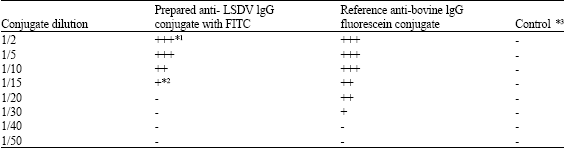
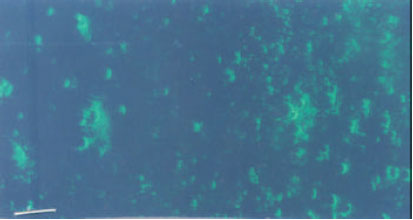
Research Article
Preparation and Evaluation of Lumpy Skin Disease Hyperimmune Serum Coniugated with Fluorescein
Veterinary Serum and Vaccine Research Institute, Abbasia, Cairo, Egypt
B. Omar
Veterinary Serum and Vaccine Research Institute, Abbasia, Cairo, Egypt
Magda M. Sayed
Central Laboratory for Evaluation of Veterinary Biologics, Abbasia, Cairo, Egypt