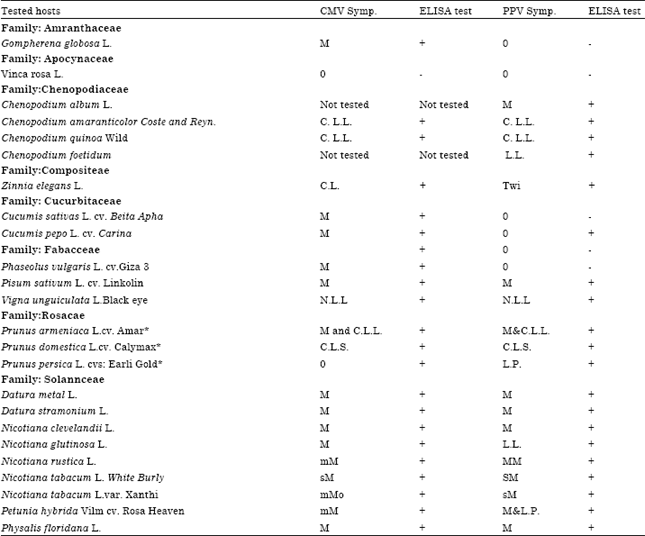
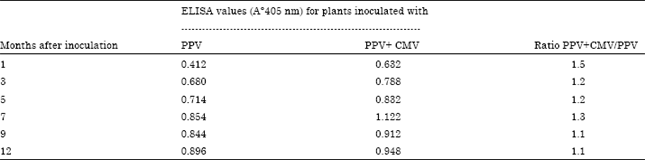
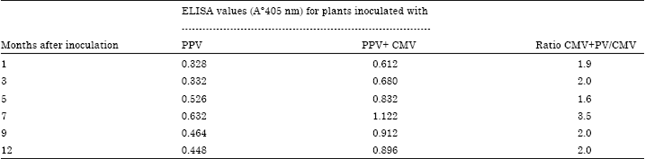
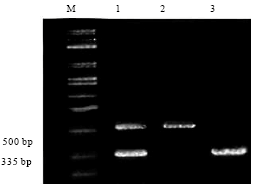
Research Article
Double Infections with Cucumber Mosaic Virus and Plum Pox Virus (Sharka) in Apricot Trees
Department of Virus and Phytoplasma Research, Plant Pathology Research Institute, Giza, Egypt
M.M. Fath-Allah
Department of Virus and Phytoplasma Research, Plant Pathology Research Institute, Giza, Egypt