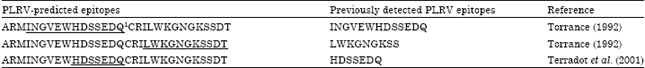
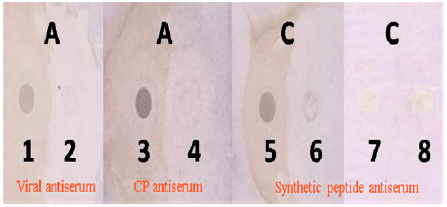
Research Article
Application of Epitopes Prediction for Antibodies Production against Potato Leaf Roll Virus
Agricultural Genetic Engineering Research Institute, Agricultural Research Center, Giza, Egypt
H.A. Nour El-Din
Agricultural Genetic Engineering Research Institute, Agricultural Research Center, Giza, Egypt
W.F. Abd EL-Wahed
Faculty of Computers and Information, Minoufiya University, Shebin El-Kom, Egypt
A.A. Hemeida
Genetic Engineering and Biotechnology Research Institute, Minoufiya University, Sadat City, Egypt