples of Datura stramonium and Physalis floridiana showing symptoms of PLRV after 15 days of vector transmission, Lane 7: 100 bp DNA ladder, Lane 8-9: Leaf sample from control plants of Datura stramonium and Physalis floridiana fed with non-viruliferous aphids, Lane 10-13: Tuber samples |
Eleven tuber samples were observed negative and all indicator plants showing symptoms of virus after transmission test were detected positive for virus in this serological study. The DAS-ELISA produced good and reproducible results in our study under standardized working conditions with highly specific antiserum supplied by Agdia Inc. Among the various possible ways to detect plant viruses, this serological detection is frequently favored because of its suitability for large scale indexing with specificity, speed and scope provided for standardization (Halterman et al., 2012).
RT-PCR: RT-PCR is an effective and efficient tool for in vitro amplification of specific region of a genome by selecting a pair of primers for that region. It requires highly purified good quality total RNA (viral and plant RNA) from infected tissues. The RNA isolated from leaves and tuber samples of plants showing symptoms of PLRV were subjected to RT-PCR assay to further confirm the presence/absence of virus. The RNA was quantified and checked for purity by A260/A280 ratio and further confirmed by separation on agarose gel before RT reaction. Total 18 ELISA positive samples were indexed by this technique which includes 10 potato leaf samples, 4 indicator plant leaf samples and 4 potato tuber samples. All ELISA positive samples yielded amplification of 336 bp corresponding to coat protein gene of PLRV in RT-PCR assay (Fig. 3). The RT-PCR is highly sensitive and reliable technique for the detection of plant viruses at very low concentrations in infected tissues (Mortimer-Jones et al., 2009; Sharma et al., 2009). The procedure is nucleotide sequence specific, extremely sensitive, fairly inexpensive and requires minimal skills to perform experiment (Matsushita et al., 2010). It has been commonly applied for the detection of PLRV in leaves and dormant tuber samples (Mortimer-Jones et al., 2009; Awan et al., 2010).
CONCLUSION
Prevalence of PLRV in Punjab was reported in the present study by using virus transmission test, ELISA and RT-PCR assay. PLRV is known to cause serious loss in yield and quality of potatoes in this region. Use of infected seed tubers and presence of aphid vectors is responsible for incidence and spread of PLRV in potato growing areas of Punjab. Survey and indexing of PLRV using different virus diagnostic methods is highly important to know the current status of this virus and to develop strategies for its control. Routine screening of seed tubers by different virus indexing methods and use of certified virus-free tubers is required to control spread of this virus in potato growing areas of Punjab.
ACKNOWLEDGMENT
Author is grateful to Science and Engineering Research Board (SERB), Government of India, New Delhi for providing financial assistance in the form of Research Project (Project No. SR/FT/LS-14/2010).
REFERENCES
- Awan, A.R., I.U. Haq, M.E. Babar and I.A. Nasir, 2010. Molecular detection of Potato leaf roll polerovirus through reverse transcription polymerase chain reaction in dormant potato tubers. Pak. J. Bot., 42: 3299-3306.
Direct Link - Clark, M.F. and A.N. Adams, 1977. Characteristics of the microplate method of enzyme-linked immunosorbent assay for the detection of plant viruses. J. Gen. Virol., 34: 475-483.
CrossRefPubMedDirect Link - Dhawan, P. and N. Rishi, 1990. Isolation and purification of potato leafroll virus in India. Indian J. Virol., 6: 38-45.
Direct Link - Flanders, K.L., D.W. Ragsdale and E.B. Radcliffe, 1990. Use of enzyme-linked immunosorbent assay to detect potato leafroll virus in field grown potato, cv. Russet Burbank. Am. Potato J., 67: 589-602.
CrossRef - Gillen, A.M. and R.G. Novy, 2007. Molecular characterization of the progeny of Solanum etuberosum identifies a genomic region associated with resistance to potato leafroll virus. Euphytica, 155: 403-415.
CrossRef - Halterman, D., A. Charkowski and J. Verchot, 2012. Potato, viruses and seed certification in the USA to provide healthy propagated tubers. Pest Technol., 6: 1-14.
Direct Link - Hamm, P.B. and D.C. Hane, 1999. Effects of seedborne potato leafroll virus on Russet Norkotah potato. Plant Dis., 83: 1122-1124.
CrossRef - Kang, S.S., A. Kumar and S.K. Hundal, 2009. Characterization and identification of viruses prevalent on potato in Punjab. Plant Dis. Res., 24: 175-177.
Direct Link - Khan, M.S., M.I. Hoque, R.H. Sarker and H.P. Muehlbach, 2003. Detection of important plant viruses in in vitro regenerated potato plants by double antibody sandwich method of ELISA. Plant Tissue Cult., 13: 21-29.
Direct Link - Kotzampigikis, A., D. Hristova and E. Tasheva-Terzieva, 2010. Virus-vector relationship between potato leafroll virus PLRV and Myzus persicae Sulzer. Bulgarian J. Agric. Sci., 16: 412-421.
Direct Link - Manzer, F.E., D.C. Merriam, R.H. Storch and G.W. Simpson, 1982. Effect of time of inoculation with potato leafroll virus on development of net necrosis and stem-end browning in potato tubers. Am. Potato J., 59: 337-349.
CrossRef - Matsushita, Y., T. Usugi and S. Tsuda, 2010. Development of a multiplex RT-PCR detection and identification system for Potato spindle tuber viroid and Tomato chlorotic dwarf viroid. Eur. J. Plant Pathol., 128: 165-170.
CrossRef - Mayo, M., D.J. Robinson, C.A. Jolly and L. Hyman, 1989. Nucleotide sequence of potato leafroll luteovirus RNA. J. Gen. Virol., 70: 1037-1051.
CrossRef - Mortimer-Jones, S.M., M.G.K. Jones, R.A.C. Jones, G. Thomson and G.I. Dwyer, 2009. A single tube, quantitative real-time RT-PCR assay that detects four potato viruses simultaneously. J. Virol. Methods, 161: 289-296.
CrossRefDirect Link - Njukeng, P.A., G.M. Chewachong, P. Sakwe, G. Chofong, L.W. Nkeabeng, P. Demo and K.D. Njualem, 2013. Prevalence of six viruses in potato seed tubers produced in informal seed system in the North West Region of Cameroon. Cameroon J. Exp. Biol., 9: 44-49.
Direct Link - Saxena, R. and P. Mathur, 2013. Analysis of potato production performance and yield variability in India. Potato J., 40: 38-44.
Direct Link - Sharma, S., B. Singh, A. Nagpal, G.S. Virk and A.A. Zaidi, 2009. Indexing tools for Indian citrus ringspot virus (ICRSV). Open Biol. J., 2: 27-31.
Direct Link - Singh, R.P., J. Kurz, G. Boiteau and G. Bernard, 1995. Detection of potato leafroll virus in single aphids by the reverse transcription polymerase chain reaction and its potential epidemiological application. J. Virol. Methods, 55: 133-143.
CrossRefDirect Link - Syller, J., 1994. The effects of temperature on the availability and acquisition of potato leafroll luteovirus by Myzus persicae. Ann. Applied Biol., 125: 141-145.
CrossRef - Syller, J., 1996. Potato leafroll virus (PLRV): Its transmission and control. Integr. Pest Manage. Rev., 1: 217-227.
CrossRef - Tiwari, J.K., J. Gopal and B.P. Singh, 2012. Marker-assisted selection for virus resistance in potato: Options and challenges. Potato J., 39: 101-117.
Direct Link








