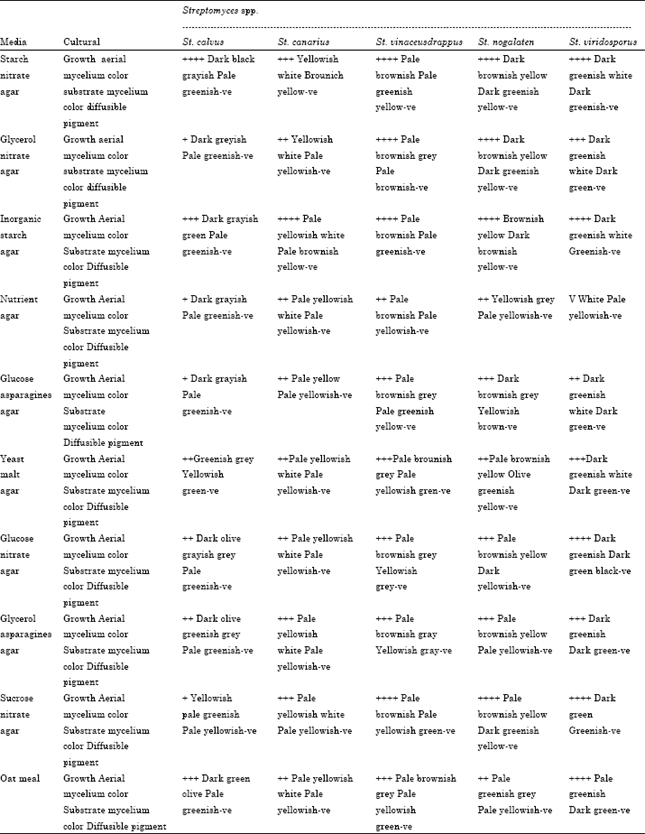
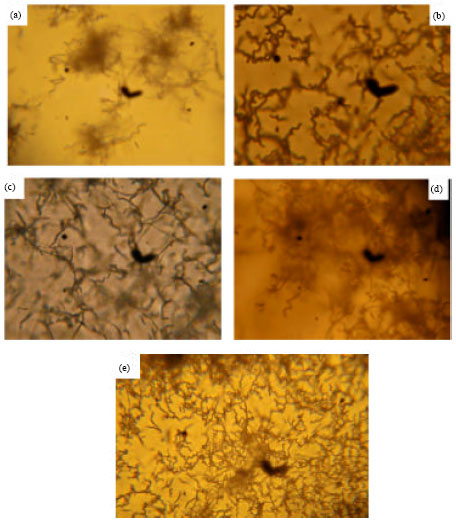
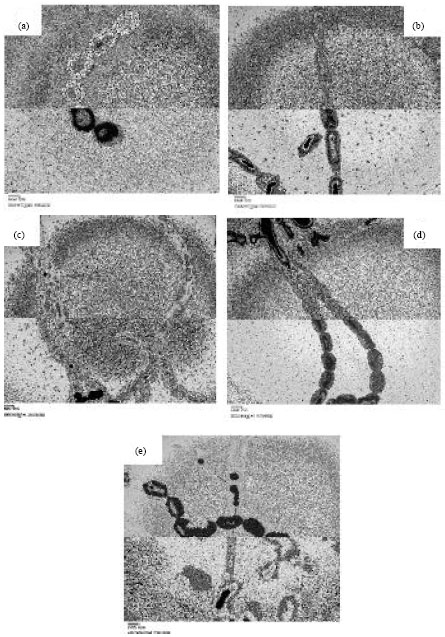
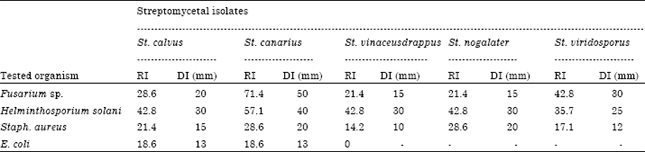
Research Article
Biological Control of Cucumber Mosaic Virus by Certain Local Streptomyces Isolates: Inhibitory Effects of Selected Five Egyptian Isolates
Department of Microbiology, Faculty of Agriculture, Ain Shams University, Egypt
M.F. Ghaly
Department of Botany, Faculty of Science, Zagazig University, Egypt
M.A. Taha
Department of Botany, Faculty of Science, Zagazig University, Egypt