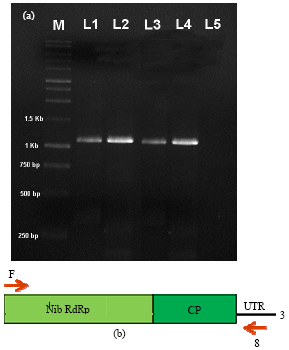
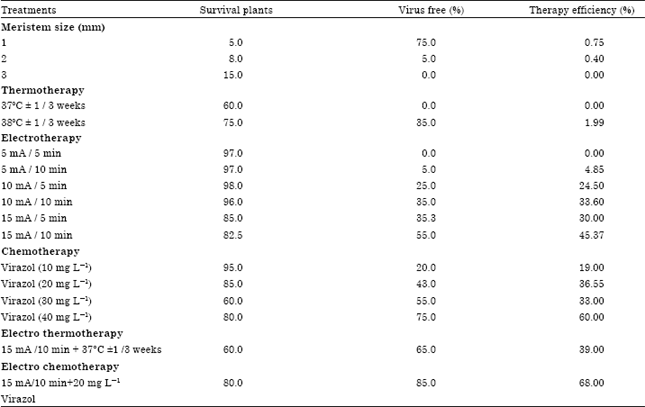
Research Article
Molecular Characterization of Onion Yellow Dwarf Virus (Garlic Isolate) with Production of Virus-free Plantlets
Virus and Phytoplasma Research Section, Plant Pathology Research Institute, Agricultural Research Center (ARC), Giza, Egypt
Sabry Y.M. Mahmoud
Department of Agricultural Botany (Agricultural Microbiology), Faculty of Agriculture, Sohag University, Sohag, Egypt
Rehab A. Dawood
Virus and Phytoplasma Research Section, Plant Pathology Research Institute, Agricultural Research Center (ARC), Giza, Egypt