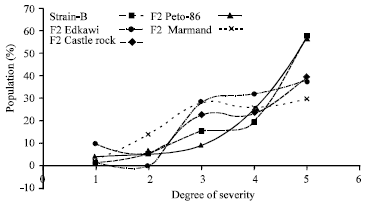
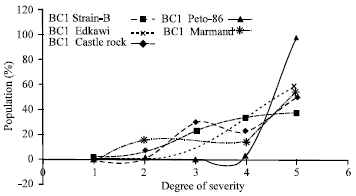
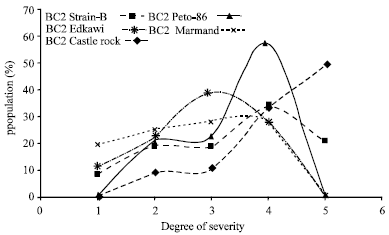
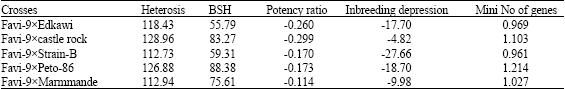
Research Article
Genetic Studies on Tomato Yellow Leaf Curl Begomovirus (TYLCV) Resistance in Egypt: Six-population Analysis
Department of Plant Virus and Phytoplasma Research, Plant Pathology Research Institue, ARC, P.O. Box 1261 9, Giza, Egypt
E. M. Khalil
Department of Vegetable, Horticultural Research Institue, ARC, P.O. Box 12619, Giza, Egypt
A. A. Rezk
Department of Plant Virus and Phytoplasma Research, Plant Pathology Research Institue, ARC, P.O. Box 1261 9, Giza, Egypt
M.A. Abdel- Hakem
Department of Plant Virus and Phytoplasma Research, Plant Pathology Research Institue, ARC, P.O. Box 1261 9, Giza, Egypt
A.E. Aboul- Ata
Department of Plant Virus and Phytoplasma Research, Plant Pathology Research Institue, ARC, P.O. Box 1261 9, Giza, Egypt