Research Article
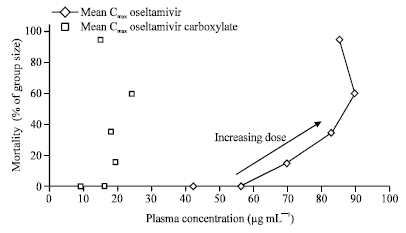
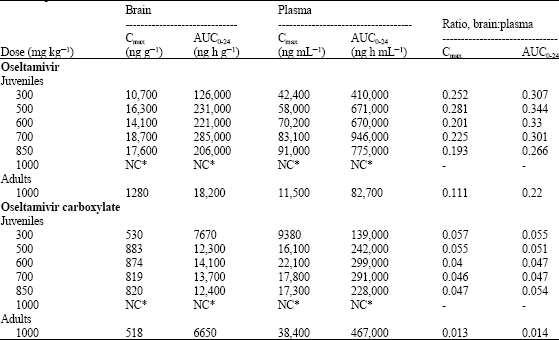
Oseltamivir is Devoid of Specific Behavioral and Other Central Nervous System Effects in Juvenile Rats at Supratherapeutic Oral Doses
Non-Clinical Safety, Pharmaceuticals Division, F. Hoffmann-La Roche Ltd., Basel, Switzerland
E. Prinssen
Discovery Neuroscience, Pharmaceuticals Division, F. Hoffmann-La Roche Ltd., Basel, Switzerland
G. Hoffmann
Non-Clinical Safety, Pharmaceuticals Division, F. Hoffmann-La Roche Ltd., Basel, Switzerland
L. Gand
Non-Clinical Safety, Pharmaceuticals Division, F. Hoffmann-La Roche Ltd., Basel, Switzerland
M. Beck
WIL Research, Ashland, OH, USA
T. Weiser
Non-Clinical Safety, Pharmaceuticals Division, F. Hoffmann-La Roche Ltd., Basel, Switzerland
A. Breidenbach
Non-Clinical Safety, Pharmaceuticals Division, F. Hoffmann-La Roche Ltd., Basel, Switzerland