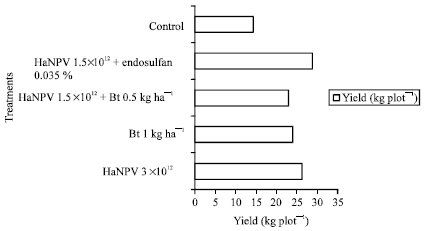
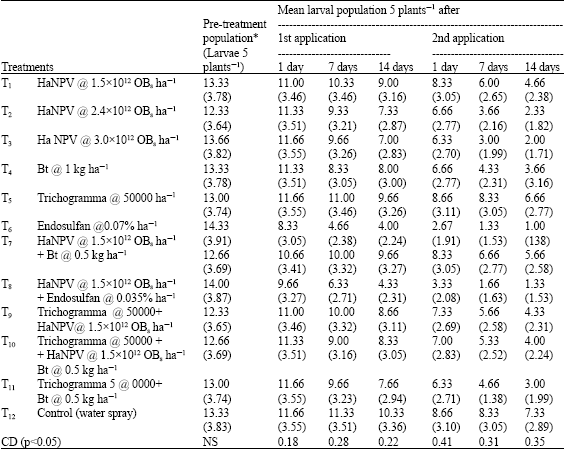
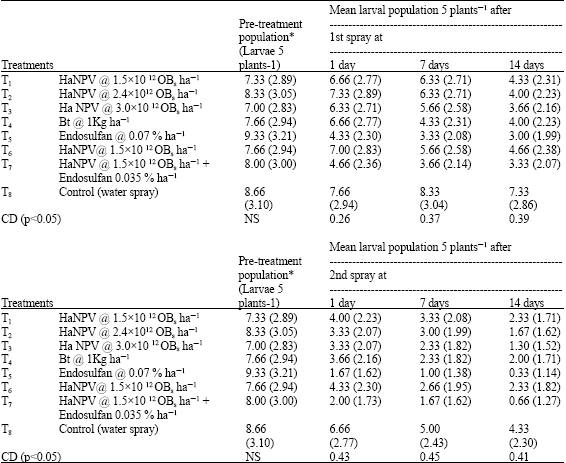
Research Article
Selection and Field Effectiveness of Nucleopolyhedrovirus Isolates Against Helicoverpa armigera (Hubner)
Division of Entomology, Faculty of Agriculture Sher-e-Kashmir University of Agricultural Sciences and Technology, Jammu, 180002, India
J. C. Raina
Division of Entomology, Faculty of Agriculture Sher-e-Kashmir University of Agricultural Sciences and Technology, Jammu, 180002, India
R. K. Arora
Division of Entomology, Faculty of Agriculture Sher-e-Kashmir University of Agricultural Sciences and Technology, Jammu, 180002, India
K. Bali
Division of Entomology, Faculty of Agriculture Sher-e-Kashmir University of Agricultural Sciences and Technology, Jammu, 180002, India